
Reference
Version number
Authorised by
Date
FS01
1
E.Lodge
28.01.11
Copyright Wm Morrison Supermarkets PLC Page 1 of 8
Guidelines for the Generation of
Guidelines for the Generation of
Guidelines for the Generation of
Guidelines for the Generation of
Microbiological
Microbiological
Microbiological
Microbiological Shelf Life Data
Shelf Life Data
Shelf Life Data
Shelf Life Data
Reference
Version number
Authorised by
Date
FS01
1
E.Lodge
28.01.11
Copyright Wm Morrison Supermarkets PLC Page 2 of 8
Version control
Version control
Version control
Version control
AMENDMENT
AMENDMENT
AMENDMENT
AMENDMENT
Version No.
Version No.
Version No.
Version No.
Date
Date
Date
Date
Change
Change
Change
Change
1
28.01.11
New document
INDEX
INDEX
INDEX
INDEX
Page
1. Bought In
4
1.1 Applicable products
4
1.2 Considerations
4
1.3 Sampling
4
1.4 Specific Product Requirements
5
1.5 Frequency of Testing & Sample
Storage
5
1.6Additional Information Requirements 6
1.7 Predictive Models and Challenge
Testing
7
1.8 Organoleptic Shelf Life
7
1.9 Enterprise
7
Appendix 1 – Bought-In Microbiological
Shelf Life
8

Reference
Version number
Authorised by
Date
FS01
1
E.Lodge
28.01.11
Copyright Wm Morrison Supermarkets PLC Page 3 of 8
Aim:
Aim:
Aim:
Aim:
The shelf life of a product is the time after production during which it remains
acceptable for consumption. The shelf life must be set before the product is
launched by either Food Technology or Food Safety Specialist to ensure the product
will, when stored and handled under the recommended conditions:
1) Remain safe
2) Retain its desired sensory, chemical, physical and microbiological
characteristics so it is of an acceptable quality that we are happy to be
associated with
The end of shelf life may be determined by the potential for growth of the micro-
organisms that are likely to be present or able to cross-contaminate the product or
by its organoleptic properties. This will be determined through the product shelf life
study.
Once the initial study has been carried out, it is important that the shelf life of a
product is re-assessed regularly after launch, which for most chilled perishable
products will be at a minimum yearly.
The guideline below details Morrison’s expectations for what should be included in
shelf life studies carried out on both bought-in and in-store prepared products.
However, as it is not possible in a guideline document like this to provide a shelf life
testing protocol that will cover every product type, any products which do not fit
this must be discussed with your Technical Manager so that a protocol can be
agreed to obtain the most realistic shelf life evaluation for that product.
It should also be noted that Morrisons do not accept accelerated shelf life
testing due to the potential inaccuracy of the results – time for a shelf life
study to be carried out over the full life of the product should be built into
the product development schedule for all applicable products. Any
exceptions to this must be agreed with the relevant Technical Manager and
Company Microbiologist.

Reference
Version number
Authorised by
Date
FS01
1
E.Lodge
28.01.11
Copyright Wm Morrison Supermarkets PLC Page 4 of 8
1. Bought
1. Bought
1. Bought
1. Bought----In (including Market Street ingredients)
In (including Market Street ingredients)
In (including Market Street ingredients)
In (including Market Street ingredients)
1.1
1.1
1.1
1.1 Applicable Products
Applicable Products
Applicable Products
Applicable Products
The following products require a microbiological shelf life study to be carried out
before launch:
1) Chilled perishable (e.g. ready meals, pies, sandwiches, bagged salads, etc)
2) Ready-to-Eat frozen products (e.g. ice-cream, cream cakes) or those with >1d
shelf life at <5
0
C post defrost
3) Ambient bakery products (Aw <0.87 then yeasts and moulds only as a
spoilage indicator)
4) Raw meat and poultry
This applies to both bought in products and those made in store
For all other products the shelf life should be determined by its organoleptic
qualities over the shelf life. This must include shelf life of the product once opened
if the customer is recommended to consume the product within a particular time
(e.g. jarred pesto, jam, etc).
1.2
1.2
1.2
1.2 Considerations
Considerations
Considerations
Considerations
The micro-organisms tested should at a minimum be those detailed in Morrison’s
microbiological specification. The following should also be taken into consideration
when determining any additional micro-organisms to test for:
- raw materials used
- product manufacturing process
- key finished product parameters (e.g. pH, Aw)
- finished product packaging
- any preservatives present
- how the customer will handle the product
1.3
1.3
1.3
1.3 Sampling
Sampling
Sampling
Sampling
Key points to note:
a) Samples tested must be from a pre-production run not
not
not
not a kitchen sample to
simulate manufacturing conditions
b) Product tested must be worst case unless with previous agreement from the
relevant Technical Manager – i.e. ingoing ingredients used at their
maximum ingoing shelf life
c) Seasonal variation may need to be taken into account for those products
containing uncooked produce as microbial loading/quality may vary through
the year. If worst case produce is not available at the point of the initial shelf
life trial then the trial should be repeated when available to ensure this
seasonal variation is captured.

Reference
Version number
Authorised by
Date
FS01
1
E.Lodge
28.01.11
Copyright Wm Morrison Supermarkets PLC Page 5 of 8
d) A representative composite sample of the product should be taken
e) In addition, if the product is multi-component or a multipack of different
flavour varieties (e.g. ready meal with separate sauce and vegetables, triple
pack sandwich) then the components should be tested separately and the
maximum shelf life based on the worst-performing component
f) A minimum of 3
3
3
3 different samples are required
1.4
1.4
1.4
1.4 Specific Product Requirements
Specific Product Requirements
Specific Product Requirements
Specific Product Requirements
The following details some specific product requirements:
a) Pizza/Sandwiches/Pies – test topping/filling only
only
only
only
b) Cream desserts -test cream and high-risk components only (e.g. custard)
c) Mould-ripened cheese should be tested for
Listeria
in the rind due to the
elevated pH making it more likely for it to be found here
d) MAP/Gas-Flushed Products – shelf life study should include ‘open life’ of
product recommended to customer on the packaging. This needs to be done
‘worst case’, i.e. last days of total product shelf life
1.5
1.5
1.5
1.5 Frequency of Testing
Frequency of Testing
Frequency of Testing
Frequency of Testing & Sample Storage
& Sample Storage
& Sample Storage
& Sample Storage
The frequency at which microbiological analysis should take place should be based
on the following:
Shelf life 1-4 days: Daily
Shelf life 5 – 14 days: Every 2 days
>14d: Weekly with 3 tests in the final week
Analysis should be carried out up to End of Life + 20% of the total life. Any
variation to the above frequencies must be agreed with the appropriate technical
Manager
The study must take into account the temperatures the product is subjected to
throughout its shelf life, including manufacturing, distribution, display, and
customer handling.
Process Stage
Temperature (
0
C)
Distribution
0 - 5
0
C
Retail
0 - 5
0
C
Customer Purchasing
22
0
C
Customer
8
0
C
Chilled products must be held at 22
0
C for 2hrs to mimic customer purchasing and
then at 8
0
C for minimum 20% of their shelf life (30% if >25d)
Bakery samples should be held at 20
0
C and at 30
0
C for worst-case customer
handling
All results should be submitted on Appendix
All results should be submitted on Appendix
All results should be submitted on Appendix
All results should be submitted on Appendix 2
2
2
2
Reference
Version number
Authorised by
Date
FS01
1
E.Lodge
28.01.11
Copyright Wm Morrison Supermarkets PLC Page 6 of 8
1.6 Additional Information Requirements
1.6 Additional Information Requirements
1.6 Additional Information Requirements
1.6 Additional Information Requirements
The shelf life study only assesses the growth of those micro-organisms which are
likely to be present in or have cross-contaminated the batch of product being
analysed. As it is unlikely that food pathogens will be present in products if good
manufacturing practice has been followed, e.g.
Cl.botulinum
, the effect of the
product formulation on growth on these organisms will not be evaluated during
shelf life studies. This means that shelf life study can not be used to establish
product safety with respect to pathogens. For those products which get an
extended shelf life due to their manufacturing process, packaging or inherent
product parameters details of these need to be included in the Enterprise
specification under ‘Finished Product Parameters’ in the ‘Comment’ box to justify
how the product is safe.
Product Type
Possible Control
Naturally acid/acid formulated chilled
products (e.g. dips, yogurt, salad dressings)
pH
Ready Meals, Soups, Custard
Hot filling temperature
CAPCOLD
Sous Vide
Chilled sponges/jellies
pH
Aw
Cheese
% moisture, pH, % salt
Naturally acid/acid formulated ambient
jars/cans (e.g. pickles, jam, olives)
pH
Low acid ambient jars/cans (e.g. canned
tuna, canned soup)
F
0
Alcohol Creams
% aqueous alcohol
Fresh Pasta
Aw
MAP Bread (e.g. part-baked rolls, naan)
Aw & pH crust & crumb
UHT
Time & Temperature
Cold Smoked Fish
% Aqueous salt
Cooked cured meat/Pate
ppm nitrate/nitrite, % aqueous salt
Fermented Meat
pH
Aw
The values required from the above can be widely found in industry guidelines – for
further detail of any Morrisons specific requirements please contact your Morrisons
Technical Manager.
NB: For all vacuum-packed and MAP products product shelf life must not exceed
10d unless additional controlling factors are in place (as above) or the product has
been put through a predictive model/challenge test (see section 1.7)

Reference
Version number
Authorised by
Date
FS01
1
E.Lodge
28.01.11
Copyright Wm Morrison Supermarkets PLC Page 7 of 8
1.7
1.7
1.7
1.7 Predictive Models and
Predictive Models and
Predictive Models and
Predictive Models and Challenge
Challenge
Challenge
Challenge Testing
Testing
Testing
Testing
Where a product shelf life is required which is longer than the typical safe shelf life
for the product, and there is no intrinsic stability control present (as listed in section
1.6) a predictive model may be used. Predictive models allow the growth rate of the
relevant shelf life limiting microorganisms to be predicted in the product over the
desired life based on its parameters (e.g. pH, Aw, % salt, etc). They are however
limited in that the growth rate will be predicted ‘worst case’ and typically will not
exactly mimic that that would be found in the product as limiting factors such as
competing microorganisms, organism stress, etc, are not taken into account.
However they are a useful tool in assessing whether an atypical shelf life can be
safely achieved or not.
In addition, as it needs to be demonstrated that for all ready to eat chilled foods
able to support the growth of
L. monocytogenes
that if contaminated the organism
will not exceed a level of >100 cfu/g by the end of its shelf life predictive models
can also be used for this.
If, when used, the predictive model does not demonstrate that the requested shelf
life is safe then a challenge test MUST be carried out. Challenge testing is where
food is deliberately inoculated with known microorganisms and/or stored under a
range of controlled environmental conditions in order to determine if those
organisms can grow in the product. It is not quick or simple and cost can potentially
be inhibitive, and so should only be resorted to if all other methods of assessing
safety/stability of the product have failed and if product reformulation/a process
change is not feasible. In these cases, prior agreement must be made with the
appropriate Technical Manager, and the proposed protocol reviewed by the
Company Microbiologist before any work commences who will, if required, seek
outside expert advice.
1.8
1.8
1.8
1.8 Organoleptic Shelf Life
Organoleptic Shelf Life
Organoleptic Shelf Life
Organoleptic Shelf Life
The standards for organoleptic qualities for the product should be determined in
conjunction with both Product Development and Commercial teams, and take place
as part of the NPD process
1.9
1.9
1.9
1.9 Enterprise
Enterprise
Enterprise
Enterprise
Copies of any microbiological or organoleptic shelf life trials carried out must be
attached to the Enterprise specification for that product. Failure to supply the
relevant information will lead to the specification being rejected.
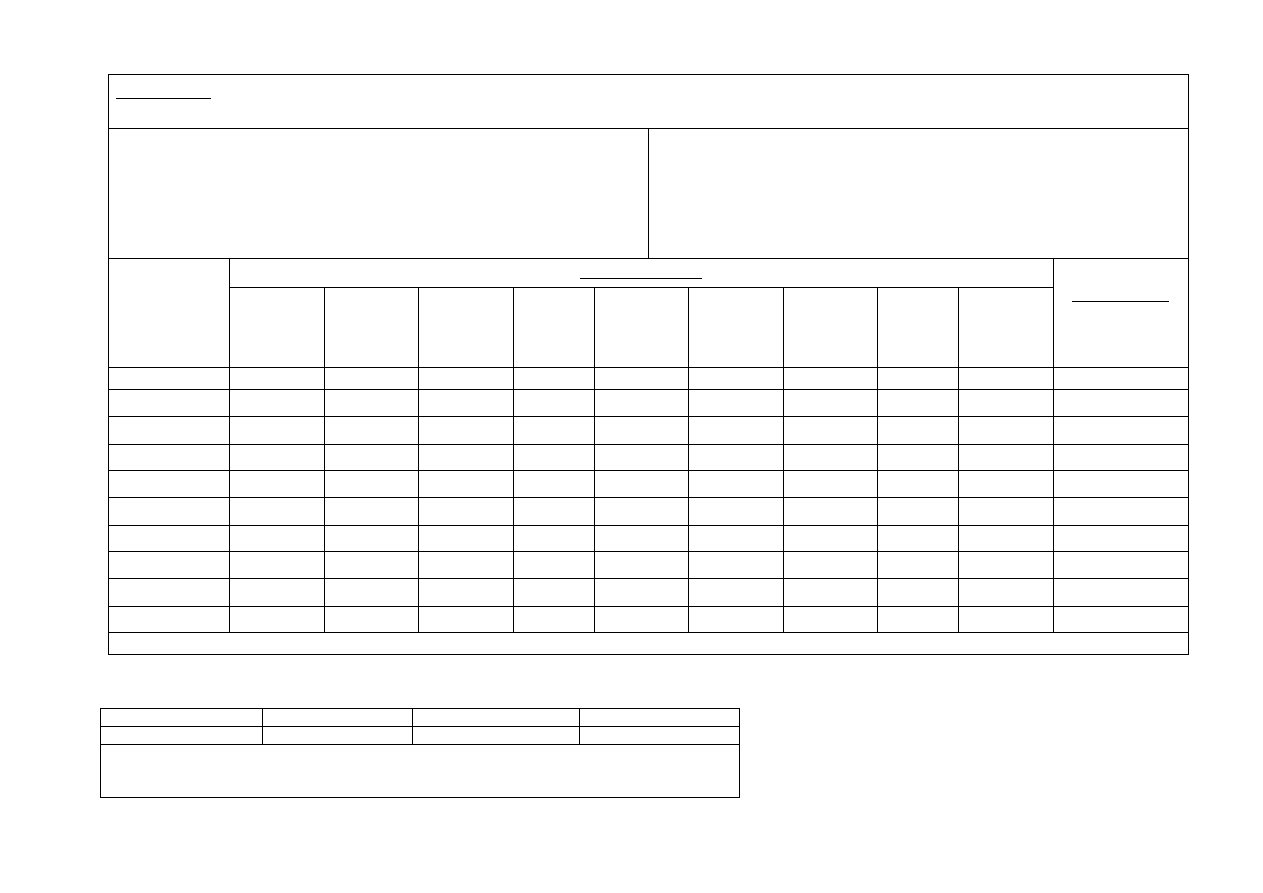
Reference
Version number
Authorised by
Date
FS01
1
E.Lodge
28.01.11
Copyright Wm Morrison Supermarkets PLC Page 8 of 8
APPENDIX 2
APPENDIX 2
APPENDIX 2
APPENDIX 2
Wm Morrisons Supermarkets PLC
Wm Morrisons Supermarkets PLC
Wm Morrisons Supermarkets PLC
Wm Morrisons Supermarkets PLC
Product:
Product:
Product:
Product:
Supplier:
Supplier:
Supplier:
Supplier:
Laboratory:
Laboratory:
Laboratory:
Laboratory:
Storage conditions:
Storage conditions:
Storage conditions:
Storage conditions:
Shelf life required:
Shelf life required:
Shelf life required:
Shelf life required:
Date Tested:
Date Tested:
Date Tested:
Date Tested:
Time (Day,
week, etc)
Micro
Micro
Micro
Micro----Organism
Organism
Organism
Organism
Temperature
Temperature
Temperature
Temperature
(
0
C)
TVC
(cfu/g)
0
4
0
C
1
2
3
8
0
C
Approved shelf life:
Approved shelf life:
Approved shelf life:
Approved shelf life:
All microorganisms tested should be as Morrison’s Microbiological Testing Specifications
Wyszukiwarka
Podobne podstrony:
APA practice guideline for the treatment of patients with Borderline Personality Disorder
Guidelines for the Management of Aneurysmal Subarachnoid Hemorrhage
GUIDELINES FOR THE APPROVAL OF FIXED WATER BASED LOCAL APPLICATION
Tips and improvements for the general occultist
DESCRIPTION FOR THE GENERAL PUBLIC
Consistency Proof for the Generalized Continuum Hypothesis
British Patent 2,812 Improvements in Methods of and Apparatus for the Generation of Electric Current
ESTRO BOOKLET 5 Practical guidelines for the impletation of in vivo dosimetry with diodes in extern
guidelines for the content of rig move procedures sept 2008
CSharp Introduction to C# Programming for the Microsoft NET Platform (Prerelease)
AMENDMENTS TO THE REVISED GUIDELINES FOR APPROVAL OF SPRINKLER SYSTEM
Vlaenderen A generalisation of classical electrodynamics for the prediction of scalar field effects
Rewicz, Tomasz i inni Isolation and characterization of 8 microsatellite loci for the ‘‘killer shri
Howe Counterexample to the generalized Ramanujan conjecture for quasi split groups (1979) [sharethe
Microsoft Word Spectre of the Black Rose Sam
więcej podobnych podstron