NOP http://www.oc-praktikum.de
July
2005
1
1021 Isolation of trimyristin from nutmeg
Classification
Reaction types and substance classes
isolation of natural products
carboxylic acid ester, triglyceride, natural product
Work methods
extracting with Soxhlet extractor, evaporating with rotary evaporator, recrystallizing, filtering,
heating under reflux, stirring with magnetic stir bar, heating with oil bath
Instruction (batch scale 25 g)
Equipment
250 mL round-bottom flask, 100 mL Soxhlet extractor with extraction sleeve, reflux
condenser, heatable magnetic stirrer with magnetic stir bar, rotary evaporator, suction flask,
suction filter, desiccator, oil bath
Substances
nutmeg,
finely
ground 25
g
tert-butyl methyl ether (bp 55 °C)
150 mL
ethanol
(bp
78
°C)
etwa
150
mL
Reaction
The reaction apparatus consists of a 250 mL round-bottom flask with a magnetic stir bar and a
100 mL soxhlet extraction unit with a reflux condenser. 25 g of finely ground nutmeg are
placed into the extraction sleeve and covered with a little glass wool. 150 mL tert-butyl
methyl ether are placed into the flask and whilst stirring, the solvent is heated to reflux until
the solvent leaving the extraction sleeve is colourless (approximately 5 hours).
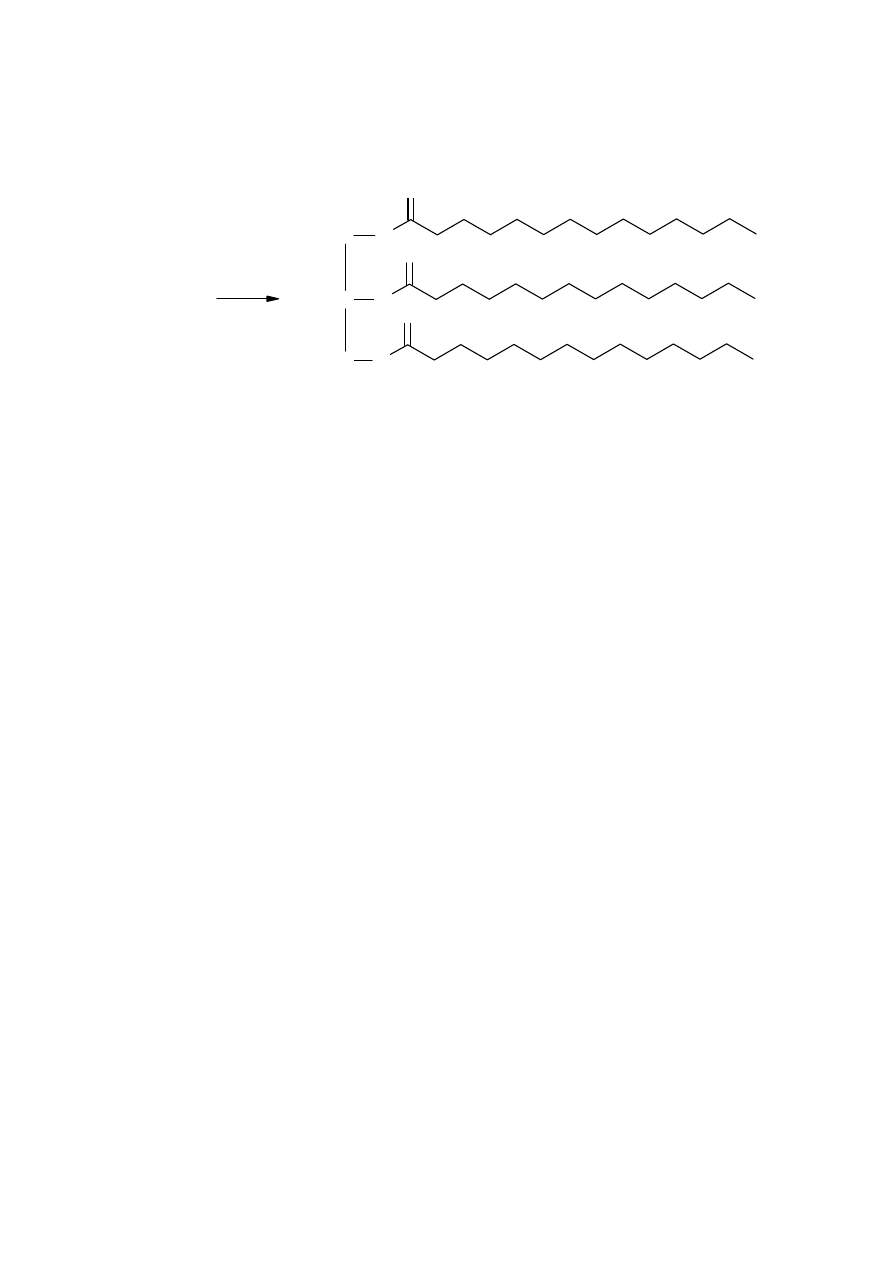
O
O
O
C
H
2
C
H
C
H
2
O
O
O
Nutmeg
C
45
H
86
O
6
(723.2)
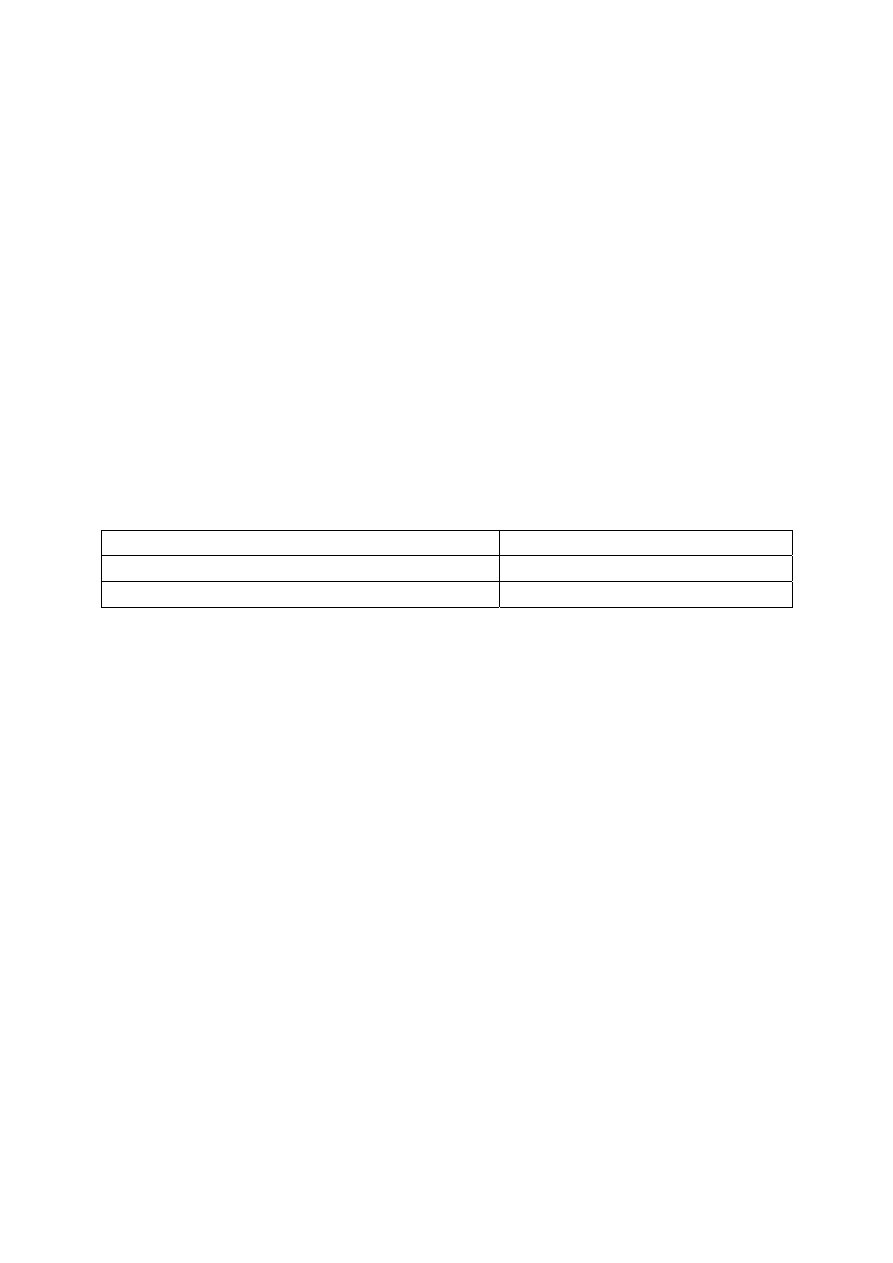
NOP http://www.oc-praktikum.de
July
2005
2
Work up
The solvent is evaporated with a final pressure of 20 hPa. The flask containing the residue is
cooled in an ice bath or the refrigerator until the contents has crystallized to a thick slurry.
Yield crude product: 12 g
The crude product is recrystallized from the minimum amount of ethanol. Prior to filtering the
crystals, the flask is placed into the refrigerator for at least 30 minutes. The crystalline slurry
is filtered and the product is dried in an evacuated desiccator over silica gel. Should the
crystals not be colourless after the first recrystallization, a second recrystallization is carried
out.
Yield 6.5 g (26% according to the amount of used nutmeg); mp 54-55 °C.
Waste management
Recycling
The evaporated tert-butyl methyl ether and the evaporated ethanol from the mother liquor are
collected and redistilled.
Waste disposal
Waste
Disposal
residue from extraction
domestic waste
residue from mother liquor
domestic waste
Time
Without recrystallization 6 hours
Break
Before and after the evaporation of the solvent
Degree of difficulty
Easy
Analytics
TLC
TLC conditions:
adsorbent:
Macherey and Nagel Polygram SilG/UV plates, 0.2 mm
elution solvent:
cyclohexane/ethyl acetate 9.5: 0.5
visualizing agent:
Vaughn`s Reagent or iodine vapour
(Vaughn’s Reagent consists of 45 mL water, 5 mL conc. sulphuric acid, 2.4 g
ammoniumheptamolybdate tetrahydrate((NH
4
)
6
Mo
7
O
24
·4H
2
O) and 0.1 g Ce(SO
4
)
2
.)
(Iodine vapour: The dried TLC foil is placed into a vessel containing a few iodine
crystals. The vessel is closed and the iodine crystals are heated with the heat gun until an
iodine vapour forms and the substance spots become visible).
R
f
(trimyristin):
0.51
NOP http://www.oc-praktikum.de
July
2005
3
1
H NMR spectrum of the pure product (300 MHz, CDCl
3
)
(ppm)
1.0
2.0
3.0
4.0
5.0
6.0
7.0
(ppm)
5.25
(ppm)
4.10
4.20
(ppm)
0.8
δ (ppm)
Multiplicity Number
of H
Assignment
0.90
m
9
14-H, 28-H
1.2-1.4
m
60
4-H up to 13-H, and 18-H up to 27-H
1.5-1.7
m
6
3-H, 17-H
2.33
m
6
2-H, 16-H
4.16
dd
2
30-H
a
4.31
dd
2
30-H
b
5.28
m
1
29-H
7.26
solvent
2.11
acetone
(impurity)
O
O
O
C
C
H
C
O
O
O
H
H
H
H
1
3
5
7
9
11
13
2
4
6
8
10
12
14
1
3
5
7
9
11
13
2
4
6
8
10
12
14
15
17
19
21
23
25
27
16
18
20
22
24
26
28
29
30
30
a
a
b
b
NOP http://www.oc-praktikum.de
July
2005
4
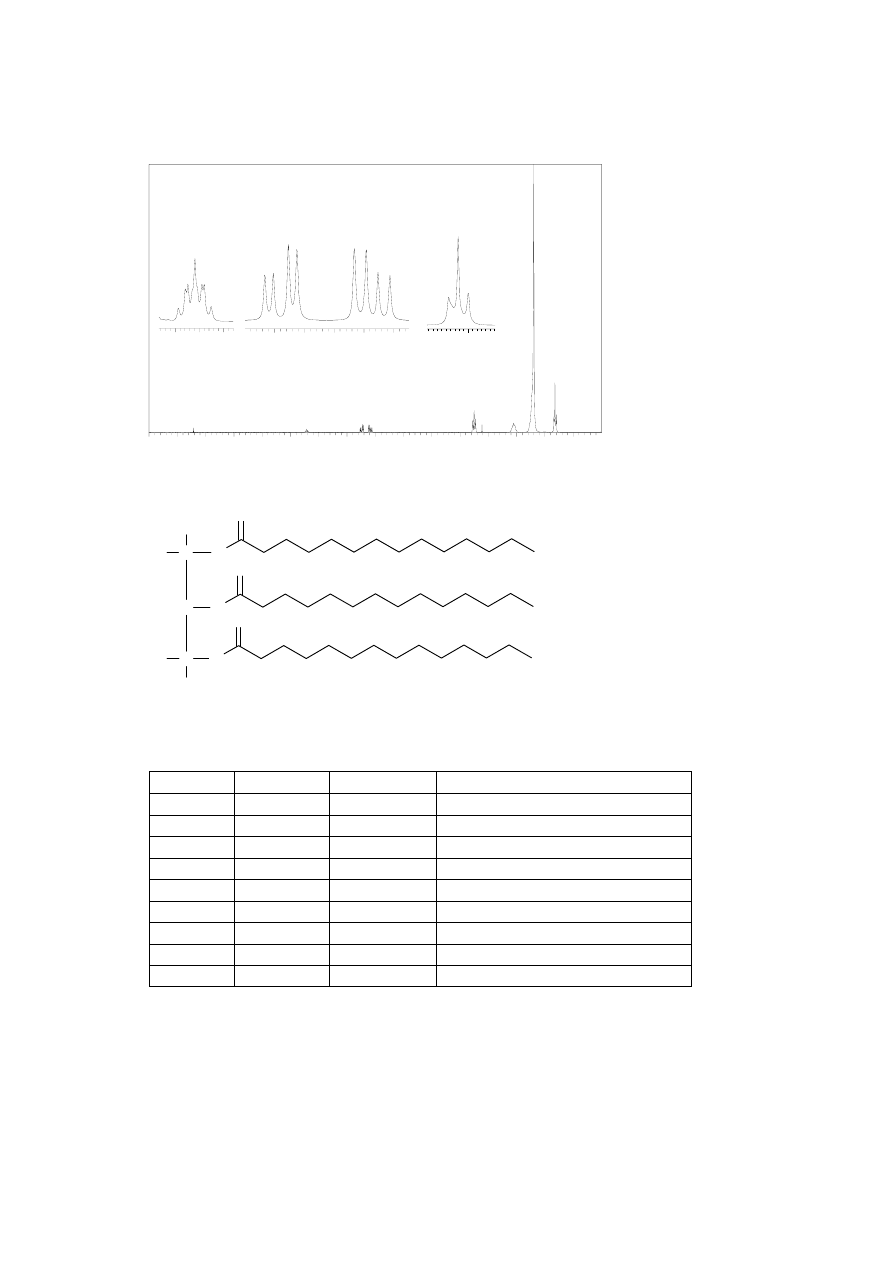
13
C NMR spectrum of the pure product (300 MHz, CDCl
3
)
(ppm)
20
40
60
80
100
120
140
160
O
O
O
C
C
H
C
O
O
O
H
H
H
H
1
3
5
7
9
11
13
2
4
6
8
10
12
14
1
3
5
7
9
11
13
2
4
6
8
10
12
14
15
17
19
21
23
25
27
16
18
20
22
24
26
28
29
30
30
a
a
b
b
δ (ppm)
Assignment
14.08
C-14, C-28
22.66
C-13, C-27
24.85, 24.89
C-3, C-17
29.06-31.90
C-4 up to C-12 and C-18 up to C-26
34.04, 34.20
C-2, C-16
62.08
C-30
68.85
C-29
172.85,
C-15
173.26 C-1
76.5-77.5
solvent
NOP http://www.oc-praktikum.de
July
2005
5
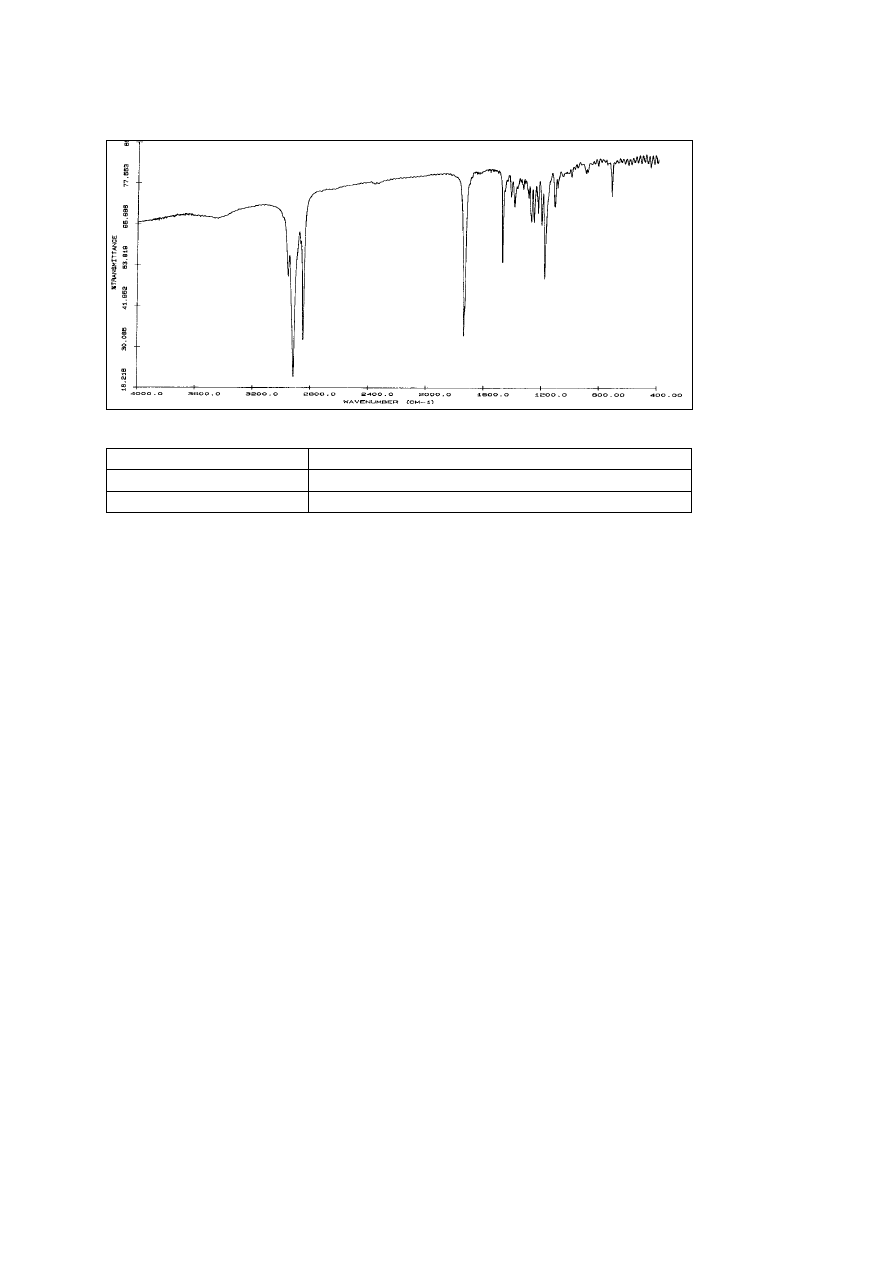
IR spectrum of the pure product (KBr)
(cm
-1
) Assignment
2950 - 2850
C – H – valence, alkane
1730
C = O – valence, ester
Document Outline
Wyszukiwarka
Podobne podstrony:
Isolation of lycopene from crude tomato extract via selective inclusion in deoxycholic acid
Morimoto, Iida, Sakagami The role of refections from behind the listener in spatial reflection
Idea of God from Prehistory to the Middle Ages
Manovich, Lev The Engineering of Vision from Constructivism to Computers
Isolation of C glabrata homologs of the S cerevisiea
Characteristics, treatment and utilization of residues from MSW
Electron microprobe dating of monazites from Western Carpathian
Russian and Chinese Methods of going from Communism to?moc
Production of Energy from Biomass Residues 020bm 496 1993
A Content Analysis of Magazine?vertisements from the United States and the Arab World
108 Pieces of?vice From Lahiri Mahasay
Morimoto, Iida, Sakagami The role of refections from behind the listener in spatial reflection
Kosky; Ethics as the End of Metaphysics from Levinas and the Philosophy of Religion
Latour, Bruno Why crtique run of steam From matters of fact to matters of concern
The Battle of Hattin from a Muslim source
więcej podobnych podstron