scan0016 (7)

345
PETROLEUM AND INDMDUAL POLYCYCLIC AROMATIC HYDROCARBONS
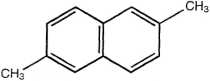
2,6-Dimethylnaphthalene
CH3
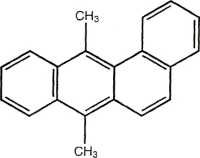
7,12-Dimethylbenz[a]anthracene
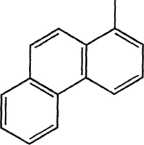
1 -Methylphenanthrene
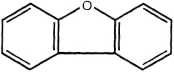
Dibenzofuran
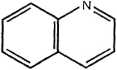
Quinoline
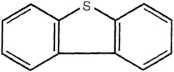
NH,
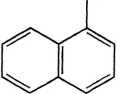
1-Naphthylamine
Dibenzothiophene
Figurę 14.3 Examples of PAH compounds with attached alkyl groups and aromatic compounds with nitrogen, sulfur, or oxygen.8 Except for the alkyl groups, hydrogen atoms bonded to carbon atoms are omitted.
14.3.2 PAHs
Most PAHs are formed by a process of thermal decomposition (pyrolysis) and subseąuent recombination (pyrosynthesis) of organie molecules.16 Incomplete combustion of organie matter produces PAHs in a high-temperature (500 to 800°C) environment. Ali forms of combustion, except flammable gases well mixed with air, produce some PAHs. Subjection of organie materiał in sediments to a low-temperature (100 to 300°C) environment for long periods of time produces PAHs as coal and oil deposits within sedimentary rock formations (a.k.a. diagenesis).7-17 Although the PAH compounds formed by high- and low-temperature processes are similar, the occurrence of alkylated PAHs is greater in the low-temperature process.14 Biological formation of PAHs occurs in chlorophyllous plants, fungi, and bacteria.28
Natural sources of PAHs include forest and grass fires, oil seeps,; yolcanoes, plants, fungi, and bacteria. Anthropogenic sources of PAHs include petroleum, electric power generation, refuse incineration, and home heating; production of coke, carbon black, coal tar, and asphalt; and intemal combustion engines.2’7 The primary mechanism for atmospheric contamination by PAHs is incom-
Wyszukiwarka
Podobne podstrony:
scan0020 (6) 349 PETROLEUM AND INDMDUAL POLYCYCLIC AROMATIC HYDROCARBONS Oil spilled on land has lit
scan0018 (7) PETROLEUM AND INDMDUAL POLYCYCLIC AROMATIC
scan0017 (7) 346 HANDBOOK OF ECOTOXICOLOGY Figurę 14.4 Sources of petroleum and PAHs in the environm
39447 scan0025 (4) Volume 34/Numbcr ll/November 1997 <3ffl -3 to-1 Om Fig. 1 Comparison of polycy
ivwiv^i V>llV^O JLJl ISillSgl CłJ/lilljUlO Wakeham S.G., Schaffner C., Giger W., 1980a. Polycycli
46894 str (86) lob BIBLIOGRA PH Y PnxoK. J. Ind. Eng. Cham., 1934. XXVI., 345. i tciKS and Thompson.
UHAM095 78 ^ UNDERSTANDING HEADACHES AND M I G RAI N ES Aromatherapy: There are many aromatherapy as
więcej podobnych podstron