
Optimal Management
of HBV:
A Partnership
Between
Primary Care and
Specialty Physicians

Primary Care
Specialist
Screening
Diagnostic testing
Identification of treatment
candidates
Initiation of treatment
On-treatment monitoring
Long-term follow-up for
disease activation
HCC screening
Vaccination

Hepatitis B:
An Important Disease
for Primary Care and
Specialty Physicians

Why Is HBV Relevant in
Primary Care?
High global impact
–
High prevalence in specific risk
groups
–
Risk of death/cancer
Effective prevention
Effective treatment
–
Improve liver disease outcomes
–
Decrease progression to cirrhosis
or HCC and improve survival

1. WHO. Hepatitis B. 2002. 2. Maynard JE, et al. In: Viral Hepatitis and
Liver Disease. New York: Alan R. Liss, Inc. 1988.
3. CDC. Epidemiology & Prevention of Vaccine-Preventable Diseases. “The Pink
Book.” 8th ed. 4. CDC. MMWR. 2001;50:
RR-11.
Global Impact of HBV
A Significant Cause of
Worldwide Morbidity
and Mortality
>2 billion have been infected
1
4 million acute cases per year
1
1 million deaths per year
1
350 million chronic carriers
1
–
25% of carriers die from chronic active
hepatitis, cirrhosis, or liver cancer
1
–
Nearly 75% of chronic carriers are Asian
2
2nd most important carcinogen behind
tobacco
3
Causes 60%–80% of all primary liver cancer
1
HBV is 100 times more contagious than HIV
4
HBV
A Global Health Problem
1. WHO. Hepatitis B. 2002. 2. Custer B. et al. J Clin Gastroenterol.
2004;38(10 suppl):S158. 3. WHO/WPRO data.
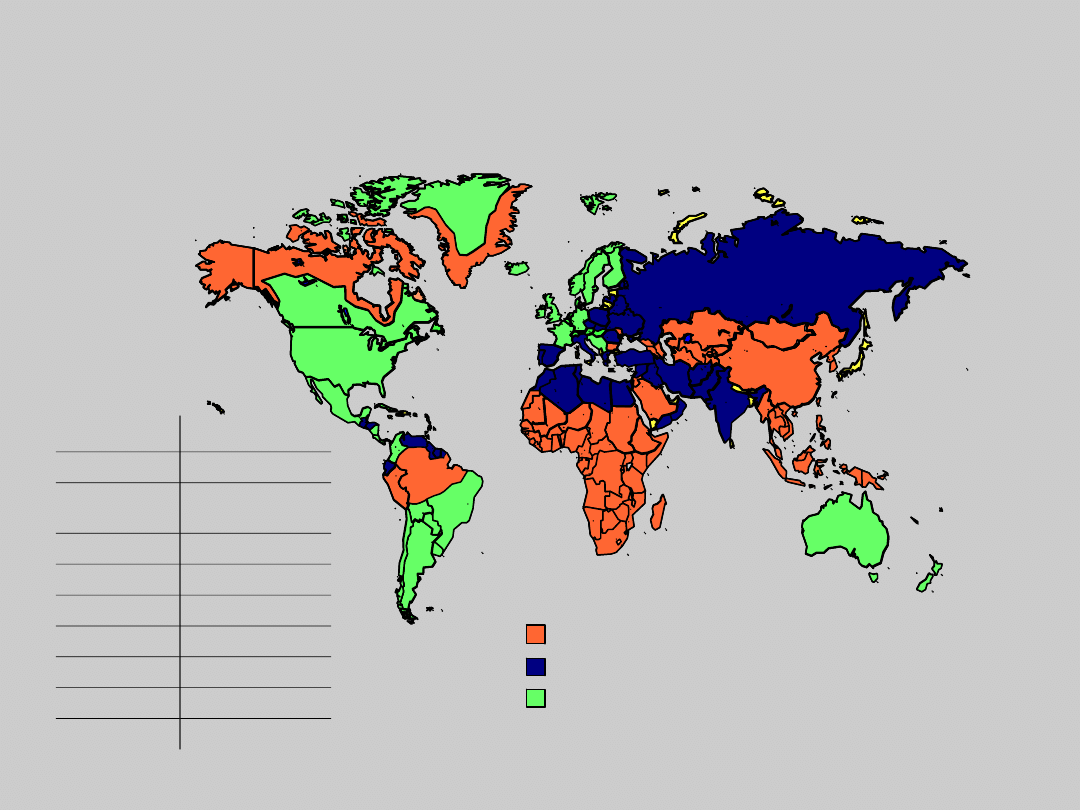
HBsAg Prevalence (%)
1
<8: High
2–8: Intermediate
<2: Low
Country
HBsAg+ (%)
China
5.3–12
2
South
Korea
2.6–5.1
2
India
2.4–4.7
2
Taiwan
10–13.8
2
Viet Nam
5.7–10
2
Japan
4.4–13
3
Africa
5–19
2
Russia
1.4–8
2
Europe
0.3–12
2
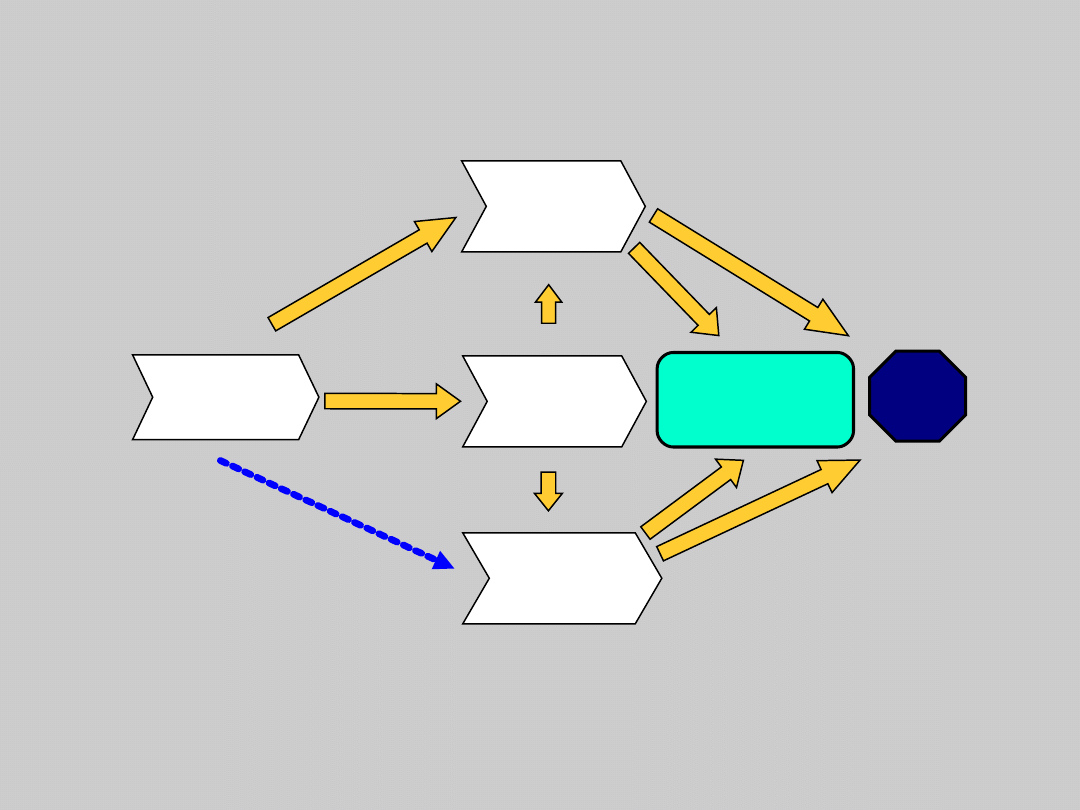
HBV Disease Progression
Chronic
Infection
Cirrhosis
Death
Torresi J, et al. Gastroenterology. 2000;118:S83. Fattovich G, et al.
Hepatology. 1995;21:77.
Perrillo RP, et al. Hepatology. 2001;33:424.
Liver
Failure
Liver
Cancer
(HCC)
Liver
Transplanta
tion
Burden of HBV Infection
in the United States
New Acute Infections
per Year
1
73,000
Chronic
Infections
1
1,250,000
Deaths per
Year
1
5000
Liver Transplants
per Year
3
230
HCC per
Year
2
3100
1. CDC. HBV Disease Burden. 2005. 2. El-Serag HB, et al. Arch Intern Med.
2000;160:3227. 3. UNOS/OPTN.
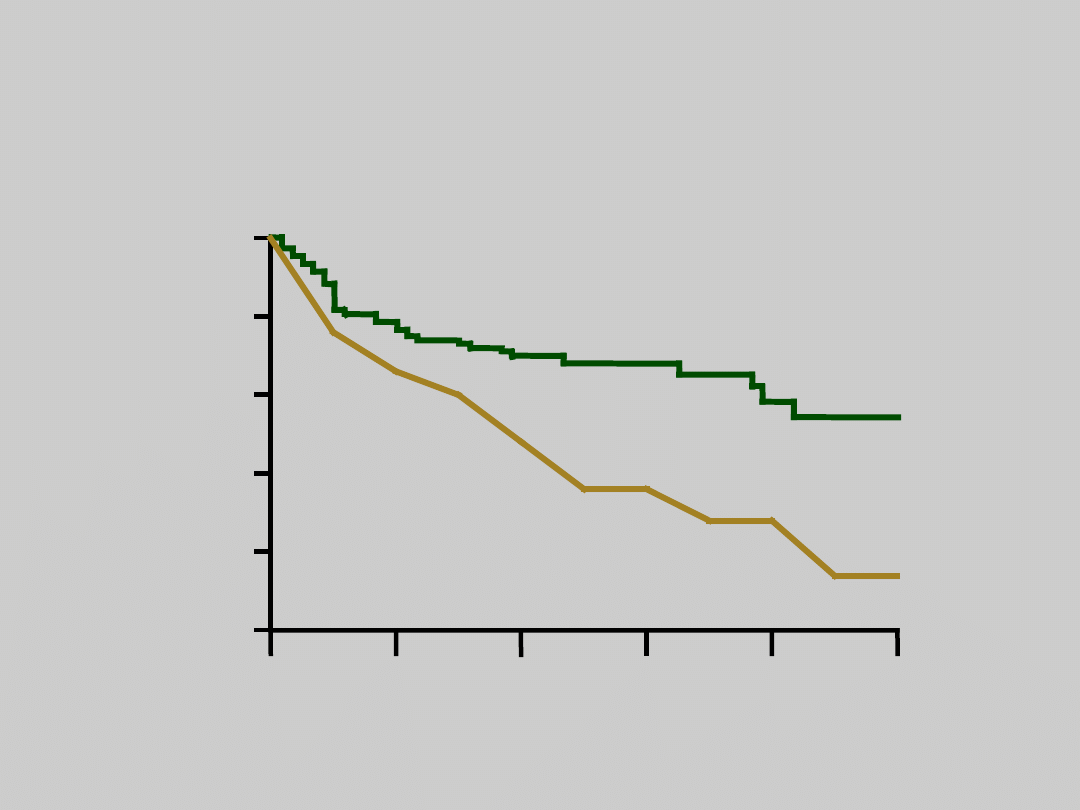
Poor Survival in
HBV-Related Cirrhosis
1. Weissberg JI, et al. Ann Intern Med. 1984;101:613. 2. De Jongh FE, et al.
Gastroenterology. 1992;103:1630.
1
3
2
4
5
0
2
0
4
0
60
10
0
8
0
Cirrhosis
1
(n = 130)
Decompensated Cirrhosis
2
(n = 21)
14%
14%
55%
P
a
t
i
e
n
t
s
S
u
r
v
i
v
i
n
g
(
%
)
Years
0

HBV Is Preventable
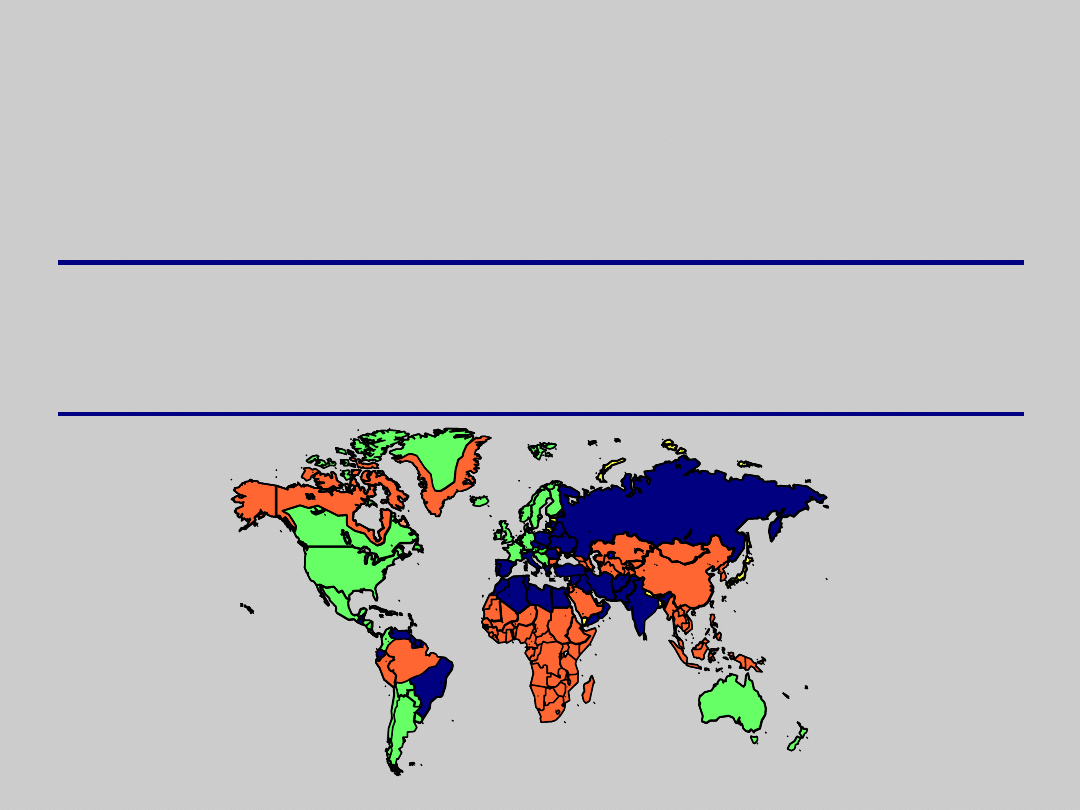
Clinical-Epidemiologic
Correlations
Age of
Mode of
ESLD
HCC
Endemicity Location
Infection Transmission Chronicity
Risk
Risk
Low
N. America
Early
Percutaneous
Rare
Low
Low
W. Europe Adulthood
Sexual
High
Sub-Sahara
Birth
Perinatal
Likely
High
High
Far East
Toddler
Horizontal

CDC. MMWR. 1991;40(RR-13):1.
HBV Vaccine
Indications
Routine vaccination of infants
–
Regardless of mother’s HBsAg status
–
With HBIG for HBsAg positive mothers
Catch-up vaccination of children and
adolescents
Vaccination of adults with risk
factors for infection
–
High-risk sexual activity
–
Illegal injection drug use
–
Occupational exposure
–
Hemodialysis patients
–
Household contacts of infected
persons
HBIG = hepatitis B immune
globulin.
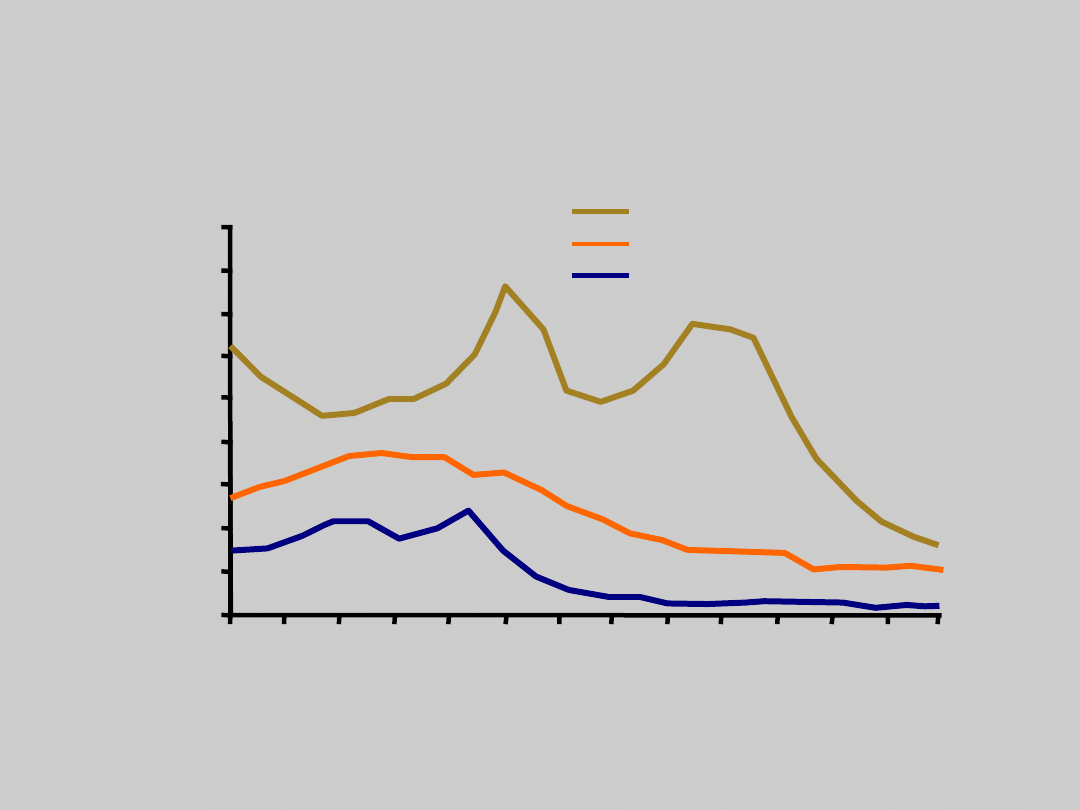
US Incidence of Viral
Hepatitis Infection Over
Time
CDC. Disease burden from viral hepatitis A, B, and C in the United
States. 2003.
Incidence of Hepatitis A, US
Incidence of Acute Hepatitis B, US
Incidence of Acute Hepatitis C, US
0
20
40
60
80
100
120
140
160
180
1990
1992
1994
1996
1998
2000
2002
2003
E
s
t
i
m
a
t
e
d
N
u
m
b
e
r
o
f
C
a
s
e
s
(
T
h
o
u
s
a
n
d
s
)
Year
1991
1993
1995
1997
1999
2001
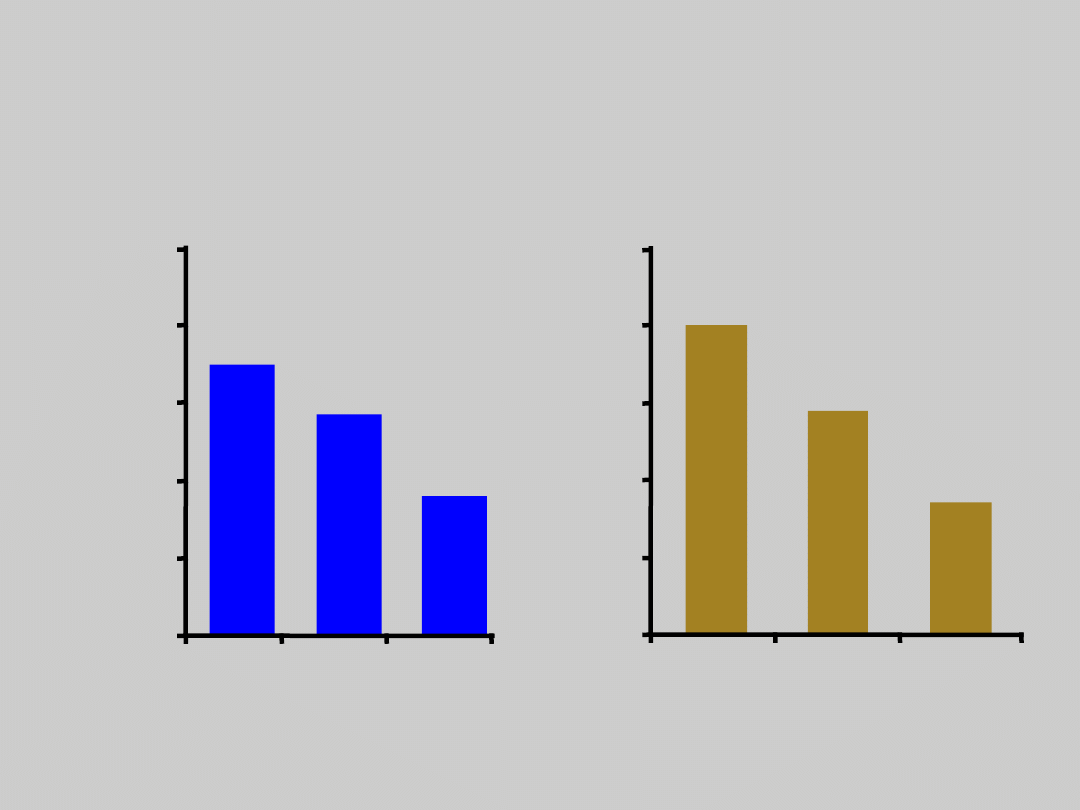
HBV Vaccination
Effect on HCC Incidence and
Mortality*
Chang M-H, et al. N Engl J Med. 1997;336:1855.
*Nationwide vaccination in Taiwan, implemented 7/84
Incidence
0
0.2
0.4
0.6
0.8
1
1981–86
1986–90
1990–94
P
e
r
1
0
0
,
0
0
0
C
h
i
l
d
r
e
n
(
6
–
1
4
Y
e
a
r
s
)
0.7
0
0.57
0.36
P
e
r
1
0
0
,
0
0
0
C
h
i
l
d
r
e
n
(
6
–
1
4
Y
e
a
r
s
)
Mortality
0
0.2
0.4
0.6
0.8
1
1981–86
1986–90
1990–94
0.80
0.58
0.34

Screening for HBV
and Evaluating
Infected Patients

Who Should Be Screened?
Patients with abnormal ALT
Patients engaged in high-risk sexual behaviors
Injection drug users
Immigrants, refugees, or adoptees from areas of high
endemicity
Immunocompromised patients
Dialysis patients
Recipients of organ/tissue transplants or blood
transfusion
Household members or sexual partners of known HBV
carriers
Occupational exposure (healthcare workers, police, EMTs)
Inmates in long-term correctional facilities or residents
in institutions for the developmentally disabled
Pregnant women
Individuals infected with HCV or HIV
Adapted from CDC. Epidemiology & Prevention of Vaccine-Preventable Diseases.
“The Pink Book.” 8th ed, 2005.
Lok ASF, et al. Hepatology. 2001;34:1225.
Lok ASF, et al. Hepatology. 2001;34:1225. Reprinted with permission of Wiley-
Liss, Inc, a subsidiary of John Wiley & Sons, Inc.
Prevalence of HBV Serologic
Markers in
Population Groups Who
Should Be Tested for HBV
Infection
Prevalence of HBV
Serologic Markers (%)
Population
HBsAg
Any Marker
Persons born in high endemic areas*
13 70–85
Men who have sex with men
6
35–80
Injection drug users
7
60–80
Dialysis patients
3–10
20–80
HIV infected patients
8–11
89–90
Pregnant females (USA)
0.4–1.5
Family/household and sexual contacts
3–6 30–60
*Africa; Southeast Asia, including China, Korea, Indonesia,
and the Philippines; the Middle East,
except Israel; South and Western Pacific Islands; the interior
Amazon River basin; and certain
parts of the Caribbean (Haiti and the Dominican Republic)

Screening Tests
HBsAg
–
If positive, indicates
infection
Anti-HBc
–
If positive, indicates HBV
exposure
Anti-HBs
–
If positive, indicates
immunity

Lok ASF, et al. Hepatology. 2001;34:1225. Tsai NCS, et al. Semin Liver
Dis. 2004;24(suppl 1):71.
History and Physical
Evaluation
Risk factors for coinfection
Alcohol use
Family history of HBV
infection and HCC
Physical findings of advanced
disease
–
Jaundice
–
Abdominal swelling
–
Upper GI bleeding

Assessment of
Liver Disease Severity
Liver disease activity, biochemical
–
ALT
–
AST
Liver function/synthetic testing
–
Albumin
–
Bilirubin
–
Prothrombin time (INR)
Ultrasonography: morphologic assessment
–
Liver size, contour, and
echogenicity
–
Splenomegaly
Lok ASF, et al. Hepatology 2001;34:1225. Tsai NCS, et al. Semin Liver Dis. 2004;24(suppl 1):71.
Keeffe EB, et al. Clin Gastroenterol Hepatol. 2004;2:87.

2nd-Phase Testing
in HBsAg+ Patients
Anti-HBc
–
IgG: if positive, indicates HBV exposure
–
IgM: if positive, indicates acute HBV
HBeAg
–
If positive, indicates active HBV replication
–
If negative
If HBV DNA negative, suggests HBV
replication is suppressed
If HBV DNA positive, most likely has
precore mutation
Anti-HBe
HBV DNA quantification
–
Quantitative level correlates with level of
HBV replication

Further Testing
for HBsAg+ Patients
HCV antibody in at-risk individuals
HIV antibody or RNA quantification
in
at-risk individuals
Consider screening for hepatitis
delta virus (HDV)
if adult-acquired HBV
–
Anti-HDV
–
Delta antigen
–
Delta RNA if any HDV testing is
available

Hepatitis Delta Virus (HDV)
Outcome
Clinical Scenario
Risk of cirrhosis
and cancer
Chronic HDV/chronic HBV
Risk of progressive
liver
disease
Acute HDV/chronic HBV
Risk of acute
liver disease
Acute HDV/acute HBV

Who Are Treatment
Candidates?
Natural History of HBV
Infection
Inactiv
e
Carrier
<5%
HBeAg+
Chronic
Hepatit
is B
Adultho
od
Courtesy of W. Ray Kim, MD
Chen DS, et al . J. Gastroenterol Hep. 1993:8(5):470.
Seeff L, et al. N Engl J Med. 1987;316(16):965.
Immune
Toleran
ce
Cirrhosis
Early
Childho
od
>95%
HBeAg-
Chronic
Hepatitis B

Phases of Chronic HBV
Infection
*Precore mutant
†
Expert opinions vary as to this value
1IU = ~5 copies/mL
Lai CL, et al. Lancet. 2003:362:2089. Lok AS, et al. Gastroenterology.
2001;120:1828.
Immune Inactive HBeAg
HBeAg
Tolerant
HBsAg Positive
Negative
Phase
Carrier
CHB
CHB*
HBsAg
+
+
+
+
HBeAg
+
–
+
–
Anti-HBe
–
+
–
+
ALT
Normal
Normal
HBV DNA
>10
5
>10
4
<10
5
>10
4†
copies/mL copies/mL copies/mL copies/mL
HistologyNormal/Mild Inactive
Active
Active
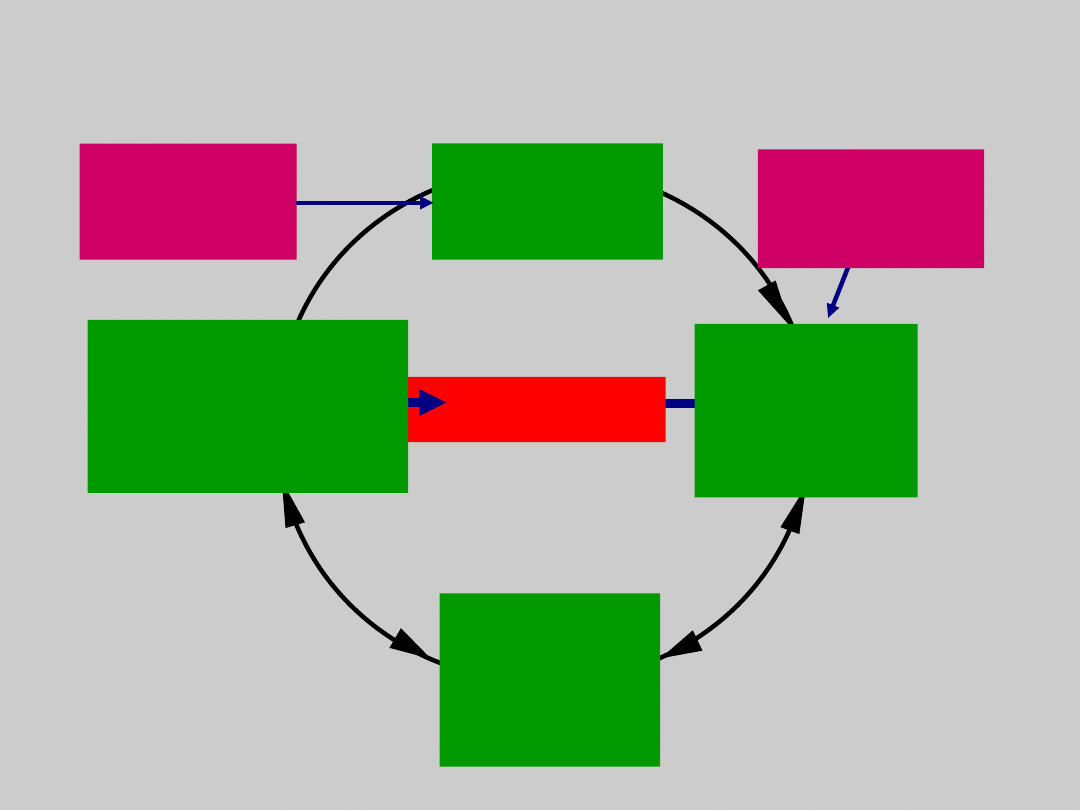
HBV Disease Progression
Chronic
Infection
Cirrhosis
Death
1. Torresi J, et al. Gastroenterology. 2000;118:S83. 2. Fattovich G, et al. Hepatology. 1995;21:77.
3. Perrillo RP, et al. Hepatology. 2001;33:424.
5%–10%
1
Liver
Failure
30%
1
23% in 5 yr
2
Liver
Cancer
(HCC)
Chronic HBV is the
6th leading
indication of liver
transplantation in
the US
3
~5%
Liver
Transplanta
tion
Acute Flare
6% in 5 yr
2

HBeAg Negative Patients
HBeAg positive patients may
develop antibodies (anti-HBe)
When HBeAg is lost, 2 possible
scenarios
–
Inactive carrier (normal ALT,
low or negative HBV DNA level)
–
Precore mutant chronic HBV
(moderate
to high HBV DNA, elevated ALT)

Inactive Carriers
HBeAg negative, normal ALT,
low/negative
HBV DNA
“Healthy carriers” = oxymoron
All patients who are HBsAg
positive need ongoing monitoring
as part of management
–
Monitor for increased ALT or HBV
DNA every 6 months
–
Monitor for HCC in at-risk
groups
Sherman M. Semin Liver Dis. 2005;25:143.

Precore Mutant (HBeAg
Negative)
Chronic HBV
HBeAg negative, elevated ALT, moderate to
high HBV DNA
Usually result of mutation in precore or
basal core promoter regions of HBV
Potentially more severe and progressive
chronic disease
Longer, more aggressive (suppression of
HBV DNA) treatment needed
29% risk of adefovir resistance at 5 years
of therapy
1
Seen predominantly in genotypes B,C, and D
1. Borroto-Esoda K, et al. J Hepatol. 2006;44(Suppl 2):179.

HBV DNA and Prognosis
High viral load predicts
Progression of liver
disease
1
Cirrhosis
1
Cirrhosis-related
complications
2
HCC
1,3
–
Independent of HBeAg,
ALT,
and cirrhosis
3
1. Chen G, et al. Abstract 996. Presented at: AASLD 2004. 2. Yuan JH,
et al. J Viral Hepat. 2005;12:373.
3. Chen C-J, et al. JAMA. 2006;295:65.
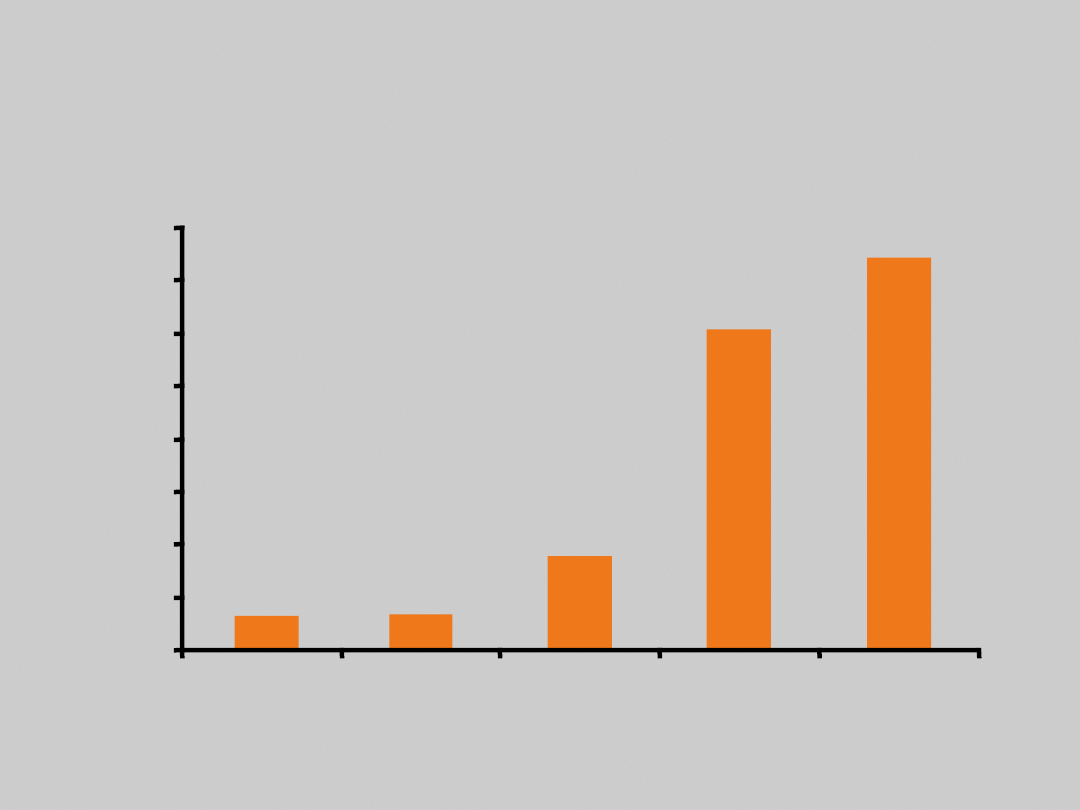
Baseline HBV DNA Level and
Cumulative Incidence of
HCC
Entire Cohort
0
2
4
6
8
10
12
14
16
<300
300–<10
4
10
4
–<10
5
10
5
–<10
6
≥10
6
N = 3653, 13-year follow-up
C
u
m
u
l
a
t
i
v
e
I
n
c
i
d
e
n
c
e
o
f
H
C
C
(
%
)
Chen C-J, et al. JAMA. 2006;295:65.
HBV DNA (Copies/mL)
1.
3
1.3
7
3.5
7
12.17
14.89
1IU = ~5 copies/mL
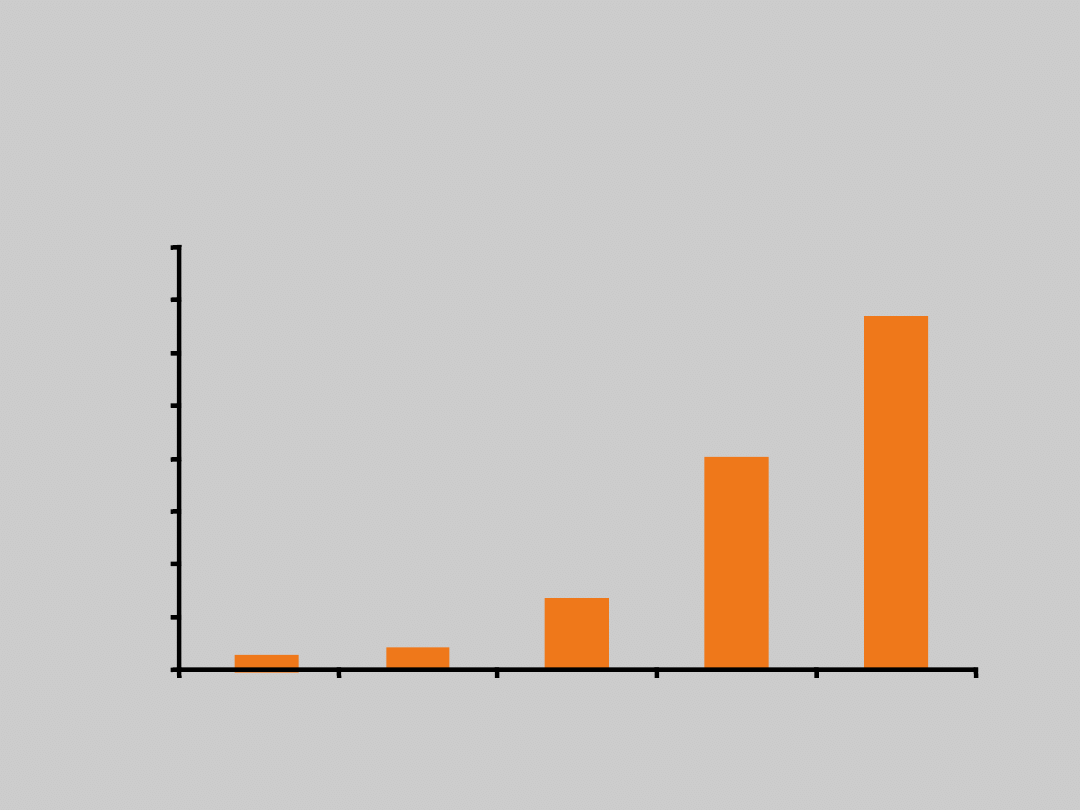
Baseline HBV DNA Level and
Cumulative Incidence of HCC
Subgroup of Noncirrhotic HBeAg– Patients
with Normal ALT
0
2
4
6
8
10
12
14
16
<300
300–<10
4
10
4
–<10
5
10
5
–<10
6
≥10
6
N = 2925
C
u
m
u
l
a
t
i
v
e
I
n
c
i
d
e
n
c
e
o
f
H
C
C
(
%
)
Chen C-J, et al. JAMA. 2006;295:65.
HBV DNA (Copies/mL)
0.7
4
0.8
9
3.1
5
7.96
13.50
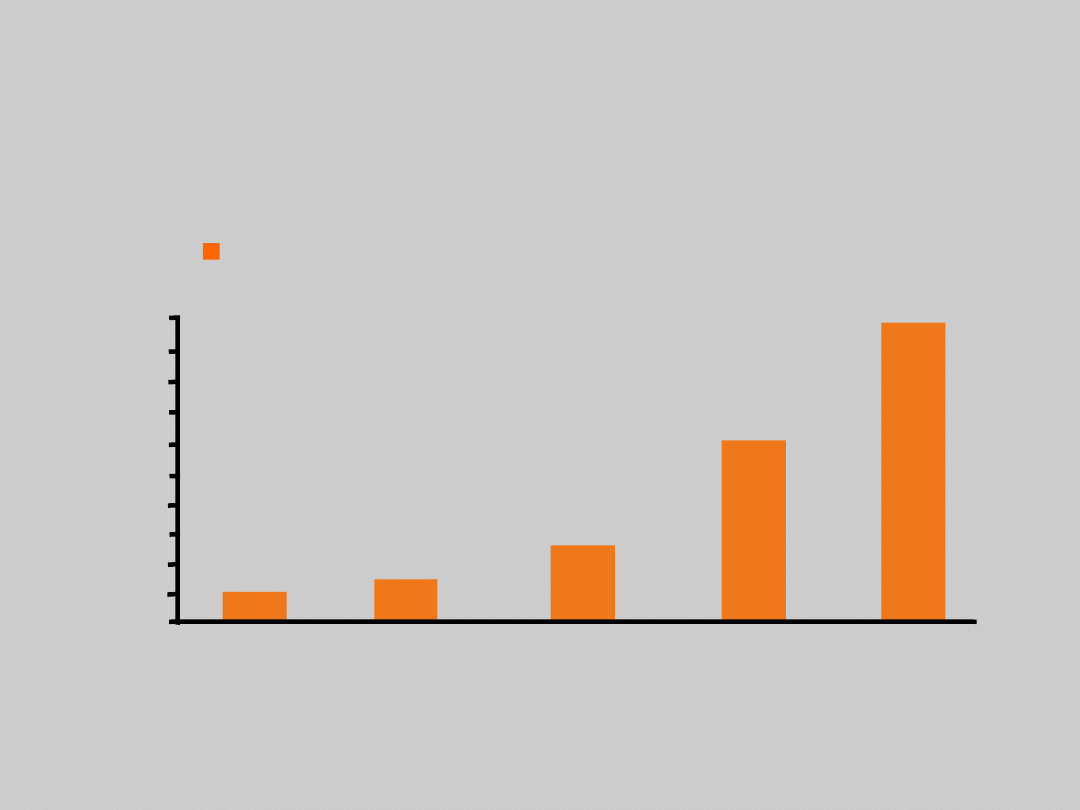
Baseline HBV DNA Level and
Relative Risk of Cirrhosis
Iloeje UH, et al, Gastroenterology. 2006;130:678.
0
10
<300
300–9.9x10
3
1.0–9.9x10
4
1.0–9.9x10
5
>10
5
N = 3582, 11-year follow-up
R
e
l
a
t
i
v
e
R
i
s
k
1
1.4
2.5
5.9
9.8
Adjusted for gender, age, smoking, alcohol consumption
HBV DNA (Copies/mL)
7
6
5
4
3
2
1
9
8

HBV DNA and Prognosis
Caveat
Low HBV DNA does not rule out risk
In HBeAg positive patients, HBV DNA
20,000 IU (<10
5
copies/mL) predicted better
histology
–
But 14.3% of patients with
HBV DNA 20,000 IU (<10
5
copies/mL) still
had fibrosis
1
In patients with cirrhosis, viral load was
the best predictor of complications
–
But even with HBV DNA 20,000 IU (<10
4
copies/mL) complications continued to
develop
2
1. Yuen MF, et al. Am J Gastroenterol. 2004;99:2031. 2. Yuan HJ, et al. J
Viral Hepat. 2005;12:373.

Candidacy for anti-HBV
Treatment
Principle
In general, a patient with chronic HBV is a
treatment candidate if there is evidence of
Liver disease (abnormal ALT) and
HBV replication (HBV DNA+)

When Do You Initiate
anti-HBV Therapy?
Parameters
–
HBV DNA levels >10
4-5
copies/mL
–
ALT levels >1–2 x ULN
Factors
–
HBeAg positive vs HBeAg
negative
–
Cirrhosis vs no cirrhosis
–
Compensated vs decompensated
disease

HBV DNA
HBeAg
(Copies/mL)
ALT
Management
+
>10
5
≤2 x ULN
Follow
+
>10
5
>2 x ULN
Treat
–
>10
5
>2 x ULN
Treat
–
–
≤2 x ULN
Follow
+/–
>10
5
Cirrhosis
If compensated, treat;
if decompensated*,
refer
for liver transplant
+/–
–
Cirrhosis
If compensated,
observe;
if decompensated*,
refer
for liver transplant
Which Patients Should Be
Treated?
AASLD Guidelines
1
1. Lok ASF, et al. Hepatology. 2004;39:857. Reprinted with permission of
Wiley-Liss, Inc, a subsidiary of John Wiley & Sons, Inc. 2. INTRON
®
A
(interferon alfa-2b) Product Information. Kenllworth, NJ: Schering
Corporation: 2004.
3. PEGASYS
®
(peginterferon alfa-2a) Product Information. Nutley, NJ: Hoffmann-
La Roche Inc.: 2004.
*Do not use interferon or peginterferon if the patient has
decompensated cirrhosis.
2,3
Specific treatment recommendations are
made elsewhere in this activity.
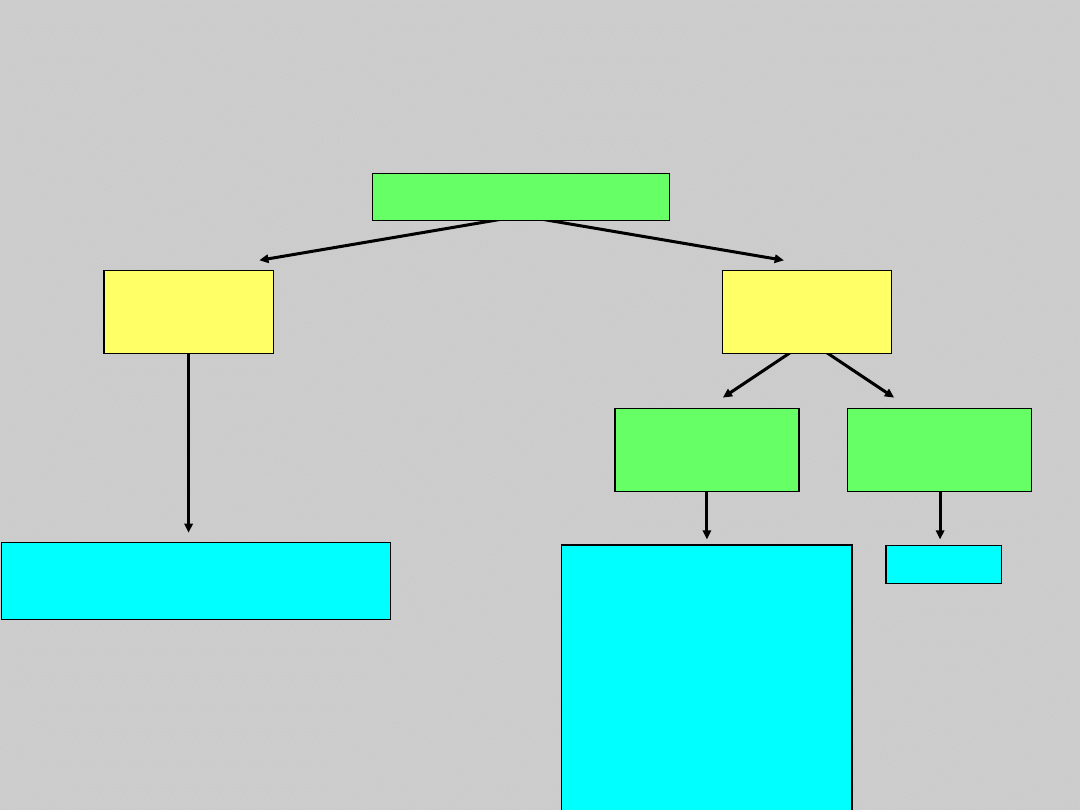
US Treatment Algorithm
Update
HBeAg Positive Compensated
Disease
No treatment
Monitor every 6–12 mo
Monitor every
3–12 mo
(immune
tolerant)
Consider
biopsy, if age
>35–40 y, and
treat if
significant
disease
Treat
HBeAg Positive
ALT
Elevated
ALT
Normal
HBV DNA
≥10
5
c/mL
HBV DNA
<10
5
c/mL
Courtesy of Emmet Keeffe, MD.
Keeffe EB, et al. Clin Gastroenterol Hepatol. 2006: In press.
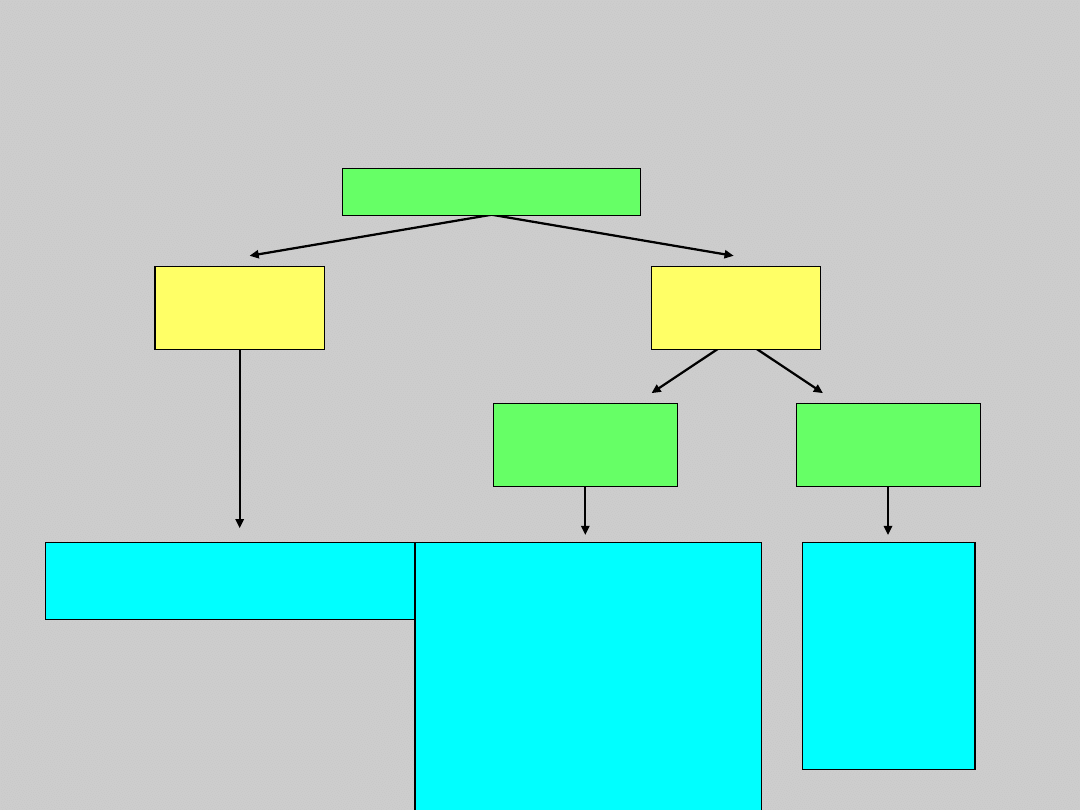
US Treatment Algorithm Update
HBeAg Negative Compensated
Disease
No treatment
Monitor every 6–12 mo
Monitor ALT, or
Consider biopsy,
since ALT often
fluctuates, and
treat if
significant
disease
Long-term
treatment required
Treat
Long-
term
treatme
nt
require
d
HBeAg Negative
ALT
Elevated
ALT
Normal
HBV DNA
≥10
4
c/mL
HBV DNA
<10
4
c/mL
Courtesy of Emmet Keeffe, MD.
Keeffe EB, et al. Clin Gastroenterol Hepatol. 2006: In press.
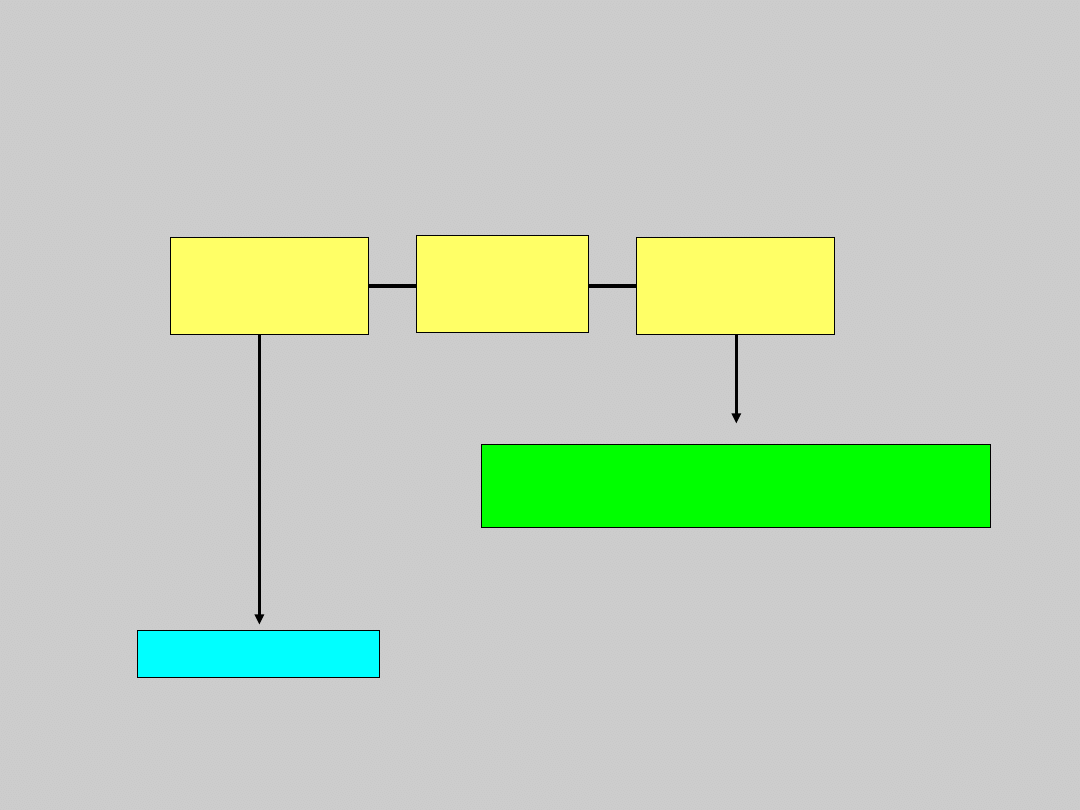
May Choose to Treat or
Observe
Treat
HBV DNA
(PCR)
HBV DNA
<10
4
c/mL
HBV DNA
≥10
4
c/mL
US Treatment Algorithm Update
Compensated Cirrhosis
Courtesy of Emmet Keeffe, MD.
Keeffe EB, et al. Clin Gastroenterol Hepatol. 2006: In press.
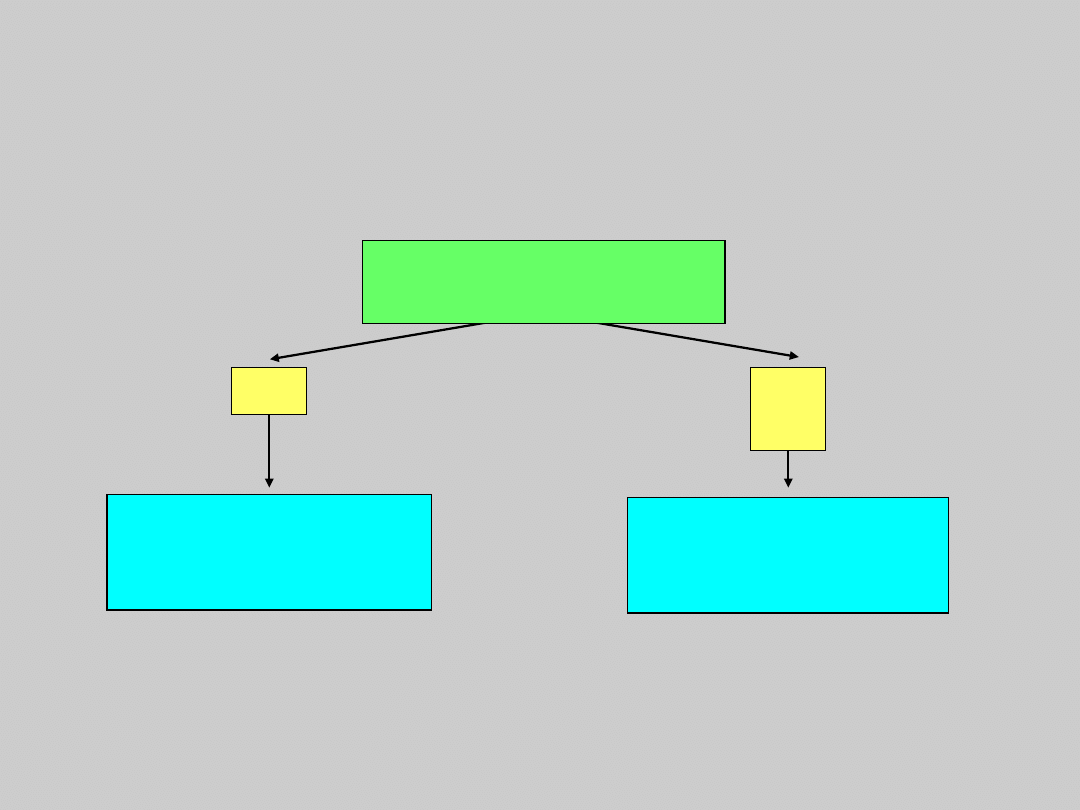
Observe
Wait list for
transplant
Treat
Wait list for
transplant
HBV DNA
Detectable by PCR?
No
Ye
s
US Treatment Algorithm Update
Decompensated Cirrhosis
Courtesy of Emmet Keeffe, MD.
Keeffe EB, et al. Clin Gastroenterol Hepatol. 2006: In press.

Who May Not Need Referral
to a Specialist?
Noncirrhotic patients with
persistently normal ALT
Inactive carrier
–
HBeAg negative
–
DNA <10
4
–
Persistently normal ALT
Immune tolerant
–
HBeAg positive
–
Persistently normal ALT

Who Is Likely to Benefit
from Referral to a
Specialist?
HBeAg positive chronic hepatitis B
–
Abnormal ALT
–
DNA >10
5
HBeAg negative chronic hepatitis B
–
Abnormal ALT
–
HBV DNA may be variable
Cirrhosis
–
Regardless of HBeAg, DNA or ALT
status

*Upper limits of normal for a person with normal BMI, 17 for women
and 25 for men;
†
Treat any patient with cirrhosis who is NAT positive, refer to
specialist;
‡
Rule out fatty liver and other causes of CLD;
§
Consider
3–5 years. NAT = nucleic acid testing, such as PCR, bDNA or TMA.
Reprinted from Gish R. Clin Liver Dis. 2005;9:541, with permission
from Elsevier.
Management of Chronic HBV
Infection
ALT <17–25* U/L
ALT >17–25* U/L
DNA
<2000 IU/mL
Observe
†
Biopsy
‡
<10,0000 c/mL
DNA
>2000 IU/mL
Biopsy
Treat:
>10,000 c/mL
and treat ifeAg (+): >6 mo post Ag
seroC
active HBV
eAg (–): prolonged Rx
(at least 24 mo) beyond
NAT negative
§
(Consider
biopsy or use
noninvasive
testing to stage
disease)

Monitoring for Patients Not
Considered for Treatment
Check ALT every 3–6 months
–
If ALT is persistently
elevated, reevaluate for
treatment
HCC surveillance in relevant
population
Lok ASF, et al. Hepatology. 2001;34:1225. Lok ASF, et al. Hepatology. 2004;39:857.

Bruix J, Sherman M. Hepatology. 2005;42:1208. Reprinted with permission of Wiley-Liss, Inc, a subsidiary of John Wiley & Sons, Inc.
HCC Surveillance Is Recommended for
the Following Groups of Patients (Level III)
Hepatitis B Carriers
Asian males ≥40 years
Asian females ≥50 years
All cirrhotic hepatitis B
carriers
Family history of HCC
Africans over age 20
For noncirrhotic hepatitis B
carriers not listed
above, the risk of HCC
varies depending on
the severity of the
underlying liver disease
and current and past
hepatic inflammatory
activity. Patients with
high HBV DNA
concentrations and those
with ongoing
hepatic inflammatory
activity remain at risk
for HCC.
Nonhepatitis B Cirrhosis
Hepatitis C
Alcoholic cirrhosis
Genetic hemochromatosis
Primary biliary cirrhosis
Although the following
groups have an
increased risk of HCC, no
recommendations
for or against surveillance
can be made
because a lack of data
precludes an
assessment of whether
surveillance would
be beneficial.
Alpha1-antitrypsin
deficiency
Nonalcoholic
steatohepatitis
Autoimmune hepatitis

1. Lok ASF, et al. Hepatology. 2001;34:1225. 2. Chevillotte G, et al. Gastroenterology. 1983;85:141.
3. Villa E, et al. Lancet. 1982;2:1243. 4. CDC. “The Pink Book.” 8th ed, 2005.
Counseling of
HBV-Infected Patients
Limit use of alcohol
1-3
Prevent transmission
1,4
Screen and vaccinate
sexual and
household contacts
1,4
Vaccinate against
hepatitis A
1

Treatment of
Hepatitis B
Primary Goal of anti-HBV Therapy
Preventing Cirrhosis, HCC, and Death
Durable Suppression
of HBV Replication

Treatment Goal
(Endpoints)
Remission of liver disease
Suppression of HBV
HBeAg positive HBV
–
Seroconversion
HBeAg negative HBV
–
Sustained suppression of
HBV DNA

Initial Therapy: What Are
the Therapeutic Options
and Considerations?
First-line therapy
–
Adefovir
–
Entecavir
–
Peginterferon alfa-2a
Lamivudine no longer considered
first-line therapy due to high
rate of resistance, except
in specific settings
Keeffe EB, et al. Clin Gastroenterol Hepatol. 2006: In press.

Peginterferon
Pegylated recombinant interferon alfa protein/immune
modulator
Subcutaneous injection, but less frequent than standard
interferon
Dual immunomodulatory and antiviral mode of action
Defined, finite treatment interval
High rate of HBeAg seroconversion in wild-type infection
High rate of HBV DNA suppression in precore mutant variant
High rate of HBsAg seroconversion
No reports of resistance mutations
Significant adverse events
–
Better than or similar to that when used to treat HCV
infection

Peginterferon
Considerations for Use
HBeAg positive
–
Response in genotype A better than B = C, better than D
1,2
–
ALT >80 IU/mL
2
–
HBV DNA <10
8
copies/mL
2
–
Compensated liver disease guidelines
3-6
–
Monoinfected
–
No psychiatric or medical contraindications
HBeAg negative
–
No specific genotype populations that benefit over others
7
–
Modest number of patients negative by PCR (20%) long term
7
Consider PEG IFN before nucleos(t)ide therapy due to defined
treatment intervals and
high HBeAg seroconversion rates
Should not be used in decompensated cirrhosis
8
1. Janssen HL, et al. Lancet. 2005;365:123. 2. Lau G, et al. N Engl J Med.
2005;352:2682. 3. Liaw YF, et al.
J Gastroenterol Hepatol. 2003;18:239. 4. De Franchis R, et al. J Hepatol.
2003;39:S3. 5. Lok AS, et al. Gastroenterology. 2001;120:1828. 6. Lok ASF, et
al. Hepatology. 2004;39:957. 7. Marcellin P, et al. Hepatology. 2005;42:580A.
8. PEGASYS® (peginterferon alfa-2a) Product Information. Nutley, NJ: Hoffmann-
La Roche Inc.: 2004.

Lamivudine
First oral nucleoside approved for
treatment of HBV
Cytidine nucleoside analog: inhibits
1st-strand DNA synthesis
Extensive database and publications
–
Half of patients who are HBeAg
positive undergo seroconversion by 5
years
1
Safety profile excellent except for
resistance
Risk of resistance is high (70%) at 4
years of therapy
2
–
Associated with flares and
decompensation
–
No longer a first-choice therapy due
to high rate of resistance
3
1. Guan R, et al. J Gastroenterol Hepatol. 2001;16(suppl):A60. 2. Lai CL, et
al. Clin Infect Dis. 2003;36:687. 3. Keeffe EB, et al. Clin Gatroenterol
Hepatol. 2006: In press.
Lai C-L, et al. Clin Infect Dis. 2003;36:687.
Lamivudine in CHB
Incidence of YMDD Mutants Over Time—
Integrated Phase 3 Data
0
20
40
60
80
100
Y
M
D
D
(
%
)
Year
1
24%
n =
426
Year
2
42%
n =
74
Year 3
53%
n =
58
Year 4
70%
n =
58

Adefovir
Adenosine nucleotide analog
First oral medication and first
nucleotide approved by regulatory
authorities for the treatment of HBV
Inhibits HBV DNA polymerase
Oral bioavailability not affected by food
No significant drug-drug interactions
Rare events of nephrotoxicity (overall
risk – low)
Effective against lamivudine-resistant
mutants
Long/indefinite duration
HEPSERA
®
Product Insert.

Safety of Adefovir
Dipivoxil
Over 4–5 Years
Resistance is 29% at 5 years of use in
HBeAg negative patients
1
Resistance is more likely if patients are
lamivudine resistant and switched to
adefovir
2
Renal safety
–
Infrequent (3%) increases in creatinine
≥0.5 mg/dL
Maximum value 1.5 mg/dL
Maximum increase 0.8 mg/dL
1. Borroto-Esoda K, et al. J Hepatol. 2006;44(Suppl 2):179.
2. Lee YS, et al. Hepatology. 2006;43:1385.
3. Hadziyannis S, et al. J Hepatol. 2006;44(Suppl 2):283.

1. Chang TT, et al. Hepatology. 2004;40:193A. 2. Sherman M, et al.
Hepatology. 2004;40:664A. 3. Shouval D, et al. Hepatology. 2004;40:728A. 4.
Colonno RJ, et al. 56th AASLD. November 11–15, 2005. Poster 962.
Entecavir
Oral deoxyguanine nucleoside analog
Inhibits priming of HBV DNA polymerase, reverse
transcription of negative DNA strand from pregenomic RNA,
and synthesis of positive DNA strand
Superior efficacy to lamivudine
1-3
Comparable safety profiles except fewer ALT flares
compared with lamivudine
1-3
ETV-associated resistance
mutations were not observed in
nucleoside-naive, HBeAg positive, or negative studies
4
ETV-associated resistance mutations with rebound were
seen in 9%
of patients with previous lamivudine resistance during
second year
of therapy
4
ETV = entecavir.
Entecavir Phase 3 Studies
Histologic Endpoints–Primary
Efficacy Endpoint
Study 022*
HBeAg+
Study 027*
HBeAg–
Study 026
†
LVD-Refractory
ETV
0.5 mg
(n =
314)
LVD
100 mg
(n =
314)
ETV
0.5 mg
(n =
296)
LVD
100 mg
(n =
287)
ETV
1 mg
(n =
124)
LVD
100 mg
(n =
116)
Overall
histologic
improvement
72%
62%
70%
61%
55%
28%
Fibrosis no
worse
89%
82%
84%
79%
87%
70%
Necroinflammato
ry
>2-point
decrease
74%
64%
73%
64%
55%
32%
Ishak fibrosis
score
improvement
39%
35%
36%
38%
34%
16%
Primary analysis: only patients with evaluable baseline biopsy
included; missing/inadequate week-48 biopsy counted as failures.
*Treatment for 52 weeks up to 96 weeks for partial responders.
†
Treatment for 48 weeks for up to 96 weeks for partial
responders. ETV = entecavir; LVD = lamivudine.
FDA. Entecavir briefing document. February 10, 2005.
Entecavir Phase 3 Studies
Selected Secondary Efficacy
Endpoints
FDA. Entecavir briefing document. February 10, 2005.
*Treatment for 52 weeks up to 96 weeks for partial responders.
†
Treatment for 48 weeks for up to 96 weeks for partial
responders. ETV = entecavir; LVD = lamivudine.
Study 022*
HBeAg+
Study 027*
HBeAg–
Study 026
†
LVD-Refractory
ETV
0.5 mg
LVD
100 mg
ETV
0.5 mg
LVD
100 mg
ETV
1 mg
LVD
100 mg
HBV DNA PCR
<400 copies/mL
72%
42%
95%
77%
22%
1%
Log HBV DNA by
PCR (mean
change from
baseline)
-7.0
-5.5
-5.2
-4.7
-5.1
-0.5
HBeAg
seroconversion
21%
18%
NA
NA
8%
3%
ALT normalization
(<1 x ULN)
69%
61%
78%
71%
65%
17%

Current FDA-Approved
Nucleoside and Nucleotide
Treatment Options
(Not Head-to-Head Studies)
Lamivudine Adefovir
Entecavir
Resistance
~20%
~3%
~0%
Naive;
9% Lam-
R
E antigen seroconversion
~17%
~12%
~22%
Flares
Moderate
Low
Low
Viral suppression
4.5–5.5
3.5–5.5
5.2–7
(log reduction)
DNA negativity
32%–78%
21%–51%
69%–91%
Availability of long-term data 5 year
5 year
2 year
AEs
Rare
Rare
Rare
Lam-R = lamivudine resistance
Slide Courtesy of Robert G. Gish, MD.
Results Following
Suppression
of Viral Replication
Preventio
n of
Death,
Cirrhosis
, and HCC
Histologic
Improvement
Virologic
Response
Reduction
in:
HBV DNA
cccDNA
Serologic
Response
HBeAg loss
HBeAg
seroconversion
HBsAg loss and
seroconversion
Biochemical
and
Liver
Synthetic
Test
Improvement
ALT
Bilirubin,
INR,
Albumin

Summary
HBV infection is a worldwide epidemiologic
and clinical challenge
Screening at-risk individuals identifies
those with HBV infection
Pretreatment evaluation includes history,
physical exam,
and additional diagnostic testing
Candidates for anti-HBV treatment include
patients with active liver disease and high
levels of HBV replication
Refer HBsAg positive patients for treatment
with entecavir, adefovir, or peginterferon
Document Outline
- Optimal Management of HBV: A Partnership Between Primary Care and Specialty Physicians
- PowerPoint Presentation
- Hepatitis B: An Important Disease for Primary Care and Specialty Physicians
- Why Is HBV Relevant in Primary Care?
- Global Impact of HBV A Significant Cause of Worldwide Morbidity and Mortality
- HBV A Global Health Problem
- HBV Disease Progression
- Slide 8
- Poor Survival in HBV-Related Cirrhosis
- HBV Is Preventable
- Slide 11
- HBV Vaccine Indications
- US Incidence of Viral Hepatitis Infection Over Time
- HBV Vaccination Effect on HCC Incidence and Mortality*
- Screening for HBV and Evaluating Infected Patients
- Who Should Be Screened?
- Slide 17
- Screening Tests
- History and Physical Evaluation
- Assessment of Liver Disease Severity
- 2nd-Phase Testing in HBsAg+ Patients
- Further Testing for HBsAg+ Patients
- Hepatitis Delta Virus (HDV)
- Who Are Treatment Candidates?
- Natural History of HBV Infection
- Phases of Chronic HBV Infection
- Slide 27
- HBeAg Negative Patients
- Inactive Carriers
- Precore Mutant (HBeAg Negative) Chronic HBV
- HBV DNA and Prognosis
- Baseline HBV DNA Level and Cumulative Incidence of HCC Entire Cohort
- Baseline HBV DNA Level and Cumulative Incidence of HCC Subgroup of Noncirrhotic HBeAg– Patients with Normal ALT
- Baseline HBV DNA Level and Relative Risk of Cirrhosis
- HBV DNA and Prognosis Caveat
- Candidacy for anti-HBV Treatment Principle
- When Do You Initiate anti-HBV Therapy?
- Which Patients Should Be Treated? AASLD Guidelines1
- US Treatment Algorithm Update HBeAg Positive Compensated Disease
- US Treatment Algorithm Update HBeAg Negative Compensated Disease
- US Treatment Algorithm Update Compensated Cirrhosis
- US Treatment Algorithm Update Decompensated Cirrhosis
- Who May Not Need Referral to a Specialist?
- Who Is Likely to Benefit from Referral to a Specialist?
- Slide 45
- Monitoring for Patients Not Considered for Treatment
- Slide 47
- Counseling of HBV-Infected Patients
- Treatment of Hepatitis B
- Slide 50
- Treatment Goal (Endpoints)
- Initial Therapy: What Are the Therapeutic Options and Considerations?
- Peginterferon
- Peginterferon Considerations for Use
- Lamivudine
- Slide 56
- Adefovir
- Safety of Adefovir Dipivoxil Over 4–5 Years
- Entecavir
- Entecavir Phase 3 Studies Histologic Endpoints–Primary Efficacy Endpoint
- Entecavir Phase 3 Studies Selected Secondary Efficacy Endpoints
- Current FDA-Approved Nucleoside and Nucleotide Treatment Options (Not Head-to-Head Studies)
- Results Following Suppression of Viral Replication
- Summary
Wyszukiwarka
Podobne podstrony:
Management of Hepatocellular Carcinoma
Diagnosis and Management of Hemochromatosis
Long term Management of chronic hepatitis B
Management of severe psoriasis
Guidelines for the Management of Aneurysmal Subarachnoid Hemorrhage
Management of Adult Patients With Ascites Due to ascites
Introduction Blocking stock in warehouse management and the management of ATP
Diagnosis and Management of hepatitis
Majewski, Marek; Bors, Dorota On the existence of an optimal solution of the Mayer problem governed
Practical Evaluation and Management of Atrophic Acne Scars
Management of gastro oesophageal reflux disease in general practice
Betcke T OPTIMAL SCALING OF GENERALIZED AND POLYNOMIAL EIGENVALUE PROBLEMS
Professional Management of Housekeeping Operations
Skill 21[1] Management of Gastrointestinal Suction
Management of infectionous diarrhoea
Optimal Control of Three Phase PWM Inverter for UPS Systems
Combination Therapy in the Management of Atrophic Acne Scars
więcej podobnych podstron