
*
Corresponding author.
AMW: FRCS; MKDB: FRCS.
Current Orthopaedics (2001) 15, 127d134
^
2001 Harcourt Publishers Ltd
doi:10.1054/cuor.2001.0176, available online at http://www.idealibrary.com on
CHILDREN
LeggdCalveHdPerthes’ disease
A. M. Wainwright and M. K. D. Benson*
Nuffield Orthopaedic Centre, Windmill Road, Headington, Oxford OX3 7LD, UK
INTRODUCTION
This intriguing condition is defined as idiopathic avascular
necrosis of the femoral capital epiphysis during
childhood. In the mild form, the disorder can be limited
to a period of hip pain and limping which resolves
without any long-term problem. If segmental collapse and
subsequent deformity complicate femoral head necrosis,
it may result in premature hip osteoarthritis. Much is
written about the disease but the aetiology remains
unknown, the course of the disease may be unpre-
dictable, and the best method of treatment has not been
determined.
HISTORICAL FACTS
The condition has been described as coxa plana,
osteochondritis deformans juvenilis, Perthes’ disease,
Legg}Perthes’ disease,
Legg}CalveH}Perthes’
disease
(LCPD), and occasionally WaldenstroKm’s name is added.
Henning WaldenstroKm,
Professor
of
Orthopaedic
Surgery in Stockholm, is generally credited with the first
description of the condition in 1909. He felt it was
a benign form of tuberculosis. However, Alben KoKhler
had previously published a radiograph in 1905, and the
first pathological description was by Fragenheim in 1909.
The next year, 1910, it was described independently by
Arthur T Legg (a Harvard surgeon), Jacques CalveH, (at
BerckdPlage tuberculosis hospital near Paris), and Georg
Clemens Perthes (a German surgeon). These three
clinicians recognized that it is a problem distinct from
tuberculosis of the hip which was prevalent at the time.
Epidemiology
The annual incidence rate is of the order of five to 10
newly diagnosed cases per 100 000 children per year.
There are several epidemiological facts of interest.
E
The condition affects boys much more often than girls,
with a 4 : 1 ratio.
E
The condition is bilateral in 10}20% of patients, in
whom the disease may run a more severe course.
E
There
is
variation
between
population
groups
and between regions within countries. For example,
within the United Kingdom the annual incidence is
5.5/100 000 in Wessex and 11.1 in Liverpool.
This is
more likely to be associated with differences in social
deprivation than because one area is rural and the
other urban.
E
There is a marked difference in the annual incidence
between white children compared to black children
(10.8 compared to 0.45 per 100 000, respectively, in
South Africa).
E
Unlike many childhood conditions there is no clear
evidence of a strong genetic component. This is based
on studies of first-degree relatives, twin studies and
the difference in the incidence between the sexes
which exclude the skeletal dysplasias.
E
The clinical onset is within a narrow age range,
predominantly affecting children aged between 4 and
7 years and rarely outside the range of 3}13 years.
E
There is a higher risk of LCPD in children who are
passive smokers.
E
Affected children are of short stature, and have
delayed bone age.
E
There is no evidence that the majority of children with
LCPD have had a preceding irritable hip. Of the few
children with recurrent hip irritability, only those with
over 2 year’s delay in bone growth were found later to
have LCPD.
Aetiology
The cause of the condition is unknown although there
are several theories. It is generally accepted that ‘in
a susceptible child the changes are the consequence of
ischaemia of variable duration, after which a process
of repair produces a growth disturbance, which if
uncontrolled leads to femoral head deformity with
subsequent arthritis’.

Table 1
I.
The evolutionary period divided into 2 stages
a.
The initial stage*the epiphysis is dense, with ‘de-
calcinated’ spots and flattened, uneven margins.
b.
The fragmentation stage*the epiphysis is flattened
and divided, granular and ‘atrophied’
II.
The healing period*the epiphysis becomes homo-
geneous and there is evidence of recalcification.
III.
The growing period*normal growth and ossification
resume within the deformed femoral head.
VI.
The definite period*permanent, residual features are
evident
It has been proposed that there is an underlying
coagulation defect which leads to vascular thrombosis
(the thrombophilia theory). Reports have shown
associations of LCPD with hypercoagulable conditions
such as protein C and protein S deficiency and resistance
to activated protein C.
Subsequent studies have failed
to confirm these findings, but have found a prolonged
activated partial thromboplastin time.
This theory
may explain the mechanism that relates it to passive
smoking.
Experimental work in animals
shows that repeated
episodes of occlusion of the arteries of the femoral head
can cause the pathological changes seen in LCPD
(vascular theory). Superselective angiography of the
lateral
epiphyseal
arteries
shows
that
68%
are
interrupted at their origin in LCPD with signs of
revascularization at later stages.
LCPD is associated
with a bone age 2}3 years behind the chronological age.
Further evidence for an hormonal theory is the
observation that there are abnormal levels of some
growth factors including insulin-like growth factor-
binding protein 3.
PATHOGENESIS
WaldenstroKm described segmental stages
for the
disease (Table 1).
These stages are now recognized as:
1. Initial stage.
2. Fragmentation stage.
3. Healing stage.
4. Remodelling stage.
Histologically, several changes follow sequential infarc-
tion of the femoral head.
E
Synovial tissue becomes inflamed and causes an
effusion.
E
Articular cartilage is nourished mainly from synovial
fluid, and continues to grow; cartilage becomes thicker
over the medial femoral head and acetabular floor; at
the deep surface it may transform to fibrocartilage.
E
Growth plate cartilage columns become distorted and
do not undergo normal ossification.
E
Epiphysis is affected variably as the trabeculae and
the subchondral bone plate become necrotic and
fragment; in adjacent unaffected areas of the epiphysis
the appearances are normal, with some remodelling;
later, in the reparative phase, there is new bone
deposition on the necrotic trabeculae and a callus-
like cartilaginous tissue adjacent to the necrotic
part.
E
Metaphysis contains adipose tissue and sclerotic-
rimmed osteolytic lesions of fibrocartilage; ossification
is disorganized and the growth plate ruptures into the
adjacent bone.
CLINICAL FEATURES
Symptoms
A typical patient would be a white boy between 4 and
8 years old who presents with pain in the hip, or more
commonly, the knee.
Relatives or teachers may report
that they have noticed a limp. The child feels well.
Signs
The first sign of LCPD may be a limp. WaldenstroKm
reported that at first the limping may be so slight that it
may not be noticeable, but heard when the child walks
across the floor with shoes on.
Initially the gait is
antalgic. Later this may become a Trendelenburg gait
because of trochanteric overgrowth and femoral head
flattening. Lying on the couch, there is limited internal
rotation in extension. The hip flexes into abduction and
external rotation. An abduction contracture is a sign of
severe disease with lateral impingement of the epiphysis
against the acetabulum.
DIFFERENTIAL DIAGNOSIS
Other conditions may simulate LCPD, affecting just one
hip, or both.
E
Infection}tuberculosis of the hip is the classic
differential diagnosis and worldwide this condition
remains prevalent. Subacute septic arthritis or osteo-
myelitis of the femoral neck can cause similar changes.
E
Gaucher’s disease differs in that there is a failure to
remodel with associated anaemia, thrombocytopenia
and hepatosplenomegaly.
E
Eosinophilic granuloma often presents with a high
erythrocyte sedimentation rate (ESR), and other
lesions may be apparent on a skeletal survey.
128
CURRENT ORTHOPAEDICS
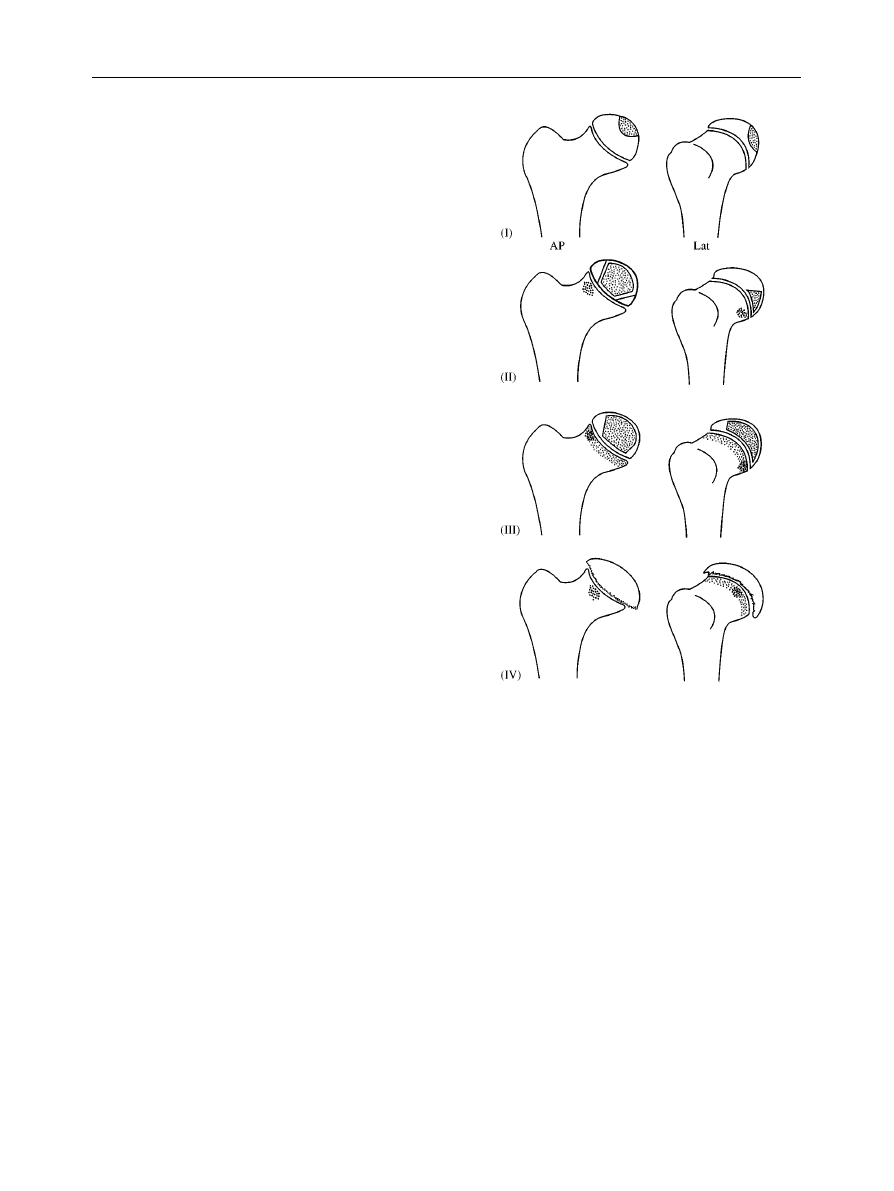
Figure 1
The Catterall classification of Legg}CalveH}Perthes’
disease.
E
Lymphoma deposits in the femoral neck cause
complete infarction and the change is progressive.
E
Sickle cell disease, haemophilia, steroid treatment,
leukaemia and immunosuppression can be associated
with femoral head osteonecrosis.
There are several other conditions which may mimic
bilateral LCPD. These conditions can be differentiated
from LCPD; the development of the femoral capital
epiphysis is usually delayed and changes are synchronous
and symmetrical.
E
Hypothyroidism produces a widened metaphysis and
classic clinical signs.
E
Spondylo-epiphyseal dysplasias produce an uninvolved
but cup-shaped metaphysis. Clinical examination
shows a short trunk and spinal radiographs confirm
platyspondyly. Other epiphyses may be affected.
E
Multiple epiphyseal dysplasia produces similar changes
to spondylo-epiphyseal dysplasia at the hip but a
normal sitting-height. A skeletal survey confirms the
diagnosis.
RADIOGRAPHIC FEATURES
The radiographic features are best seen on an antero-
posterior view
of
the
pelvis and
a
frog-lateral
(Lauenstein) view of the hip in flexion, abduction and
external rotation. The changes depend upon the
duration, stage and severity of the disease, which is the
basis for classification as outlined below. One of the
earliest signs is medial joint space widening, which is
thought to reflect thickening of the acetabular floor
cartilage and may be present in the opposite hip.
OTHER INVESTIGATIONS AND
THEIR VALUE
Ultrasound scans offer an investigation of hip pain, which
is safe, inexpensive and reproducible. It can be useful in
combination with aspiration to exclude the important
differential diagnosis of septic arthritis. Capsular dis-
tension that persists for more than 6 weeks is an early
manifestation of LCPD.
Other diagnostic markers
which may be useful in making the diagnosis are cartilage
thickening and quadriceps atrophy.
Radioisotope bone scans have a higher sensitivity and
specificity than radiographs and identify the onset of
revascularization earlier.
Arthrography is as good or better than MRI in
determining the shape of the articular surface and lateral
subluxation. MRI scans can give earlier information on
the extent and location of involvement and healing, but
there is little evidence that MRI produces information
that would alter management.
CLASSIFICATION OF SEVERITY
There are three classification systems commonly used to
classify the severity of LCPD.
1. Catterall
described four groups based upon antero-
posterior (AP) and lateral radiographs (Fig. 1).
E
Group 1eonly the anterior part of the epiphysis is
involved on the lateral view. No collapse of the
femoral head is seen and complete absorption of the
involved
segment
occurs
without
sequestrum
formation, followed by regeneration. The AP may
show a cystic epiphysis, but there is no loss of height
and metaphyseal changes are unusual.
E
Group 2emore of the anterior epiphysis is involved
and this may collapse, leaving a dense sequestrum. On
the AP a dense oval mass may be visible with viable
fragments medially and laterally which maintain height.
On the lateral view, a ‘V’, characteristic of this group,
LEGG}CALVED}PERTHES’ DISEASE
129
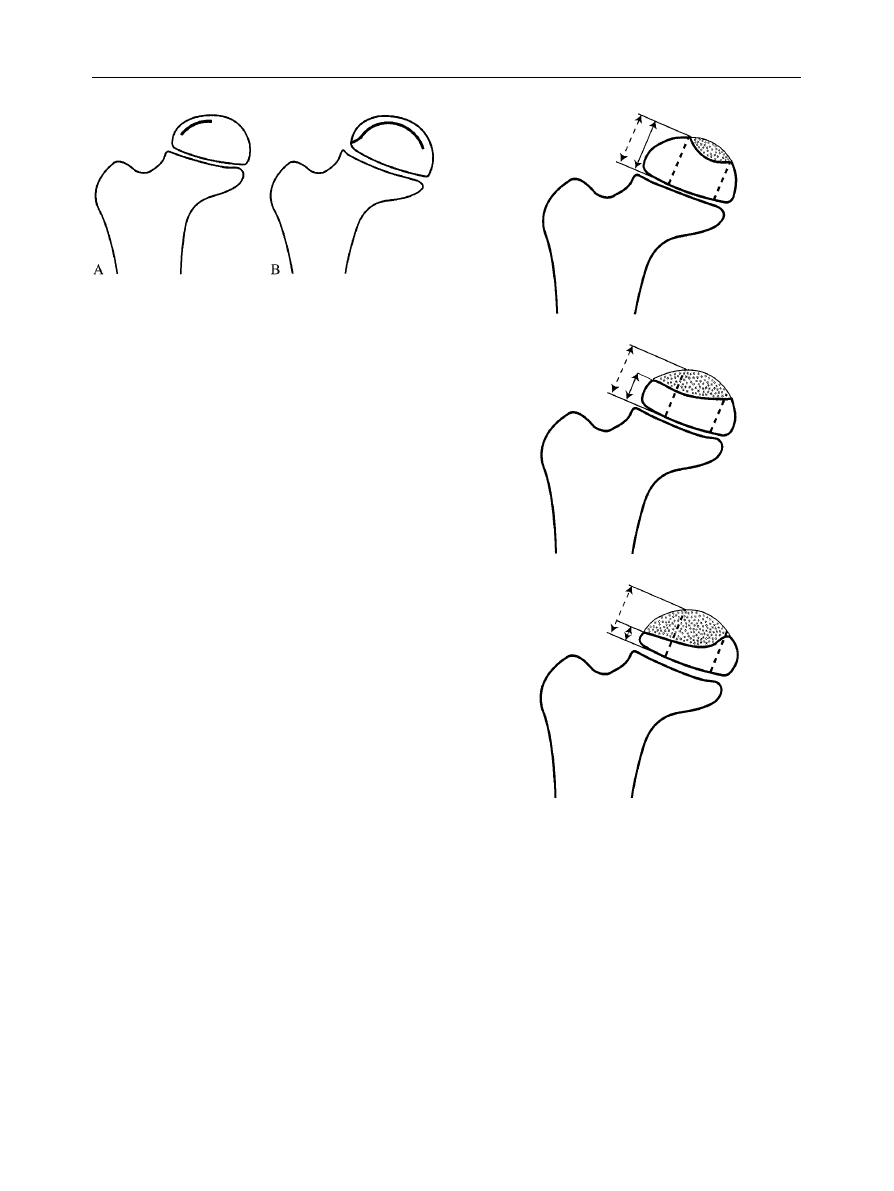
Figure 2
The Salter}Thompson classification of Legg}
CalveH}Perthes’ disease.
Figure 3
The Herring lateral pillar classification of Legg}
CalveH}Perthes’ disease.
may separate the sequestrum posteriorly from the
viable fragments.
E
Group 3eonly a small part of the epiphysis is not
sequestrated. The AP shows the appearances of
a ‘head within a head’, with later collapse of the central
sequestrum and very small normal segments medially
and laterally. The lateral segment is often compressed
and osteoporotic, displacing laterally to produce
a broadened neck. Metaphyseal changes are more
generalized and associated with a broad neck.
E
Group 4ethe whole epiphysis is sequestrated and
collapses to produce a dense line on the AP view. Early
loss of height between the physis and the acetabular
roof indicates flattening of the head. The epiphysis can
‘mushroom’ anteriorly and posteriorly. Metaphyseal
changes are extensive.
2. Salter and Thompson described a simple two-group
classification
based upon the extent of the involvement
of the femoral head (Fig. 2).
E
Group A (less than half of the head),
E
Group B (more than half of the head)
The authors state that it can be applied early in the stage
of the disease when the subchondral fracture is
detectable. This fracture may not be visible in two thirds
of cases, however.
3. Herring et al. described a classification system
based
upon the extent of involvement of the ‘lateral pillar’ of
the femoral head (Fig. 3). The lateral pillar is the area in
the lateral 15}30% of the femoral head on a true antero-
posterior film. An assessment of the height of this pillar is
compared to the unaffected hip, ignoring the amount of
collapse of the central and medial pillars.
E
Group A (no involvement of the lateral pillar).
E
Group B ('50%
of the lateral pillar
height
maintained).
E
Group C ((50% of the lateral pillar height
maintained).
All three of these classification systems have been
assessed to find which system is most repeatable when
different individuals try to classify the severity of disease
based on X-ray appearances. In these studies, there were
Kappa values (a measure of agreement, with a maximum
of 1.0) of more than 0.5 for all three systems.
The
best value was obtained when consultant staff from the
originating unit assessed the Salter and Thompson
classification, with Kappa scores of 0.99.
PROGNOSIS AND THE
‘HEAD AT RISK’
At least 50% of involved hips do well with no
intervention. There is a group of children with LCPD
130
CURRENT ORTHOPAEDICS

Table 2
Clinical signs
1 The obese child
2 A decreasing range of movement
3 Adduction contracture
Radiological signs
1 Lytic area in the lateral epiphysis and metaphysis (‘Gage’s
sign’)
2 Calcification lateral to the epiphysis
3 Diffuse metaphyseal reaction
4 Femoral head lateral subluxation
5 A horizontal growth plate
Figure 4
The Stulberg classification of the radiological appear-
ance of the hip at maturity following Legg}CalveH}Perthes’ disease.
who tend to have poor results associated with
deterioration in femoral head shape.
The most widely accepted prognostic factor is the
patient’s age at onset. There is a better chance of
recovery when the onset is before the age of 6 years and
a worse chance after the age of 8. This may reflect the
time available for remodelling.
Catterall reported that patients who have a femoral
‘head at risk’, have several clinical and radiological signs
as outlined in Table 2. Three clinical signs of a ‘head at
risk’ were described.
The obese child is at greater risk
(although the criteria for obesity were not specified).
A decreasing range of movement may be the earliest sign
of subluxation of the femoral head, in contrast to an
increasing range of movement that may be the earliest
sign of healing.
An adduction contracture may also be
a sign of lateral subluxation.
Five radiological signs are associated with a poor
prognosis.
A lytic area in the lateral epiphysis and
adjacent metaphysis was called Gage’s sign by Catterall
and has been universally known by this eponym
subsequently. It has been suggested that the lytic
appearance should be called Catterall’s sign
as Gage’s
original description of an early radiological sign in
Perthes’ disease was of the lateral metaphysis being
curved rather than straight.
Calcification lateral to
the epiphysis is caused by ossification of the extruded
femoral
head
(as
the
sphere
is
squashed
and
‘mushrooms’ out). The
diffuse metaphyseal reaction is
caused by non-ossified nests of cartilage cells, giving
a cystic radiological appearance.
Femoral head lateral
subluxation appears to be present due to the thickened
medial femoral head and acetabular floor cartilage. This
causes loss of containment and alters the shape of the
acetabulum, causing high pressures on the softened
femoral head. A
horizontal growth plate reflects the fact
that hip is lying in external rotation.
According to Stulberg and Salter,
the four major
factors that determine the prognosis are
E
Age of the patient at onset.
E
Extent of involvement of the femoral head.
E
Loss of containment of the femoral head when the
acetabulum in the weight-bearing position.
E
Loss of motion of the hip.
OUTCOMES
Stulberg suggests that it is
possible to
predict
progression to osteoarthritis based upon the radio-
graphic features at maturity. From a longitudinal study of
three to four decades, prognosis was shown to be
related to the congruency between the femoral head and
acetabulum.
Five classes of deformity are described
E
Class I, Completely normal hip joint
E
Class II, Spherical femoral head but with one or more
of the following characteristics: larger than normal
although spherical femoral head (coxa magna). shorter
than normal femoral neck (coxa brevis). abnormally
steep acetabulum.
E
Class III. Non-spherical (ovoid, mushroom-shaped or
umbrella-shaped), but not flat, femoral head. Class II
characteristics are present.
LEGG}CALVED}PERTHES’ DISEASE
131

E
Class IV. Flat femoral head and abnormalities of the
femoral head, neck or acetabulum
E
Class V. Flat femoral head but a normal femoral neck
and acetabulum, so that congruency is lost.
From this classification, three types of congruency are
recognized
1. Spherical congruency (class I and II hips)ein hips in
this category early arthritis does not develop.
2. Aspherical congruency (class III and IV hips)emild
to moderate arthritis develops in late adulthood.
3. Aspherical incongruency (class V hips)esevere
arthritis develops before early or middle age in
these hips.
There is considerable interobserver variability using this
system. This is because some of the criteria are not
clearly defined (for example, a flat head, and a steep
acetabulum).
MANAGEMENT
There is still doubt as to which is the best method of
treatment, partly owing to the fact that 50% of patients
will have a good result with no treatment. There are
enthusiasts for the different methods of treatment and it
appears that several treatment options are available
which may provide very similar results.
There are no studies available that compare different
forms of treatment with a long enough follow-up to
determine which is best for preventing early osteo-
arthritis. Measures of joint sphericity and containment
and often used to assess the outcome of treatment.
There have been difficulties in assessing comparable
series of children,
as several factors influence the
outcome including the age at onset, stage at diagnosis,
stage at treatment, whether both hips are involved, and
the severity of disease. There is notable interobserver
variability in the systems used for classifying the severity
of disease and outcome. Finally there is a long lag period
of three or four decades between the onset of the
disease and the main effects upon the hip. There is a
prospective, multicentre study in progress which may help
to clarify the situation ( J. A. Herring, pers. comm. 2001).
Pragmatically, it is important to determine what the
prognosis is likely to be, as this will influence the choice
of intervention. Other important considerations are the
range of movement of the hip joint and the impact of
treatment on the child physically, socially and at school.
Good prognosis
If the prognosis is likely to be good without treatment
then the aim of any intervention will be for symptomatic
control. The symptoms may last several months and can
fluctuate from day to day. The majority of these children
can rest at home and avoid weight bearing, using
crutches if old enough to comply. During this period the
child should not participate in competitive sports and
should be in a wheelchair for long walks. Activities such
as swimming and cycling may be helpful. Physiotherapy is
used to treat muscle spasm and regain abduction.
Poor prognosis
For patients who have features associated with a poor
prognosis, the aim of any intervention is to improve the
range of movement and decrease the likelihood of
premature osteoarthritis. Most methods of treatment
are based upon the concept of ‘containment’ which
assumes that the acetabulum will contain and mould the
softened femoral head. The problem with this concept is
that the acetabulum is 1/3 of a sphere and the femoral
head, 2/3 of a sphere.
Although containment is not
clearly defined, over 80% head coverage may be required
to prevent extrusion of the femoral head and lateral
compression.
Historically, treatment was aimed at preventing weight
bearing until the femoral head had reossified. This
included prolonged bed-rest, often in hospital, using
traction, a frame or a spica cast. Complications included
muscle atrophy, osteoporosis, leg shortening, loss of
thoracic kyphosis, urinary calculi, and social, academic
and emotional problems.
Currently, the three widely used methods of contain-
ment are weight-bearing orthosis, femoral osteotomy
and innominate osteotomy.
1. Weight-bearing abduction brace
Devices such as the Atlanta Scottish Rite brace keep
the hips abducted, but permit mobility at the hip.
Arthrography is often used prior to bracing in order to
assess congruency in different positions, avoiding hinge
abduction. Serial X-rays should be performed every
3}4 months, with clinical assessment of the range of
movement, and gradual weaning from the brace. Some
clinicians keep children in a brace until the lateral column
reossifies and sclerotic areas of the epiphysis are gone.
Problems
E
In practice this form of treatment is not recommended
for the severely involved hip.
E
Pain and spasm may result in pelvic tilting and failure of
containment of the affected hip.
E
Hinge abduction may occur if the supero-lateral
portion of a deformed femoral head impinges on the
lateral acetabulum. When a decreasing range of
abduction results from femoral head overgrowth
132
CURRENT ORTHOPAEDICS
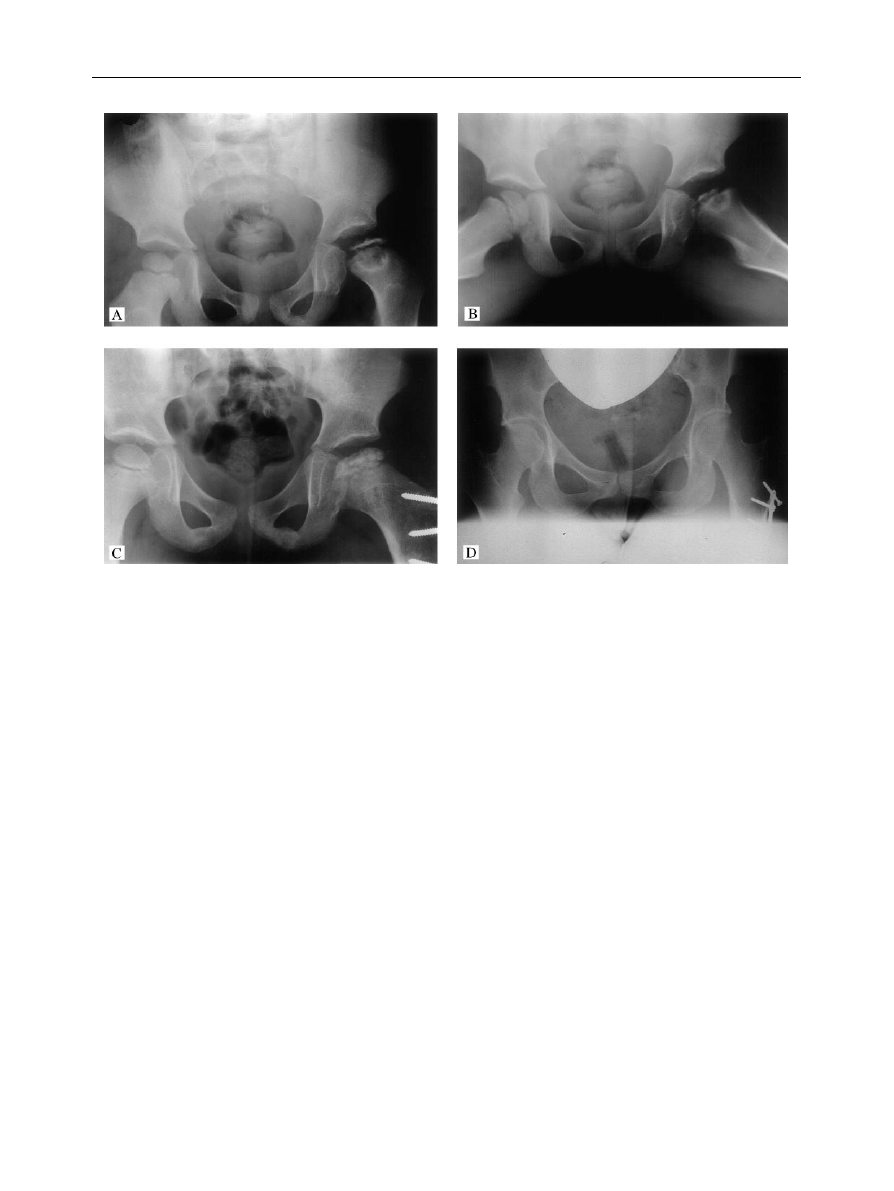
Figure 5
(A, B) Antero-posterior and lateral views of the hip of a girl with Legg}CalveH}Perthes’ disease at presentation. (C) Hip after
a varus femoral osteotomy. (D) Radiological appearance of the hip several years later. (Note, as the operation was performed several
years ago, the technique has changed from that shown; using an intertrochanteric osteotomy and blade plate fixation).
a valgus extension osteotomy can be an effective
salvage procedure. This aims to increase abduction
and bring the more normal medial femoral head into
the weight-bearing area to relieve pain and correct
deformity.
2. Femoral varus osteotomy
Varus osteotomy of the proximal femur centres the
femoral head more deeply within the acetabulum, so that
weight bearing proceeds without bracing (Fig. 5). The aim
is to cover the entire ossified femoral epiphysis with the
ossified acetabulum.
Problems are associated with
a varus neck-shaft angle of less than 1003, especially in the
older child who is less likely to remodel the deformity.
Typically, the varus corrects within 3 years. The greater
trochanter should be kept distal to the level of the
femoral head in order to prevent an abductor lurch.
3. Acetabular osteotomy
Salter recommended that his osteotomy should only be
performed in hips with a full, or almost full, range of
movement and a round or almost round femoral head
with reasonable congruence in abduction. The child
should be more than 6 years old, with over 50% of the
femoral head involved (Salter}Thompson group B) and
subluxation in a weight-bearing position.
In some cases this procedure should be performed in
combination with a femoral osteotomy. The approach
permits containment of a large, deformed femoral head,
avoids an extreme varus neck-shaft angle, maintains leg
length equality and decreases intra-articular pressure.
Other salvage procedures
After reossification late remodelling may still occur.
Salvage procedures include:
E
Excision of the extruded part of the femoral head
(cheilectomy).
E
Acetabular osteotomy.
E
Chiari osteotomy to cover the femoral head.
E
Lateral shelf osteotomy.
E
Arthrodesis at skeletal maturity in unilateral cases for
severe functional impairment.
E
Valgus, extension femoral osteotomy.
E
Combined pelvic and femoral osteotomies if done in
the early stages.
LEGG}CALVED}PERTHES’ DISEASE
133

SUMMARY
One group of children with LCPD only require
symptomatic treatment as there will be little residual
effect from the disease. There is another group which
progresses to a symptomatic arthropathy at an early
age. The challenge of this condition is to differentiate
between these two groups and manage these children
without adversely affecting their life at home and
school.
REFERENCES
1. Rang M. Perthes’ disease, in: Wenger D R, Rang M (eds). The Art
and Practice of Children’s Orthopaedics. New York: Raven Press,
1992; Chapter 10.
2. Barker D J P, Dixon E, Taylor J F. Perthe’s disease of the hip in
three regions of England. J Bone Jt Surg 1978; 60B: 478}480.
3. Catterall A. Legg}CalveH}Perthes’ Disease. Edinburgh: Churchill
Livingstone, 1982.
4. Van de Bogaert G, de Rosa E, Moens P, Fabry G, Dimeglio A.
Bilateral Legg}CalveH}Perthes’ disease: different from unilateral
disease? J Paediatr Orthop B; 8: 165}168.
5. Kealey W D C, Moore A J, Cook S, Cosgrove A P. Deprivation,
urbanisation and Perthes’ disease in Northern Ireland. J Bone Jt
Surg 2000; 82B: 167}171.
6. Purry N A. The incidence of Perthes’ disease in three population
groups of the Eastern Cape region of South Africa. J Bone Jt Surg
1982; 64B: 286}288.
7. Wynne-Davis R. Some etiologic factors in Perthes’ disease. Clin
Orthop Rel Res 1980; 150: 12}15.
8. Mata S G, Aicua E A, Ovejero A H, Grande M M.
Legg}CalveH}Perthes disease and passive smoking. J Paediatr
Orthop 2000; 20: 326}330.
9. Burwell R G, Dangerfield P H, Hall D J, Vernon C L, Harrison
M H M. Perthes’ disease. An anthropometric study revealing
impaired and disproportionate growth. J Bone Jt Surg 1978; 60B:
461}477.
10. Keenan W N W, Clegg J. Perthes’ disease after ‘irritable hip’:
delayed bone age shows the hip is a ‘marked man’. J Paediatr
Orthop 1996; 16: 20}23.
11. Glueck C J, Glueck H I, Greenfield D et al. Protein C and
S deficiency and hypofibrinolysis: pathophysiologic causes of Legg
Perthes’ disease. Pediatr Res 1994; 35: 383}388.
12. Glueck C J, Brandt G, Gruppo R et al. Resistance to activated
protein C and Legg}Perthes’ disease. Clin Orthop 1997; 338:
139}152.
13. Gallistl S, Reitinger T, Linhart W, Muntean W. The role of inherited
thrombotic disorders in the etiology of Legg}CalveH}Perthes’
disease. J Paediatr Orthop 1999; 19: 82}83.
14. Sirvent N, Fisher F, El Hayek T et al. Absence of congenital
prethrombotic disorders in children with Legg}Perthes’ disease.
J Paediatr Orthop B 2000; 9: 24}27.
15. Thomas D P, Morgan G, Tayton K. Perthes’ disease and the
relevance of thrombophilia. J Bone Jt Surg 1999; 81B: 691}695.
16. Kealey W D C, Mayne E E, McDonald W, Murray P, Cosgrove A P.
The role of coagulation abnormalities in the development of
Perthes’ disease. J Bone Jt Surg 2000; 82B: 744}746.
17. Salter R B, Bell M. The pathogenesis of deformity in Legg}Perthes’
diseaseean experimental investigation. J Bone Jt Surg 1968; 50B:
436.
18. Atsumi T, Yamano K, Muraki M, Yoshihara S, Kajihara T. The blood
supply of the lateral epiphyseal arteries in Perthes’ disease. J Bone
Jt Surg 2000; 82B: 392}398.
19. Matsumoto T, Enomoto H, Takahashi K, Motokawa S. Decreased
levels of IGF binding protein-3 in serum from children with
Perthes’ disease. Acta Orthop Scand 1998; 69: 125}128.
20. WaldenstroKm H. The definite form of the coxa plana. Acta Radiol
1922; 1: 384}394.
21. Catterall A, Pringle J, Byers P D. A review of the morphology of
Perthes’ disease. J Bone Jt Surg 1982; 64B: 269}275.
22. WaldenstroKm H. The first stages of coxa plana. J Bone Jt Surg 1938;
20: 559}566.
23. Eggl H, Drekonja T, Kaiser B, Dorn U. Ultrasonography in the
diagnosis of transient synovitis of the hip and Legg}CalveH}Perthes’
disease. J Paediatr Orthop B 1999; 8: 177}180.
24. Robben S G F, Meradji M, Diepstraten A F M, Hop W C J. US of the
painful hip in childhood: the diagnostic value of cartilage thickening
and muscle atrophy in the detection of Perthes’ disease. Radiology
1998; 208: 35} 42.
25. Sutherland A D, Savage J P, Paterson D C, Foster B K. The nuclide
bone-scan in the diagnosis and management of Perthes’ disease.
J Bone Jt Surg 1980; 62B: 300}306.
26. Kaniklides C, LoKnnerholm T, Moberg A, Sahlstedt. Legg}
CalveH}Perthes’ disease. Comparison of conventional radiography,
MR imaging, bone scintigraphy and arthrography. Acta Radiol 1995;
36: 434}439.
27. Jaramillo D, Galen T A, Winalski C S et al. Legg}CalveH}Perthes’
disease. MR imaging evaluation during manual positioning of the
hipecomparison with conventional arthrography. Radiology 1999;
212: 519}525.
28. Catterall A. The natural history of Perthes’ disease. J Bone Jt Surg
1971; 53B: 37}53.
29. Salter R B, Thompson G H. Legg}CalveH}Perthes’ disease. The
prognostic significance of the subchondral fracture and a two-
group classification of the femoral head involvement. J Bone Jt Surg.
1984; 66A: 479}489.
30. Herring J A, Neustadt J B, Williams J J, Early J S, Browne R H. The
lateral
pillar
classification
of
Legg}CalveH}Perthes’
disease.
J Paediatr Orthop 1992; 12: 143}150.
31. Simmons E D, Graham H K, Szalai J P. Interobserver variability in
grading Perthes’ disease. J Bone Jt Surg 1990; 72B: 202}204.
32. Christiansen F, So balle K, Ejsted R, Luxho j T. The Catterall
classification of Perthes’ disease. An assessment of reliability.
J Bone Jt Surg 1986; 68B: 614}615.
33. Wenger D R, Ward W T, Herring J A. Legg}CalveH}Perthes’
disease. J Bone Jt Surg 1991; 73A: 778}788.
34. Catterall A. Legg}CalveH}Perthes’ disease. Clin Orthop Rel Res
1981; 158: 41}51.
35. Schlesinger I, Crider R J. Gage’s signerevisited! J Paediatr Orthop
1988; 8: 201}202.
36. Gage H C. A possible early sign of Perthes’ disease. Br J Radiol
1933; 6: 295}297.
37. Stulberg S D, Salter R B. The natural course of Legg}Perthes’
disease and its relationship to degenerative arthritis of the hip.
A long-term follow-up study. Orthop Trans 1977; 1: 105}106.
38. Stulberg S D, Cooperman D R, Wallensten R. The natural history
of Legg}CalveH}Perthes’ disease. J Bone Jt Surg 1981; 63A:
1095}1108.
39. Neyt J G, Weinstein S L, Spratt K. Stulberg classification system for
evaluation of Legg}CalveH}Perthes’ disease: intra-rater and inter-
rater reliability. J Bone Jt Surg 1999; 1209}1216.
40. Herring J A. The treatment of Legg}CalveH}Perthes’ disease.
A critical review of the literature. J Bone Jt Surg 1995; 76A:
448}458.
134
CURRENT ORTHOPAEDICS
Document Outline
- INTRODUCTION
- HISTORICAL FACTS
- PATHOGENESIS
- CLINICAL FEATURES
- DIFFERENTIAL DIAGNOSIS
- OTHER INVESTIGATIONS AND THEIR VALUE
- CLASSIFICATION OF SEVERITY
- PROGNOSIS AND THE ‘HEAD AT RISK’
- OUTCOMES
- MANAGEMENT
- SUMMARY
- REFERENCES
Wyszukiwarka
Podobne podstrony:
Osteochondritis dissecans in association with legg calve perthes disease
Interruption of the blood supply of femoral head an experimental study on the pathogenesis of Legg C
Modified epiphyseal index for MRI in Legg Calve Perthes disease (LCPD)
Hip Arthroscopy in Legg Calve Perthes Disease
Legg Calve Perthes’ disease
Legg Calve Perthes disease The prognostic significance of the subchondral fracture and a two group c
Computerized gait analysis in Legg Calve´ Perthes disease—Analysis of the frontal plane
Coxa magna quantification using MRI in Legg Calve Perthes disease
Osteochondritis dissecans in association with legg calve perthes disease
Interruption of the blood supply of femoral head an experimental study on the pathogenesis of Legg C
Intertrochanteric osteotomy in young adults for sequelae of Legg Calvé Perthes’ disease—a long term
Legg Calvé Perthes Disease in Czech Archaeological Material
Legg Calvé Perthes disease multipositional power Doppler sonography of the proximal femoral vascular
Multicenter study for Legg Calvé Perthes disease in Japan
Femoral head vascularisation in Legg Calvé Perthes disease comparison of dynamic gadolinium enhanced
A recurrent mutation in type II collagen gene causes Legg Calvé Perthes disease in a Japanese family
Acute chondrolysis complicating Legg Calvé Perthes disease
Legg Perthes disease in three siblings, two heterozygous and one homozygous for the factor V Leiden
Perthes Disease
więcej podobnych podstron