Triazamate
Methomyl
Aldicarb
Carbofuran
Carbaryl
Thiodicarb
Benfuracarb
Carbosulfan
Oxamyl
Methiocarb
Fenobucarb
O
CH
3
CH
3
OCONHCH
3
CH
3
NHCO
2
N
C
SCH
3
CH
3
CH
3
CH
3
C
CH
CH
3
S
NOCONHCH
3
OCONHCH
3
CH
3
NCO
2
N C
SCH
3
CH
3
S
CH
3
NCO
2
N C
SCH
3
CH
3
O
OCO
CH
3
CH
3
CH
3
N S
NCH
2
CH
2
CO
2
CH
2
CH
3
CH(CH
3
)
2
O
CH
3
CH
3
OCO
CH
3
N S N[(CH
2
)
3
CH
3
]
2
(CH
3
)
2
NCOC
NOCONHCH
3
SCH
3
O
C
O
CH
3
NH
SCH
3
CH
3
CH
3
O
CHCH
2
CH
3
CH
3
CH
3
NHC
O
1A Carbamates
1B Organophosphates
Chlorpyrifos
Dimethoate
Profenofos
Monocrotophos
Acephate
Malathion
Methamidophos
Terbufos
Parathion-methyl
Diazinon
N
Cl
Cl
Cl
OP(OCH
2
CH
3
)
2
S
CH
3
NHCOCH
2
SP(OCH
3
)
2
S
O
CH
3
SP
OCH
3
NHCOCH
3
S
(CH
3
O)
2
PS
CO
2
CH
2
CH
3
CHCH
2
CO
2
CH
2
CH
3
O
NH
2
CH
3
OPSCH
3
(E )
CONHCH
3
O
(CH
3
O)
2
P
O
H
C
C
CH
3
S
P(OCH
2
CH
3
)
2
(CH
3
)
3
CSCH
2
S
O
2
N
OP(OCH
3
)
2
S
N
N
CH
3
(CH
3
)
2
CH
OP(OCH
2
CH
3
)
2
S
SCH
2
CH
2
CH
3
P
O
Br
Cl
OCH
2
CH
3
O
Group 1: Acetylcholinesterase (AChE) inhibitors
(Only major representatives of the groups are shown)
Group 2: GABA-gated chloride channel antagonists
2B Phenylpyrazoles (Fiproles)
2A Cyclodiene Organochlorines
Cl
Cl
Cl
Cl
Cl
Cl
Cl
Cl
Chlordane
Cl
Cl
Cl
Cl
Cl
Cl
O
SO
O
Endosulfan
F
3
C
N
Cl
Cl
N
CN
S
O
CF
3
NH
2
Fipronil
N
N
Cl
Cl
CF3
H2N
CN
CH3CH2SO
Ethiprole
3A Pyrethroids
Pyrethrins
Deltamethrin
Lambda-
cyhalothrin
Cypermethrin
Bifenthrin
Alpha-
cypermethrin
Esfenvalerate
Tefluthrin
Cyfluthrin
Zeta-
cypermethrin
Etofenprox
CN
O
C
H
CO
2
CH
3
CH
3
C
Br
Br
CH
H
H
(Z)-(1R)-cis -
(Z)-(1S)-cis -
(R)
(S)
CH
3
CH
3
CH
C
C
F
3C
Cl
H
H
O
2
C
CN
O
H
CH
3
C
CH
CH
3
C
F
3C
Cl
H
H
CN
O
2
C
H
O
C
CH
Cl
Cl
CH
3
CH
3
CO
2CH
O
CN
(Z)-(1R)-cis-
(Z)-(1S)-cis-
CH
3
H
2
CO
2C
CH
3
CH
3
CH
C
F
3C
Cl
H
H
CO
2C
CH
C
F
3C
Cl
CH
3
CH
3
H
H
CH
3
H
2
(1R)-cis -
(1S)-cis -
(R)
(S)
CH
3
CH
3
CH
C
C
Cl
Cl
H
H
O
2
C
CN
O
H
CH
3
C
CH
CH
3
C
Cl
Cl
H
H
CN
O
O
2
C
H
O
C
CN
O
C
CH(CH
3)2
Cl
O
H
H
(Z)-(1S)-cis-
(Z)-(1R)-cis-
C CH
F
3C
Cl
CH
3
CH
3
H
H
F
F
F
F
CH
3
CO
2CH2
CH
3
CH
3
CO
2CH2
F
F
F
F
CH
3
H
H
CH
F
3C
Cl
C
O
CH
CN
F
CH
3
CH
3
CH
C
Cl
Cl
CO
2
O
CH
3
CH
3
CO
2CH
CH
C
Cl
Cl
CN
CH
3
CH
2
O
C
CH
3
CH
2
OCH
2
CH
3
O
Pyrethrins
(Pyrethrum)
DDT
Methoxychlor
Cl
CH
CCl
3
Cl
CH
3
O
CH
CCl
3
OCH
3
R = -CH3 (chrysanthemates) or -CO2CH3 (pyrethrates)
R1 = -CH=CH2 (pyrethrin) or -CH3 (cinerin) or -CH2CH3 (jasmolin)
H
CH3 CH3
C
CH
H
O
O H
CH3
O
CH2 C
C
R1
H
H
C
CH3
R
3B DDT,
Methoxychlor
Group 3: Sodium channel modulators
(Only major representatives of group 3A are shown)
4A Neonicotinoids
Acetamiprid
N
C
CH
3
CH
3
N
CN
CH
2
N
Cl
Clothianidin
N
Cl
CH
2
NH
H
N
CH
3
N
S
NO
2
Dinotefuran
O
H
N
H
N
CH
3
N
NO
2
Imidacloprid
N
Cl
CH
2
N
N
N
NO
2
H
Nitenpyram
N
C
CH2CH3
CH3NH
C
NO2
CH2
N
Cl
H
Thiacloprid
S
N
N
Cl
CH
2
N
CN
Thiamethoxam
S
N
CH
2
N
O
N
Cl
N
CH
3
NO
2
Group 4: Nicotinic acetylcholine receptor (nAChR) agonists
4B Nicotine
N
CH
3
N
H
Nicotine
5 Spinosyns
Group 5: Nicotinic acetylcholine
receptor (nAChR) allosteric modulators
Spinosad
O
O
O
CH3
(CH3)2N
CH3
CH3CH2
HH
H H
R
H
O
O
O
O
OCH3
OCH3
CH3
OCH3
spinosyn A, R = H-
spinosyn D, R = CH3-
Spinetoram
Group 6: Chloride channel activators
6 Avermectins, Milbemycins
Abamectin
O
CH3
CH3
CH3
O
O
O
OH
CH3
OH
O
O
H
H
H
OCH3
O
CH3
O
OCH3
HO
O
CH3
CH3
H
H
R
(i) R = -CH2CH3 (avermectin B1a)
(ii) R = -CH3 (avermectin B1b)
Emamectin
benzoate
NH2
CH3
O
O
O
CH3
CH3
O
O
O
OH
CH3
HO
O
H
H
O
CH3
R
CH3
H
O
CH3
CH3
OCH3
CH3O
CO2
B1a R = CH3CH2-
B1b R = CH3-
Milbemectin
CH3
CH3
O
O
CH3
R
O
O
O
CH3
OH
OH
H
H
H
H
Milbemycin A3: R = -CH3
Milbemycin A4: R = -CH2CH3
7A Juvenile hormone analogues
7B Fenoxycarb
7C Pyriproxyfen
C
C
C
C
CH2
CH
CH2
CH2
CH2
CH
CH3
CH3
CH3
CH3
H
H
H
CO2CH2CH3
Hydroprene
Kinoprene
C
C
C
C
CH2
CH
CH2
CH2
CH2
CH
CH3
CH3
CH3
CH3
H
H
H
CO2CH2C CH
Methoprene
Fenoxycarb
Pyriproxyfen
O
OCH
2
CH
2
NHCO
2
CH
2
CH
3
N
O
CH
CH
3
CH
2
O
O
CH
2
C C
C C
CH
3
CO
2
CH(CH
3
)
2
H
H
H
CH
(CH
2
)
3
CH
3
(CH
3
)
2
C
OCH
3
8A Alkyl halides
8B Chloropicrin
8C Sulfuryl fluoride
CH
3
Br
Cl
3
C NO
2
S
O
O
F
F
Chloropicrin
Sulfuryl
fluoride
Methyl
bromide
Tartar emetic
Borax
Na B O 10H O
O
O
Sb
O
O
O
O
O
O
Sb
O
O
O
O
.
2K+ .3H
2O
-
-
-
-
-
-
-
-
3+
3+
8D Borax
8E Tartar emetic
Group 9: Selective homopteran
feeding blockers
9B Pymetrozine
9C Flonicamid
N
CH
N
N
N
H
N
CH3
O
N
CF3
CONHCH2CN
Pymetrozine
Flonicamid
10A Clofentezine
10B Etoxazole
Group 10: Mite growth inhibitors
10A Hexythiazox
Cl
N N
N
N
Cl
N
S
O
Cl
CH3
NH
C
O
CH2CH3
O
C(CH3)3
F
F
O
N
Clofentezine
Hexythiazox
Etoxazole
Group 11: Microbial disruptors of insect midgut membranes and derived toxins
Group 7: Juvenile hormone mimics
Group 8: Miscellaneous non-specific (multi-site) inhibitors
12C
Propargite
12D
Tetradifon
12B Organotin
miticides
Group 12: Inhibitors of mitochondrial ATP synthase
Diafenthiuron
Azocyclotin
Cyhexatin
Fenbutatin
oxide
Propargite
Tetradifon
O
CH(CH
3
)
2
NHCSNHC(CH
3
)
3
CH(CH
3
)
2
Sn
N
N
N
OH
Sn
C CH2
CH3
CH3
C
CH2
CH3
CH3
Sn O Sn
3
3
C(CH
3
)
3
O
OSO
2
CH
2
C CH
Cl
SO
2
Cl
Cl
Cl
12A
Diafenthiuron
Bensultap
SO
2
S CH
2
CH
CH
2
S
N(CH
3
)
2
SO
2
CH
2
CH
2
H
2
NCOS
CH
H
2
NCOS
N(CH
3
)
2
.HCl
S
S
S
(CH
3
)
2
N
CH
CH
2
SSO
3
Na
CH
2
SSO
3
Na
CH
3
N
CH
3
Thiocyclam
Thiosultap-
sodium
Cartap
hydrochloride
Group 13: Uncouplers of oxidative phos-
phorylation via disruption of proton gradient
Chlorfenapyr
DNOC
N
CH2OCH2CH3
CF3
Br
CN
Cl
O
2
N
OH
CH
3
NO
2
Group 14: Nicotinic acetylcholine
receptor (nAChR) channel blockers
14 Nereistoxin
analogues
Group 16: Inhibitors of
chitin biosynthesis,
type 1
N
N
S
NC(CH
3
)
3
CH(CH
3
)
2
O
Buprofezin
Group 17: Moulting
disruptor, Dipteran
N
N
N
H
2
N
NH
2
NH
Cyromazine
HN N
O
O
C(CH
3
)
3
CH
3
CH
3
CH
3
O
Chromafenozide
CH
3
O
CH
3
NH N
O
O
C(CH
3
)
3
CH
3
CH
3
HN N
O
O
C(CH3)3
Cl
HN N
O
O
C(CH
3
)
3
CH
3
CH
3
CH
3
CH
2
Halofenozide
Methoxyfenozide
Tebufenozide
18 Diacyl-
hydrazines
Group 18: Ecdysone receptor agonists
Group 20: Mitochondrial complex III electron
transport inhibitors
CF
3
CH
CH
C
CH
CH
N
N
NH
NH
CH
3
CH
3
CF
3
CH
3
N
CH
3
CH
CH
CH
3
CH
3
CH
3
N
N
Amitraz
Hydramethylnon
20A
20B
O
O
O
(CH
2
)
11
CH
3
COCH
3
Acequinocyl
C
CH
O
O
O
CH
3
CH
2
O
N
N
CF
3
O
CH
CH
3
CH
3
CH
3
20C
Fluacrypyrim
Fenazaquin
Fenpyroximate
Pyrimidifen
Pyridaben
Tebufenpyrad
Tolfenpyrad
Rotenone
CH
3
O
OCH
3
O
O
O
C
CH
3
CH
2
O
H
H
CH2
C
NH
O
N
N
CH3
CH3CH2
Cl
O
CH3
CH2
C
NH
O
C(CH3)3
N
N
CH3CH2
Cl
CH3
(CH
3
)
3
C
CH
2
S
N
N
Cl
O
C(CH
3
)
3
N
N
CH3CH2
Cl
CH3
CH3
(CH2)2OCH2CH3
CH2
O
NH
CH2
C
O
N
C
N
N
CH
3
O
CH
3
H
O
CH
2
O C(CH
3
)
3
N
N
O
CH2
CH2
C(CH3)3
Group 21: Mitochondrial complex I electron transport inhibitors
21A METI acaricides and insecticides
21B Rotenone
Indoxacarb
O
N N
N
O
CO
2
CH
3
CO
2
CH
3
Cl
OCF
3
Group 23: Inhibitors of acetyl
CoA carboxylase
Metaflumizone
22A Indoxacarb
22B Metaflumizone
23 Tetronic & Tetramic acid derivatives
Spirodiclofen
Spiromesifen
C
CH2
C(CH3)3
O
O
O
O
CH3
CH3
CH3
O
O
O
C
O
Cl
Cl
CH3 CH
2CH3
CH3
Group 24: Mitochondrial complex IV electron transport inhibitors
24A
Phosphine
Aluminium
Phosphide
24B Cyanide
Cyanide
CN-
PH
3
Calcium
Phosphide
Zinc
Phosphide
Phosphine
Al PH
3
Zn PH
3
Ca PH
3
O
OH
CO
2
CH
3
O
O
CH
3
OC
O
O
OH
C
CH
3
CH
3
O
CH
3
O
O
O
HO
CH3
H
O
CH
3
C
Azadirachtin
Pyridalyl
Dicofol
Chinomethionat
Benzoximate
O
O
O
N
Cl
Cl
CF
3
Cl
Cl
Cl
C
OH
CCl3
Cl
N
N
S
S
O
CH
3
C
C
NOCH2CH3
OCH3
Cl
CH3O
O
O
Bifenazate
NHNH
OCH
3
COOCH(CH
3
)
2
Al
F
F
F
F
F
F
3Na
Cryolite
Group UN: Compounds of unknown or uncertain mode of action
15 Benzoylureas
Group 15: Inhibitors of chitin biosynthesis, type 0
Flucycloxuron
Flufenoxuron
Hexaflumuron
Lufenuron
Novaluron
Noviflumuron
Teflubenzuron
Triflumuron
CH2
F
F
NHCONHCO
O
N
C
Cl
F
F
F
NHCONHCO
O
CF3
Cl
Cl
F
F
NHCONHCO
Cl
CHF
2
CF
2
O
Cl
CF
3
CHFCF
2
O
Cl
F
F
NHCONHCO
Cl
CF3OCHFCF2O
F
F
NHCONHCO
NH
C
O
NH
C
O
F
F
CF
3
CHFCF
2
O
F
Cl
Cl
Cl
F
F
F
F
NHCONHCO
Cl
NHCONHCO
Cl
CF
3
O
Chlorfluazuron
Diflubenzuron
Cl
F
F
NHCONHCO
CONHCONH
F
F
O
N
CF
3
Cl
Cl
Cl
Bistrifluron
Group 19: Octopamine
receptor agonists
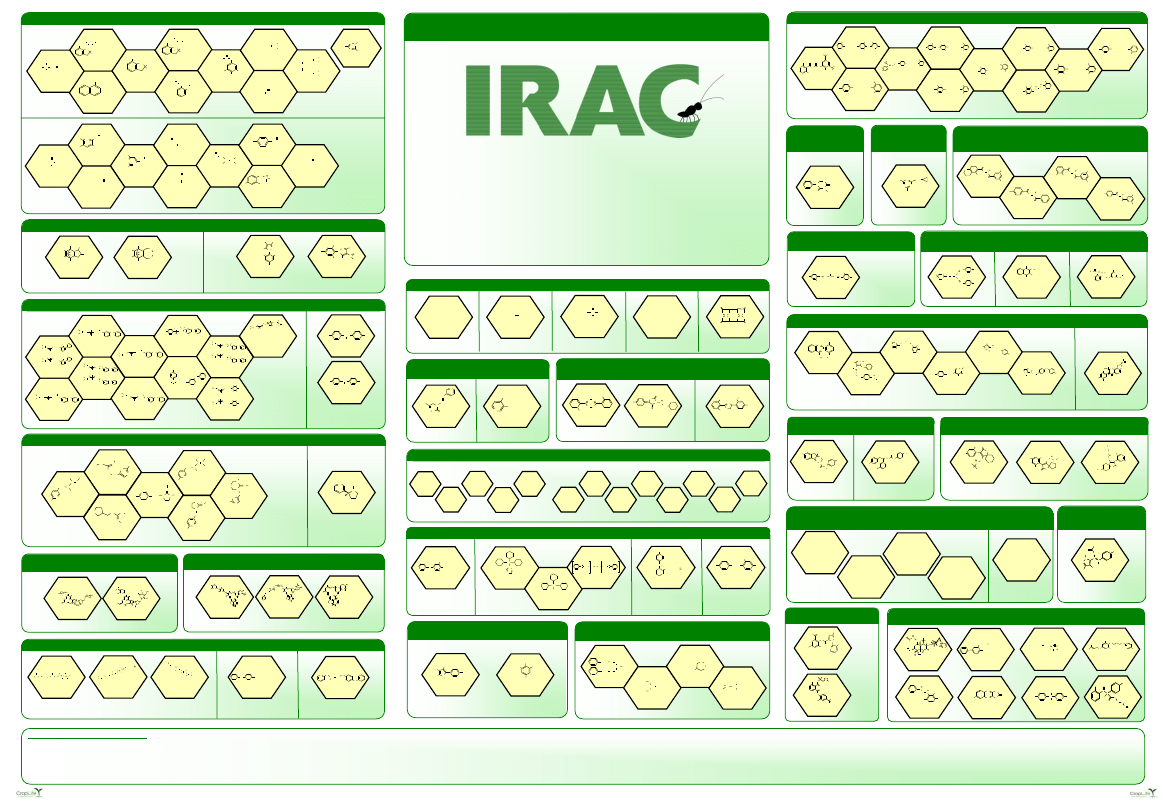
Mode of Action Classification
Insecticide Resistance Action Committee
The Key to Resistance Management
Guidance on the use of Sub-Groups:
• Represent distinct structural classes believed to have the same mode of action
• Provides differentiation between compounds that may bind at the same target site
• Are structurally different such that risk of metabolic cross-resistance is lower than for close chemical analogs
• Are likely to be metabolized by different enzymes - may bind differently enough within the target site that the chance of
selection for metabolic/target-site resistance is reduced compared to close analogs.
• 3A & 3B - If there are no other alternatives, compounds may be rotated in situations where cross-resistance mechanisms (e.g.
kdr) are known to be absent in the insect populations to be treated. DDT is no longer used in agriculture and therefore this is
only applicable for the control of human disease vectors such as mosquitoes, because of a lack of alternatives.
•10A - Clofentezine & Hexythiazox are grouped because they commonly exhibit cross-resistance even though they are structurally
distinct, and the target site for neither compound is known.
• 22A & 22B - Although these compounds are believed to have the same target site, they have been sub-grouped because they
are chemically distinct, and current evidence indicates that the risk of metabolic cross-resistance is low.
• In the absence of other alternatives, it may be possible to rotate compounds between sub-groups if it is clear that cross
resistance mechanisms do not exist in the target populations.
• Not all of the current groupings are based on knowledge of a shared target protein. For further information please refer to the
IRAC Mode of Action Classification document.
•1A & 1B - If there are no other alternatives, compounds may be rotated in situations where cross-resistance mechanisms are
known to be absent in the insect populations to be treated.
More information on IRAC and the Mode of Action Classification is available from:
www.irac-online.org or enquiries@irac-online.org
Structures are reproduced from the Pesticide Manual with permission from the British Crop Protection Council
Poster Version 2.5, July 2009. Based on the Mode of Action Classification - Version 6.3
The poster is for educational purposes only. Details presented are accurate to the best of our knowledge at the time of publication but IRAC or its member companies cannot accept responsibility for how this information is used or interpreted
N
CF
3
N
H
N
H
O
O
CF
3
NC
N
H
O
O
N
H
F
F
Cl
F
F
F
F F
F
B.t.
israelensis
B.t.
aizawai
B.t.
aizawai
B.
sphaericus
B.t.
kurstaki
B.t.
tenebrionis
Cry1Ab
Cry1Ac
Cry1Fa
Cry2Ab
mCry3A
Cry3Ab
Cry3Bb
Cry34/35Ab1
Spirotetramat
N
H
O
O
O
O
CH
2
C
H
3
O
C
H
3
C
H
3
CH
3
O
O
O
O
O
N
O
O
O
O
O
H
H
H
H
H
R
5
6
C5 C6, R = H
C5 C6, R = CH3
-
=
Group 22: Voltage-dependent
sodium channel blockers
N
N
N
(CH
3
)
3
C
CON(CH
3
)
2
SCH
2
CO
2
C
2
H
5
13 Pyrroles, Dinitrophenols
16 Buprofezin
17 Cryomazine
19 Amitraz
25 Cyenopyrafen
Group 25: Mitochondrial
complex II electron
transport inhibitors
Cyenopyrafen
Cyflumetofen
Group 28: Ryanodine
receptor modulators
Chlorantraniliprole
28
Diamides
N
H
CH
3
O
Cl
O
N
N
N
Cl
Br
N
H
C
H
3
S
O
O
I
O
N
H
N
H
O
CF
3
CF
3
F
Flubendiamide
CF
3
C
C
O
NC
C(CH
3
)
3
O
O
H
2
C
CH
2
O
CH
3
C
C
CN
C(CH
3
)
3
N
N
O
H
3
C
CH
3
H
3
C
C(CH
3
)
3
O
Wyszukiwarka
Podobne podstrony:
BPMN2 0 Poster EN id 92566 Nieznany (2)
BPMN Poster PL id 92560 Nieznany (2)
bpmn 2 posterPL id 92565 Nieznany (2)
Grad SW Dev and Test Poster id Nieznany
poster id 378305 Nieznany
BPMN2 0 Poster EN id 92566 Nieznany (2)
Gor±czka o nieznanej etiologii
02 VIC 10 Days Cumulative A D O Nieznany (2)
Abolicja podatkowa id 50334 Nieznany (2)
45 sekundowa prezentacja w 4 ro Nieznany (2)
4 LIDER MENEDZER id 37733 Nieznany (2)
Mechanika Plynow Lab, Sitka Pro Nieznany
katechezy MB id 233498 Nieznany
2012 styczen OPEXid 27724 Nieznany
metro sciaga id 296943 Nieznany
Mazowieckie Studia Humanistyczn Nieznany (11)
cw 16 odpowiedzi do pytan id 1 Nieznany
więcej podobnych podstron