
What is Acid-base balance
Acid-base balance is defined by the
concentration of hydrogen ions.
In order to achieve homeostasis, there
must be a balance between the intake or
production of hydrogen ions and the net
removal of hydrogen ions from the body.
An Acid
Molecules containing
hydrogen atoms that
can release
hydrogen ions in
solutions are referred
to as an acid.
An example of an
acid is hydrochloric
acid (HCL)

A Base
A base is an ion that
can accept a
hydrogen ion.
An example of a base
is is the bicarbonate
ion.( HCO3

How is Acid-Base balance
measured
Hydrogen ion concentration is expressed
on a logarithm scale using pH units
(part/percentage hydrogen).
7.0 being neutral
Body systems carefully control pH of the
body within the range of 7.35-7.45

pH
A low pH corresponds
to a high hydrogen
ion concentration
The term “Acidosis”
refers to the
addition of excess
hydrogen ions and
the body has a pH
that falls below 7.35
A high pH corresponds
to a low hydrogen
concentration
The term “Alkalosis”
refers to excess
removal of hydrogen
ions from the body
and has a pH that
rises above 7.45

How the Body defends
against fluctuations in pH
Three Systems in the body:
1.
Buffers in the blood
2.
Respiration through the lungs
3.
Excretion by the kidneys

Buffers in the Blood
Buffers are substances that neutralize
acids or bases
Bicarbonate which is a base and carbonic
acid in the body fluids protect the body
against changes in acidity
These buffer systems serve as a first line
of defense against changes in the acid-
base balance

Respiration through the lungs
Carbon Dioxide which is formed during
cellular metabolism forms carbonic acid
in the blood decreasing the pH
When the pH drops respiration rate
increases this hyperventilation increases
the amount of CO2 exhaled thereby
lowering the carbonic acid concentration
and restoring homeostasis

Excretion by the Kidneys
The kidneys play the primary role in
maintaining long term control of Acid-
Base balance
The kidney does this by selecting which
ions to retain and which to excrete
The kidneys adjust the body’s Acid-Base
balance

The Importance of the Body’s
Buffering Systems
Can be quickly realized if one considers the
low concentration of hydrogen ions in the body
fluids and the relatively large amounts of acids
produced by the body each day
Example: 80 milliequvilalants of hydrogen is
either ingested or produced each day by
metabolism.
Whereas the hydrogen ion concentration of
the body fluids normally is only about .
0004meq/L

There are 4 Types of Acid-
base Imbalances
Respiratory Alkalosis
Respiratory Acidosis
Metabolic Alkalosis
Metabolic Acidosis

Respiratory Alkalosis
Is a decrease in CO2, decrease in H+
(Hydrogen ions) and in increase in pH
This condition can be caused by severe
exercise, or anxiety reaction
The body compensates by the lungs
slowing the respiration rate and kidneys
excreting more bicarbonate

Respiratory Acidosis
Is an increase in CO2, an increase in H+ and
a decrease in pH
This condition can be caused by emphysema,
restrictive or obstructive lung diseases
The body compensates by the lungs
increasing respiration rate and the kidneys
by conserving bicarbonate ions and
increasing renal net acid excretions

Metabolic Alkalosis
Is a decrease in CO2, a decrease in H+
and an increase in pH
This condition can be caused by
vomiting, diarrhea, increased ingestion of
alkali
The body compensates by the lungs
creating a slow respiration rate and the
kidneys excreting more bicarbonate

Metabolic Acidosis
Is an increase in CO2, and increase in H+
and a decrease in pH
This condition can be caused by diabetes,
diarrhea, alkaline loss,excess acid
production or ingestion
The body compensates by the lungs
increasing respiration rate and the kidneys
increasing renal net acid excretion

How does the diet effect Acid-
base balance
After nutrients from
food are metabolized in
the cell they may yield
an excess of mineral
elements that are
potentially acid or base
This consideration is
based on whether the
food ingested contains
more cations or anions

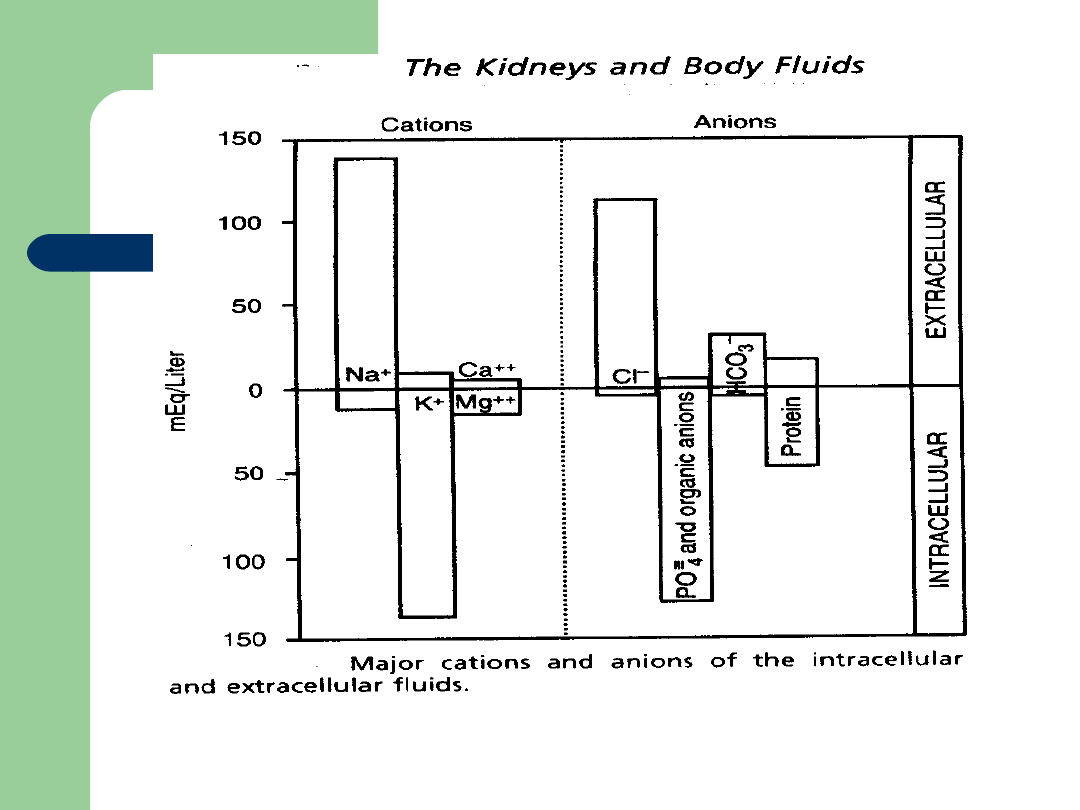
Food Components
Cations
Have a positive
charge
Na+
K+
Ca+
Mg+
Anions
Have a negative
charge
Cl-
HCO
3
-
S-
P-
Organic Acids
Proteins

What happens when we put
our bodies into acid overload
Frassetto (1998) stated
“Normal adults eating
an average American
diet (as reported in the
National Health and
Nutrition Examination
Survey)
characteristically have
chronic, low grade
metabolic acidosis”.

“Metabolic Acidosis”
This persisting upsetting of the body’s Acid-Base
balance occurs because metabolism of the diet
releases noncarbonic acids into the systems
circulation (e.g. sulfuric acid from the metabolism
of proteins) in amounts that exceeds the base
released (e.g. bicarbonate from the metabolism of
potassium in vegetables foods
The size of the difference between acid and base
production determines the net acid load of the diet

Metabolic Acidosis
This condition
overloads the bodies
buffering systems
and they cannot keep
up with the necessary
excretions of acids in
the body.
Four ways in which to
measure Acid-base balance in
the body
1.
Blood pH
2.
Urine pH
3.
Renal net acid
excretion
4.
Net acid load
Studies conducted on Diets
and Acid-base balance

Subjects and Methods
141 subjects ranging in ages from 17-73
20 different whole food diets for a period
of one week(previously shown to be
sufficient for establishing a steady state
of acid-base balance)
Diets: Values for protein, potassium were
taken for the 20 diets ingested

Diets &
Results
Protein content
ranged from 39-
193g/d
Potassium content
ranged from 40-
133mEq/d
Renal net acid
excretion then ranged
from 12-136 mEq/d
Protein content and
potassium content
were independent
predictors of renal net
acid excretion
Renal Net Acid
Excretion
1.
For protein= 0.94
2.
For potassium = -0.61

Results
This suggests that increasing protein content
increases renal net acid excretion and
increasing potassium content decreases it.
The results here indicate that in normal subjects
steady state renal net acid excretion is highly
correlated with the ratio of the dietary content
of total protein to potassium.
The information for the 2 components, protein
and potassium used in this diet is widely
available in standard food composition tables

Results
Given that steady state Renal net acid
excretion in normal subjects corresponds
closely with the diet-dependent rate of
acid-production. These results provide a
relatively simple and reliable method for
determining and controlling the net acid
load of the diet.

Subjects and Methods
6 Adults
3 Diets were given that consisted of Low
protein 49(g/d), Moderate Protein 95(g/d)
and High Protein 120(g/d)
Diets consisted of foods for which
nutrient composition was taken and
analyzed

Diets
The study was carried out in 4
consecutive diet periods, was a repeated
measure design in which all subjects
received the above diets in the same
chronological order.
Each diet period lasted 5 days
During each 5 day period the respective
food diet was kept constant

Results
During the last 2 days of each diet period
timed 24 hour urine samples were
collected
Acid-base status was determined
The mean urinary pH for the low protein
diet was 6.7, for the moderate protein
diet it was a pH of 5.4 and the high
protein diet was a urinary pH of 5.5

Conclusion
This study then concluded that urine pH
values confirmed that diet composition
can significantly influence acid-base
balance

What happens in the body
during Acid-Base imbalances
Researchers Barzel and Massey (1998)
stated that “The average American diet
which is high in protein and low in fruits
and vegetables generates a large amount
of acid and this type of diet can
adversely effect bone.”
The key in this statement is “large
amount of acid”

The kidney’s can compensate for only so
much but when there is too much acid in the
body the kidney can’t keep up so then other
body systems move in to help neutralize the
acid
One of these systems is a release of calcium
(Ca+) from our bones.This helps to act as a
buffer to neutralize the excess amount of
acid in the body

Calcium in the urine is directly related to
net acid excretion
A diet high in acid foods causes excessive
calcium loss
Overtime it is the balance of dietary acid
and base that determines calcium
balance

Calcium
“Bone and Mineral investigators should
look at acid-base effects of the diet and
use appropriate methods to measure
these affects. The 24-hour urine
collection as a part of total calcium
balance measurement is the gold
standard of acid-base research”

Study
Todd (2000) stated in her research that hip
fractures is a major health problem in the
elderly and is directly related to animal
protein intake.
They analyzed reported hip fracture
incidences among different countries in
women aged 50 years and older in relation to
their consumption of vegetables and animal
foods.

Study cont.
Hip fracture incidences correlated directly
with the ratio of vegetable and animal protein
intake and accounted for 70% of the total
variation in hip fracture incidences
Conclusion was stated as the finding suggest
that the critical determinate of hip fracture
risk in relation to the acid-base effects of diet
is the net acid load of the diet

Study
Kerstetter, Brien and Insogna (2003) stated in
their research that “Almost 30 million Americans
are affected by Osteoporosis and women are 4x
more likely to suffer from this disease than men.
The health problem is reaching near epidemic
proportions in the United States and world wide.
Less than 30-50% of U.S. adults consume
dietary protein that could be considered
moderate

Study cont.
Given the increasing prevalence of
osteoporosis and the clear impact dietary
protein has on calcium metabolism it is
imperative that we gain a better
understanding of the complex interplay
between dietary protein and skeletal
health”

Systemic effects of metabolic
acidosis
Cardiovascular
1.
Heart: Bradycardia; arrhythmias;
reduced contractility
2.
Vascular: Arteriolar dilation; increased
venous tone

Systemic effects of metabolic
acidosis
Gastrointestinal: Gastric distention;
Intestinal effects
Renal: Sodium/potassium wasting; Uric
acid retention; Hypercalciuria
Electrolytes:
Potassium,calcium,magnesium,phosphor
us

Systemic effects of Metabolic
acidosis
Metabolic/Hormonal
1.
Protein wasting
2.
Organic acid synthesis;
3.
Aldosterone secretion may be increased;
4.
Parathyroid hormone;
5.
Action on bone enhanced;
6.
Vitamin D activation may be impaired

Standard of Care
Respiratory Alkalosis- slowing down the
respiration rate and increasing the CO2
production
Respiratory Acidosis- increasing minute
ventilation and decreasing CO2
production

Standard of Care
Metabolic Alkalosis- replacing potassium
losses, and trying to restore electrolyte
imbalances
Metabolic Acidosis- ventilation must be
increased, less acid production or
ingestion, and try to increase the
systemic pH

Recommended Treatment
Consultation with Patient
Diet History
Lab Values
pH testing to see if there are in acid-base
balance
Create an individualized nutritional plan
for your patient

Recommended Treatments
Putting patient on a diet that help to maintain
the acid-base balance in their bodies
We have sources available to us as Dietitians
one of these is from the USDA Nutrient Data
Laboratory which has Lists of Food Composition
Tables
This lists 1000’s of foods and the nutrient
contents of them
Webpage is www.nal.usda.gov/fnic/foodcomp/
Document Outline
- Slide 1
- Slide 2
- Slide 3
- Slide 4
- Slide 5
- Slide 6
- Slide 7
- Slide 8
- Slide 9
- Slide 10
- Slide 11
- Slide 12
- Slide 13
- Slide 14
- Slide 15
- Slide 16
- Slide 17
- Slide 18
- Slide 19
- Slide 20
- Slide 21
- Slide 22
- Slide 23
- Slide 24
- Slide 25
- Slide 26
- Slide 27
- Slide 28
- Slide 29
- Slide 30
- Slide 31
- Slide 32
- Slide 33
- Slide 34
- Slide 35
- Slide 36
- Slide 37
- Slide 38
- Slide 39
- Slide 40
- Slide 41
- Slide 42
- Slide 43
- Slide 44
- Slide 45
- Slide 46
Wyszukiwarka
Podobne podstrony:
Wykład X Podstawy równowagi kwasowo zasadowej
10 GOSPODARKA WODNO ELEKTROLITOWA I RÓWNOWAGA KWASOWO ZASADOWAid 10549 ppt
ZABURZENIA RÓWNOWAGI KWASOWO-ZASADOWEJ, Wykłady
10 11 2010 Równowagi kwasowo zasadowe
10 GOSPODARKA WODNO ELEKTROLITOWA I RÓWNOWAGA KWASOWO ZASADOWAid 10549 ppt
Równowaga kwasowo zasadowa fizjot3
Równowaga kwasowo zasadowa fizjot3
Fizjologia kolo RKZ, II rok, II rok CM UMK, Giełdy, od Joe, FIZJOLOGIA, KOLOKWIA, RÓWNOWAGA KWASOWO-
woda jako składnik żywności oraz równowagi kwasowo- zasadowe w żywności, żywienie człowieka i ocena
01 rownowaga kwasowo zasadowa
SPEKTROFOTOMETRYCZNE?DANIE RÓWNOWAG KWASOWO ZASADOWYCH W ROZTWORACH WODNYCH sprawozdanie równowag
wyklad 16 Kataliza kwasowo zasadowa
więcej podobnych podstron