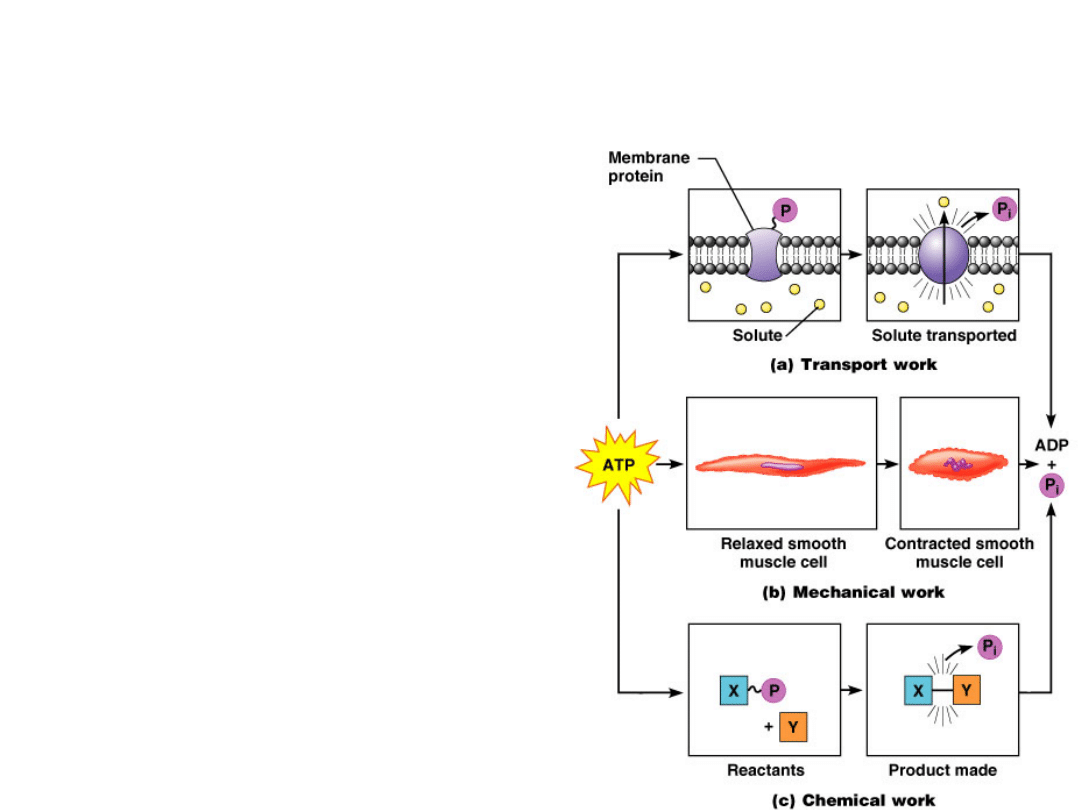
Membrane
potentials
Energy Currency
"High energy" chemical
species
Redox reactions
Transport
works
Energy Utilization
Chemical
reactions
Mechanical
work
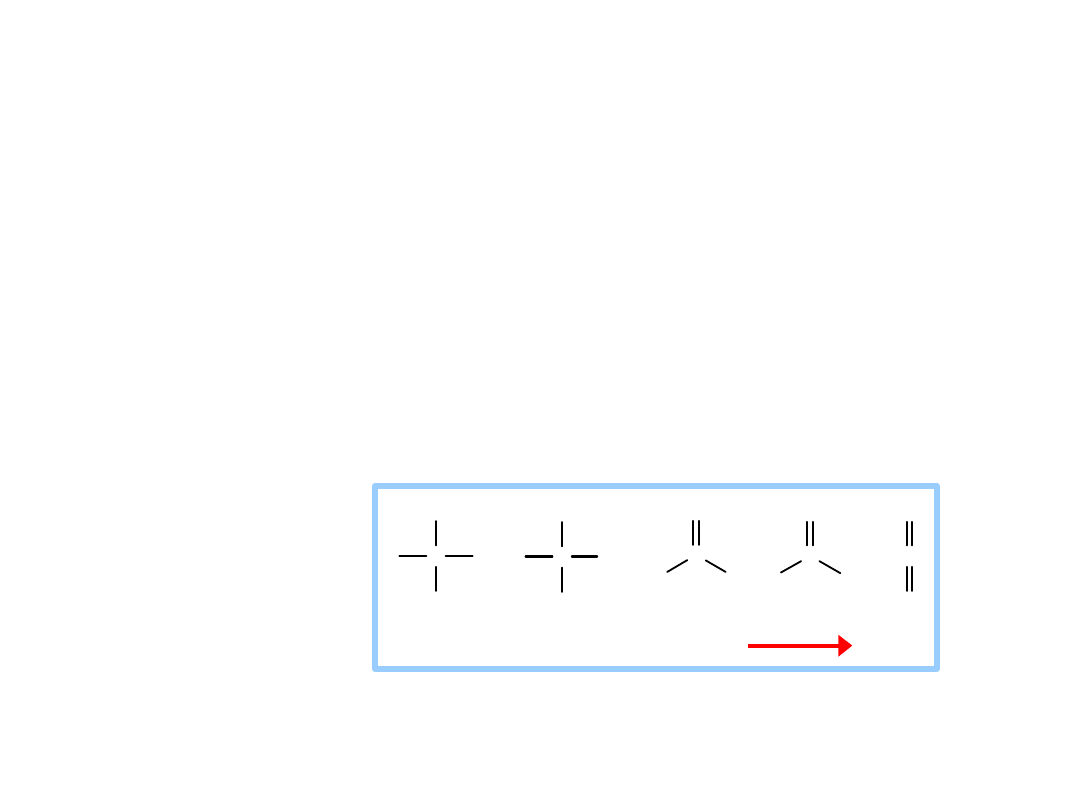
Oxidation of carbon atoms of
glucose is the major source of
energy in aerobic metabolism
Gain of electrons or hydrogen =
reduction =
energy increase
Loss of electrons or hydrogen =
oxidation =
energy decrease
Redox reactions
Oxidation of
carbon is
spontaneous.
In c re a s in g o x id atio n o f c a rb o n
H
C
H
H
H
H
C
H
O H
H
H
C
H
O
O
C
O
O H
C
H
O

]
[
]
[
ln
'
ox
red
o
A
A
nF
RT
E
E
Redox
potential - empirical measure of tendency to gain e's
Where [
A
red
] = [
A
ox
], E = E°'
E°'
is the
mid-point potential
, or standard redox
potential, the potential at which
[oxidant] =
[reductant]
for the half reaction.
ox
red
red
ox
B
A
B
A
Whenever one substance is oxidized, another substance is reduced
The more negative the potential
, the greater the
tendency of the reduced compound to donate electron
(reducing ability).
The more positive the potential
, the greater the
affinity for the electron of the oxidized compound
(oxidizing ability).

An electron transfer reaction is
spontaneous
(negative ΔG) if E°' is
positive
.
For an
electron
transfer:
'
'
'
'
)
(
'
)
(
'
)
(reductant
'
)
(
o
o
o
E
nF
G
E
E
E
E
E
o
donor
o
acceptor
o
o
oxidant
ΔE, unlike ΔG, is not a state function and
depends on the path of the reaction.
For a series of sequential redox reactions
A B C D
G
AD
= G
AB
+ G
BC
+
G
CD
The free energy is additive
E
AD
≠ E
AB
+ E
BC
+
E
CD
Possibility for
biological
modifications !!!

The location of the generation
site of ATP is the same as the
site
which
provided
the
energy for its generation.
Substrate level
phosphorylation
nATP
nP
nADP
i
O
H
e
H
O
2
2
2
4
4
The process in which ATP is formed as a result
of the transfer of electrons from NADH and
FADH
2
to O
2
by a series of electron carriers.
Oxidative Phosphorylation
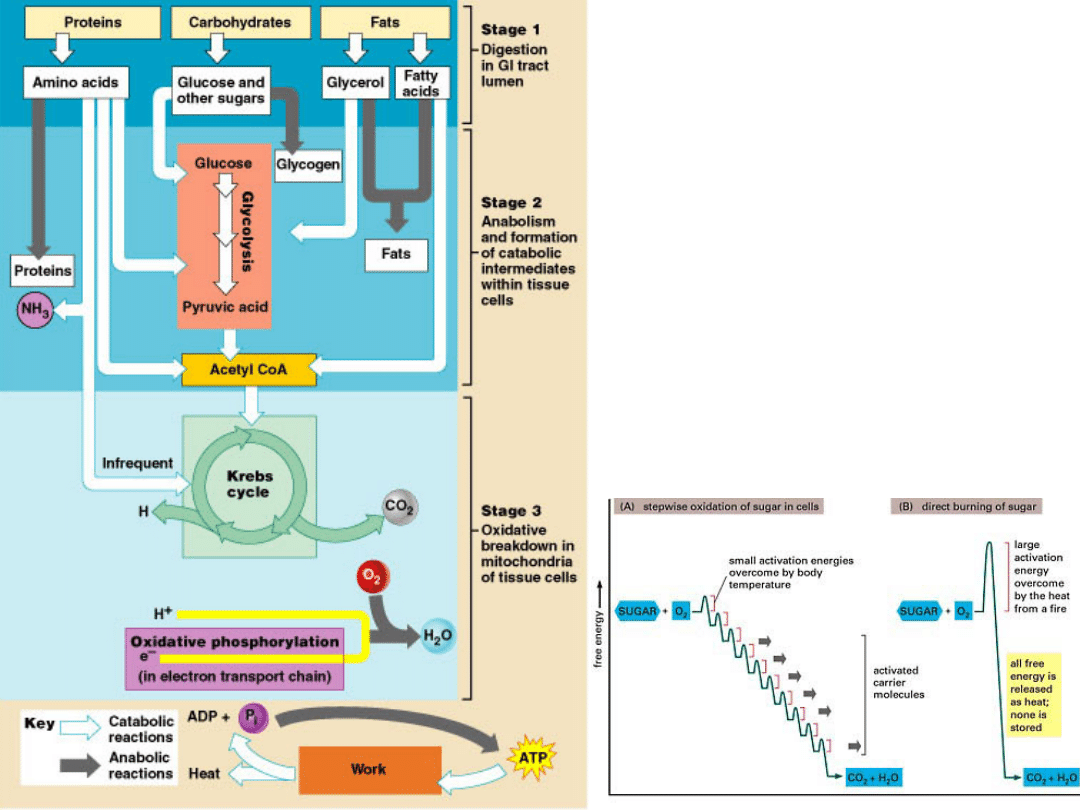
Stages of
metabolism
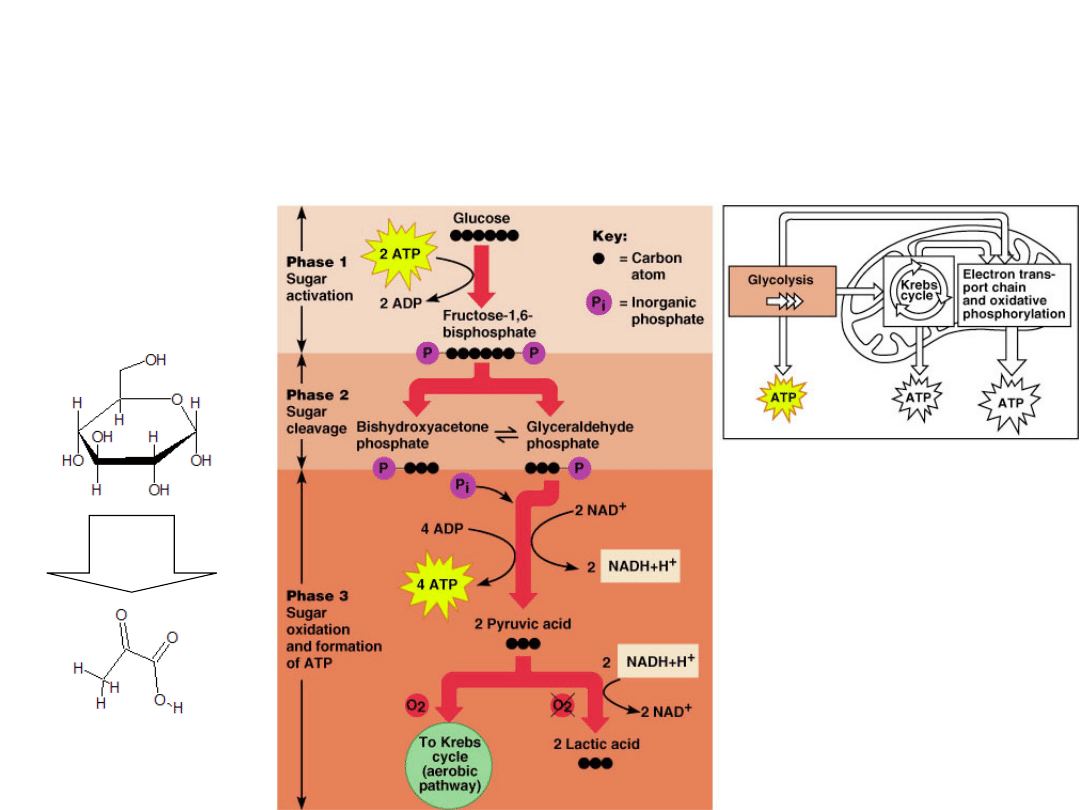
Glycolysis -
found in all living organisms.
It occurs in the cytoplasm outside the
mitochondria.
When rapid production of ATP is needed.
The outer membrane
contains porins, voltage-
dependent anion channels which regulate metabolite
flux, ie., phosphate, Cl
-
, adenine nucleotides and
organic anions.
There are no pH and potential
gradients across the
mitochondrial outer membrane.
porin proteins large pores
(5,000 -10,000 D)
permeable to ions and
molecules < 1 kDa

The inner membrane
is impermeable to ions and
polar molecules. Specific transporters shuttle
metabolites such as ATP, pyruvate, and citrate.
It serves two important functions in energy
generation.
capacitor
- it allows charge separation to
build up between the cytoplasm and the outside
of the cell
structural
- the membrane holds many of the
components involved in electron transport in the
an exact confirmation necessary to enable them
to perform their duties correctly
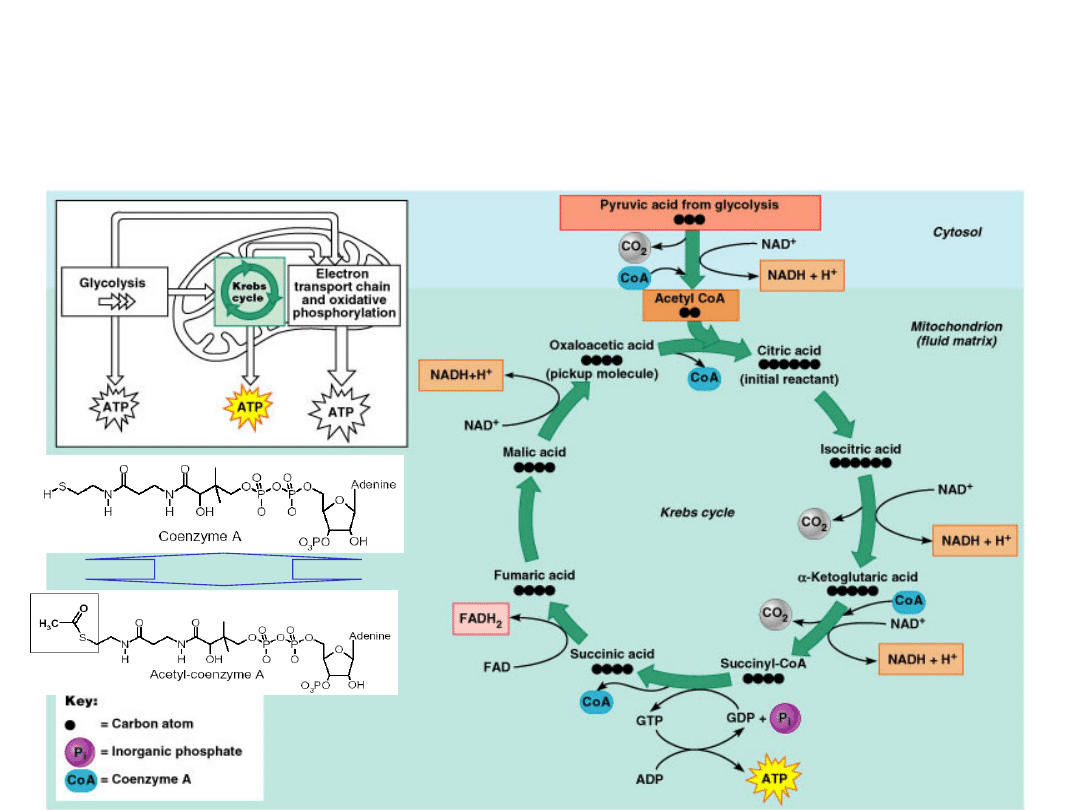
Krebs Cycle
Pyruvate
is carried into the mitochondria where it
is converted into Acetyl Co-A which enters the
Kreb's cycle.
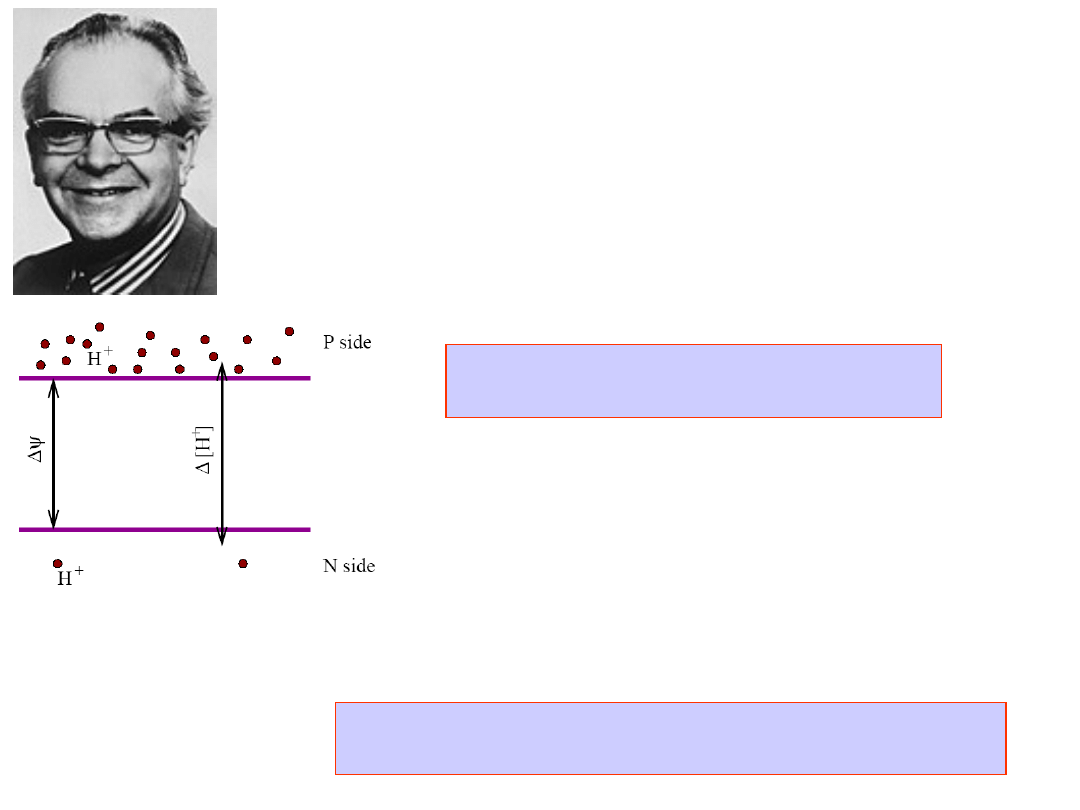
Peter Mitchell
Nobel Prize in Chemistry,
1978
Chemiosmotic Hypothesis
n
p
i
nH
O
H
ATP
nH
P
ADP
2
The ATPase activity and the transmembrane
H
+
flux are reversible:
One of the great
unifying principles
of 20th century
biology
pH
RT
F
H
3
.
2
~
Electron transport (oxidation) and ATP
synthesis (phosphorylation) are
coupled by a proton gradient across
the inner mitochondrial membrane.
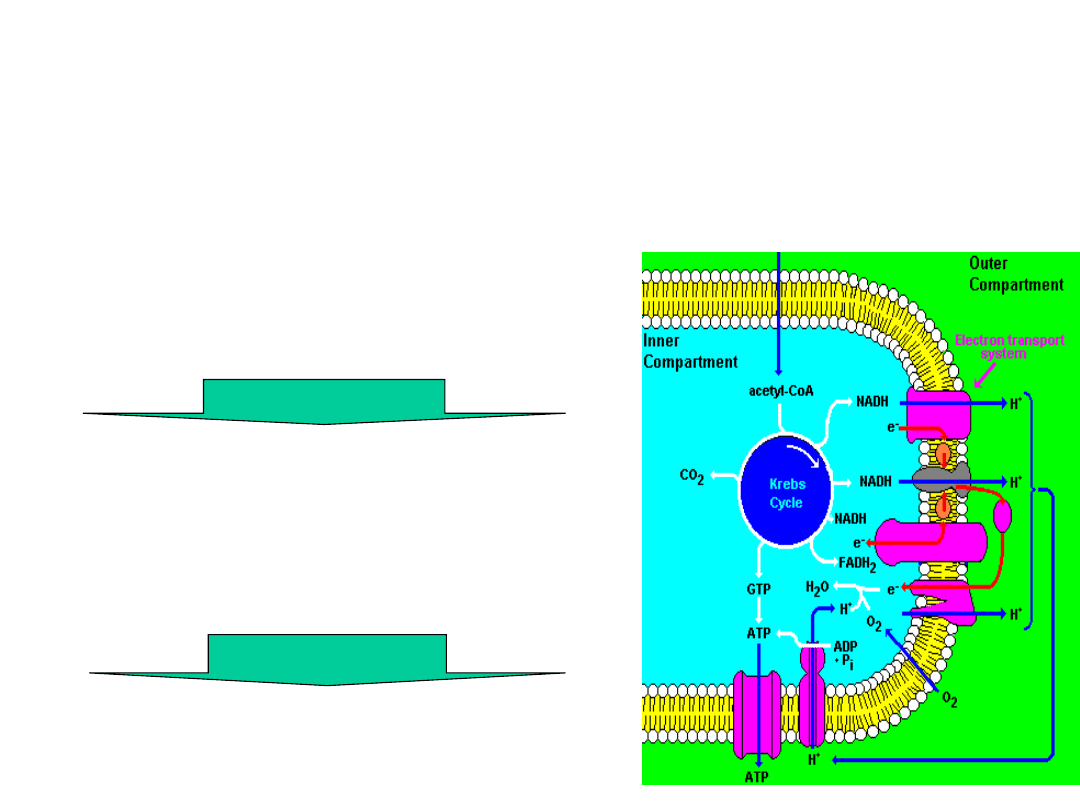
Energy Transformations
Electron–motive force
(NADH & FADH
2
)
Proton-motive force (a
proton gradient (ΔpH = 1.0
units; 8.0 matrix vs. 7.0
peri-mito]) and a membrane
potential (140mV; in(-)
out(+)).
Phosphoryl-transfer potential
in the form of ATP
The chemiosmotic process
the movement of substances across
a membrane is coupled to chemical
reactions.
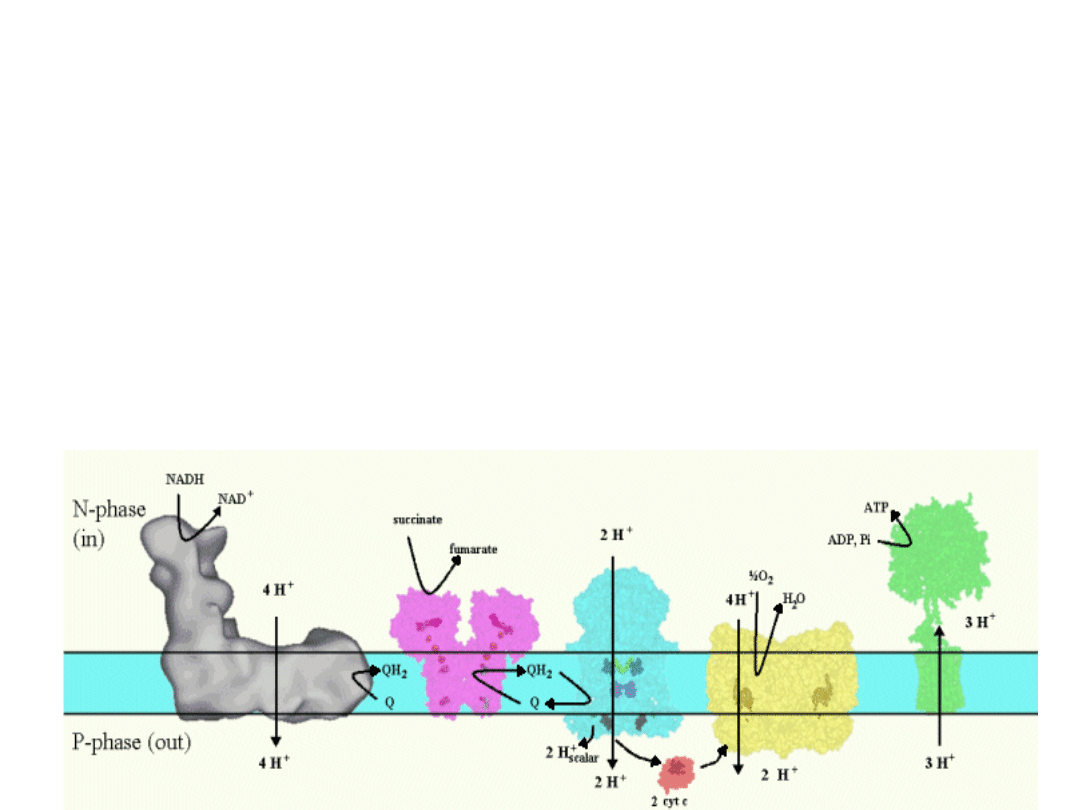
Mitochondrial respiratory chain
Complex I
- Transfers e- from NADH to quinone pool &
pumps H
+
.
Complex II
- Transfers e
-
from succinate to
quinone pool.
Complex III
- Transfers e
-
from quinol to cyt. c &
pumps H
+
.
Complex IV
- Accepts e
-
from cyt. c, reduces O
2
to H
2
O
& pumps H
+
.
Complex V
- Harvests H
+
gradient &
regenerates ATP.
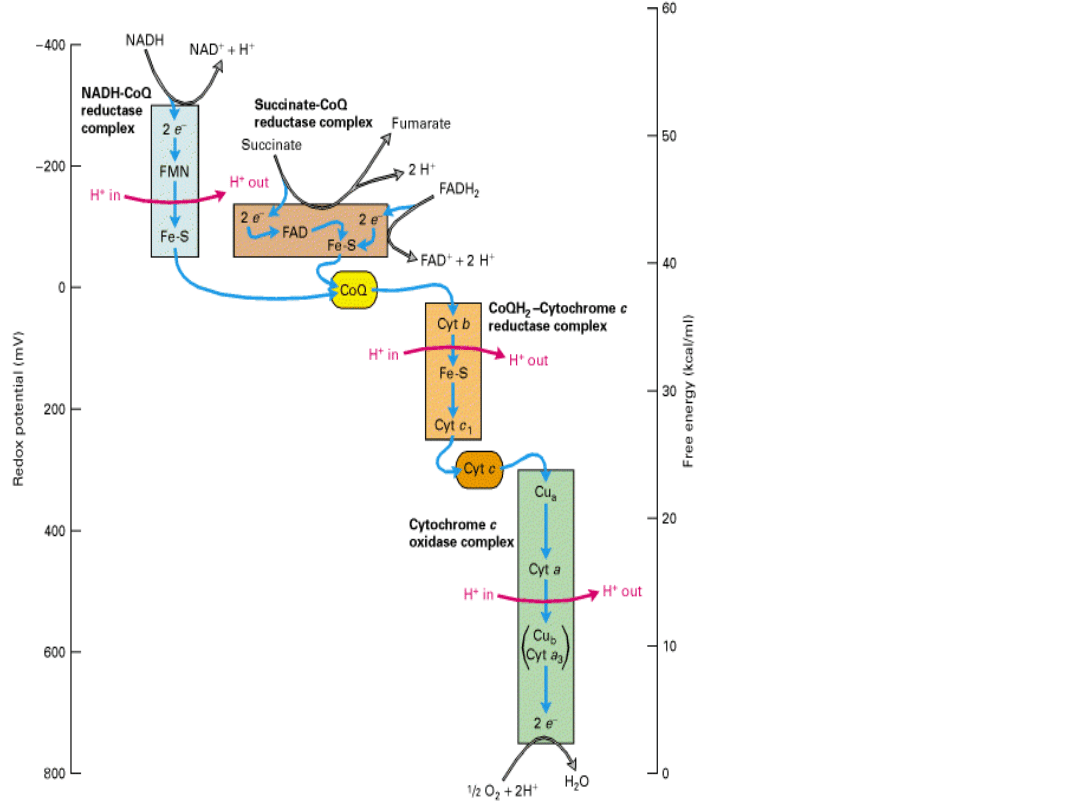
Electronic
energy
gradient
in
mitochon
dria
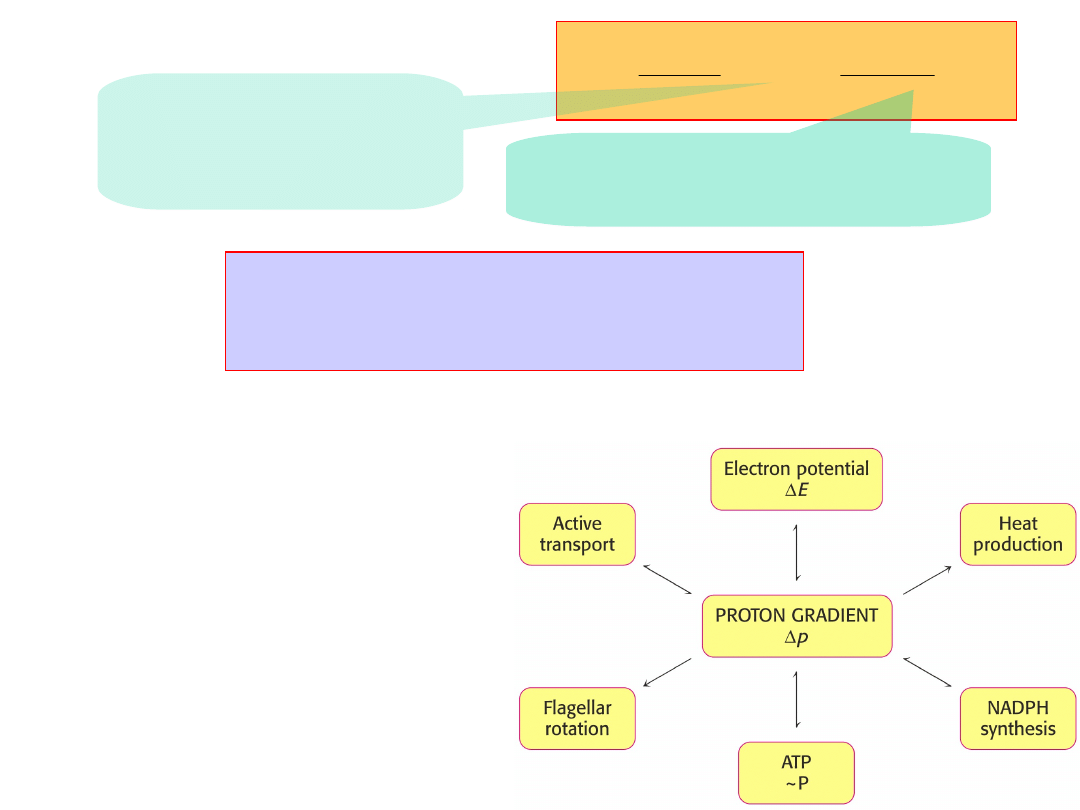
pH
F
RT
F
p
H
3
.
2
~
“Proton-motive
force”,
p.
H+
= 0.224 V = ~22
kJ/mol
CHEMICAL
- H
+
concentration gradient
pH (0.5) – 37%
ELECTRICAL
-
membrane
potential
(
150mV
) – 63%
The voltage gradient experienced by each of
proton pumps is about 30x10
6
volts/m.
The mitochondrial
membranes is rich
in an acidic
phospholipid -
cardiolipin which
makes it
impermeable to
proton leaks.
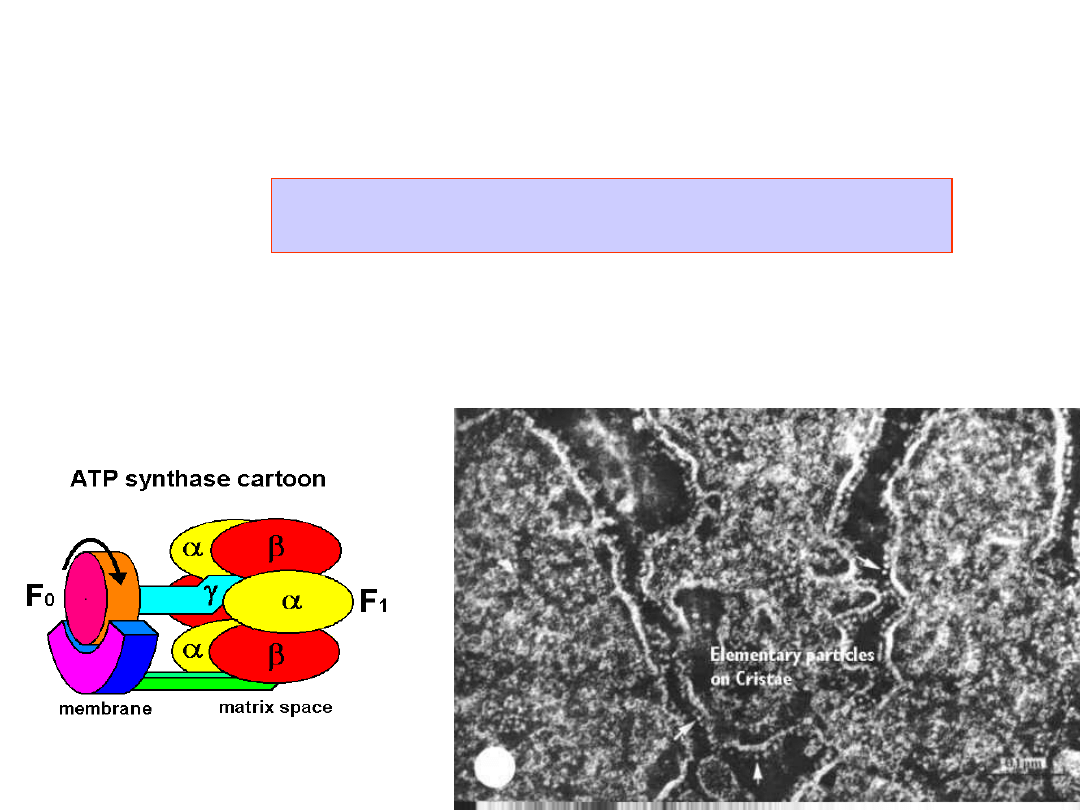
ATP synthesis
ATP Synthase
- makes 100 ATP per 300
H
+
per sec
Synthesis of ATP from ADP and orthophosphate is
coupled to a proton flux.
n
p
i
nH
O
H
ATP
nH
P
ADP
2
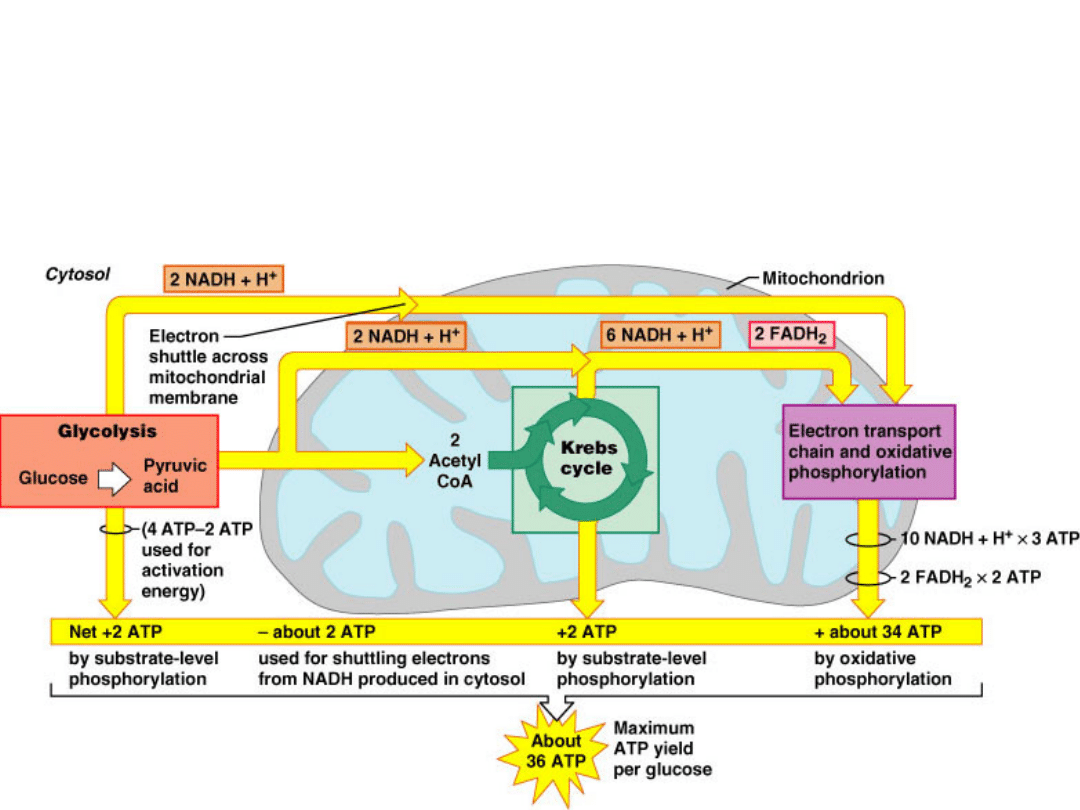
C
6
H
12
O
6
+ 6O
2
+ 36 ADPs + 36 P
i
6CO
2
+ 6H
2
O +
36 ATPs
36 ATPs can be generated from one
molecule of glucose
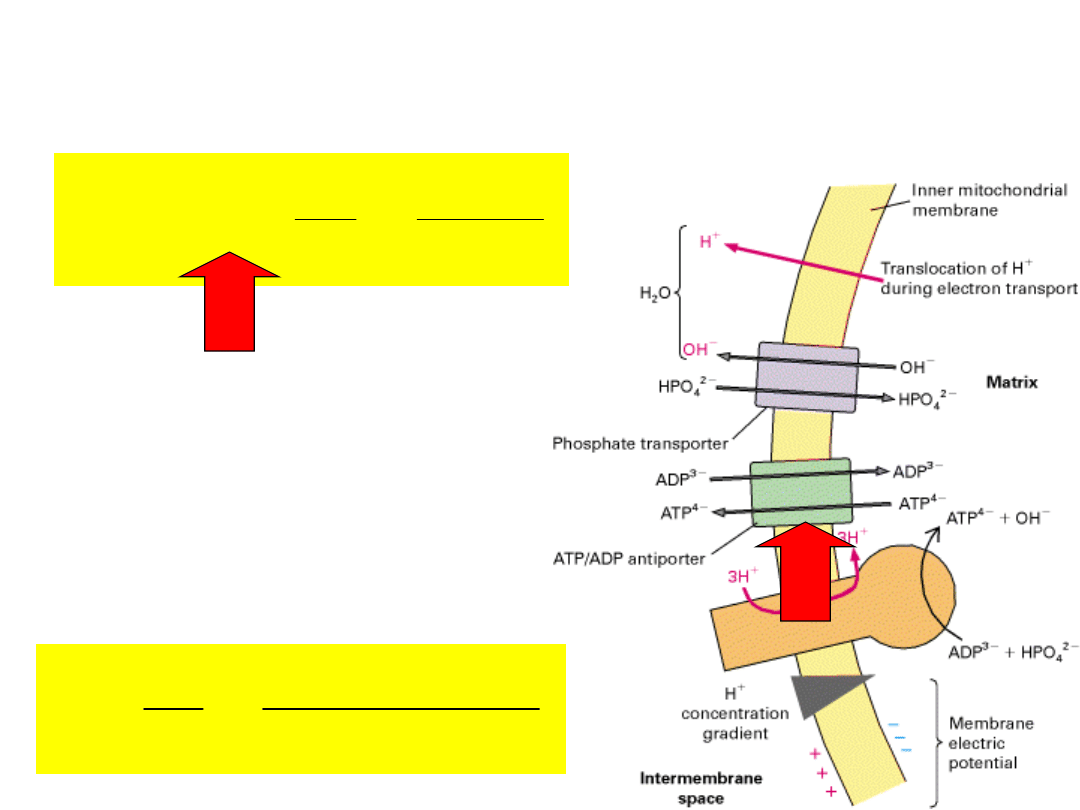
out
in
H
H
H
F
RT
]
[
]
[
ln
out
in
in
out
ADP
ATP
ADP
ATP
F
RT
]
[
]
[
]
[
]
[
ln
The membrane potential
will
drive
ATP/ADP
exchange
in
the
direction of ATP efflux
and
ADP
influx
–
electrically dissipative.
Adenine nucleotide translocase
catalyzes
1:1 exchange of ADP for ATP.
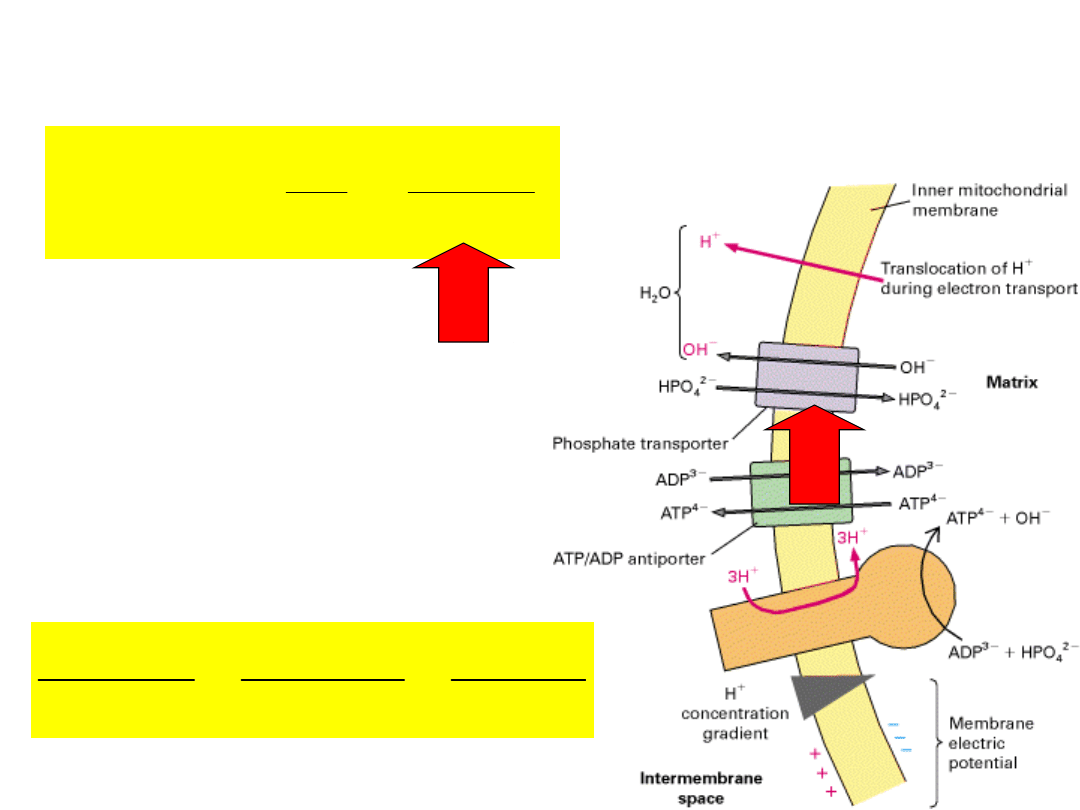
out
in
H
H
H
F
RT
]
[
]
[
ln
PO
PO
4
4
–
–
–
–
enters mitochondria via PO
enters mitochondria via PO
4
4
–
–
/OH
/OH
–
–
exchange (electroneutral).
exchange (electroneutral).
in
out
out
in
out
in
H
H
OH
OH
PO
PO
]
[
]
[
]
[
]
[
]
[
]
[
4
4
PO
4
–
will accumulate in
mitochondria because of
the higher internal OH
–
concentration.

Biological question
How does a molecular motor convert
chemical energy, a scalar quantity, into
directed motion, a vector?
Physical idea
Mechanochemical coupling arises from
a free energy landscape with a direction
set by the geometry of the motor and its
track. The motor executes a biased
random walk on this landscape.
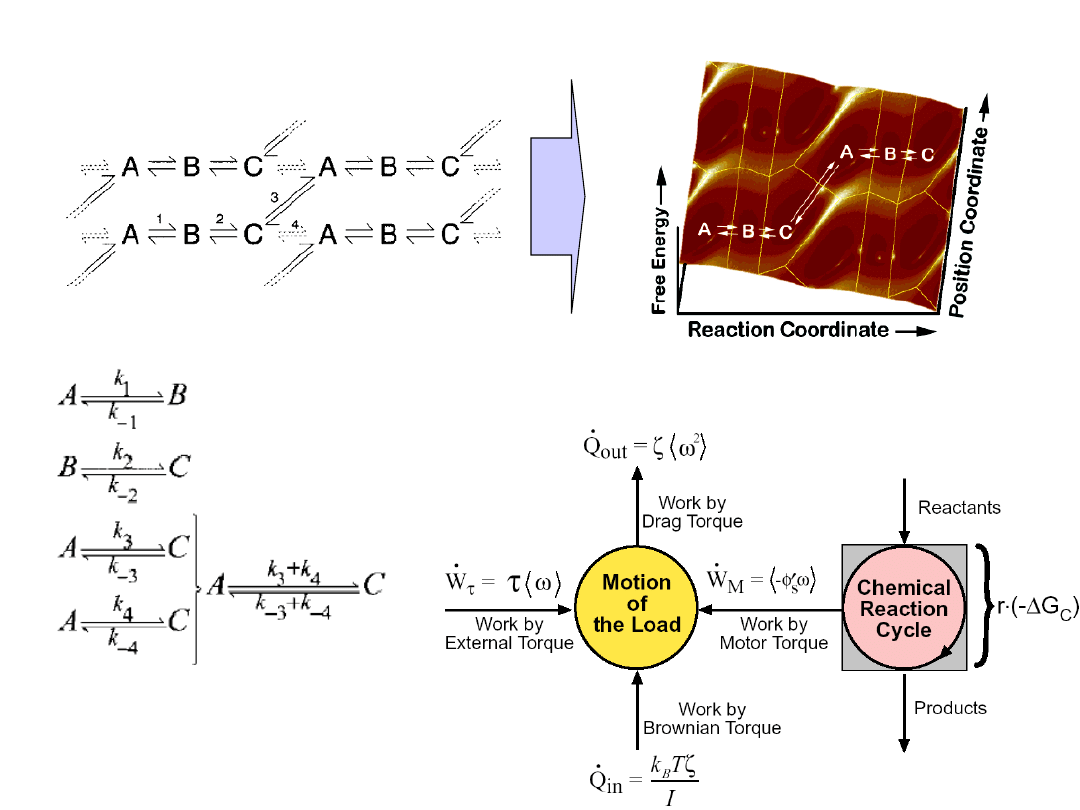
„Active” transport system
The motor, the transport complex and
the link between the two.
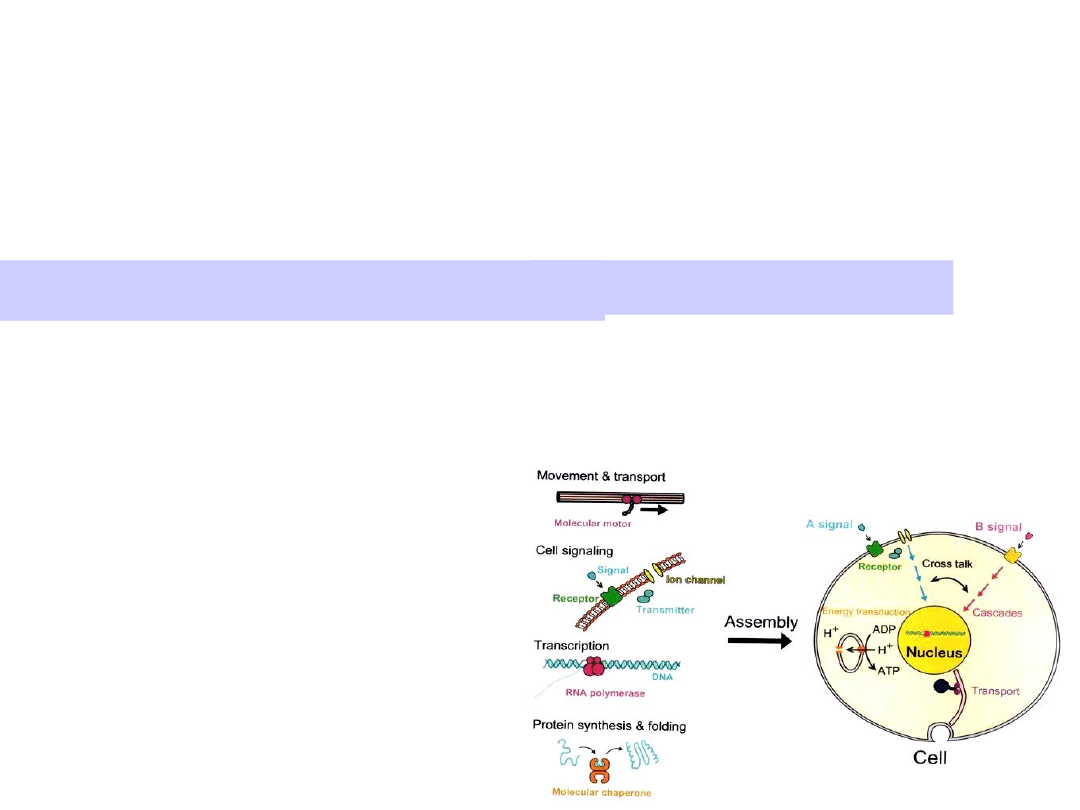
Molecular motors transduce chemical
energy into mechanical motion.
Motors are driven by ATP
ATP hydrolysis ~ 25k
B
T = 1×10
-
19
J.
Force ~ 1 to 100 pN
Thermal energy k
B
T ~ 4.1 × 10
-21
J
Thermal force ~ 1 pN
Molecular machines are encoded by a
genetic material and have the potential
to evolve.
The relation
between
molecular device
and other
devices
determines its
mode of action.
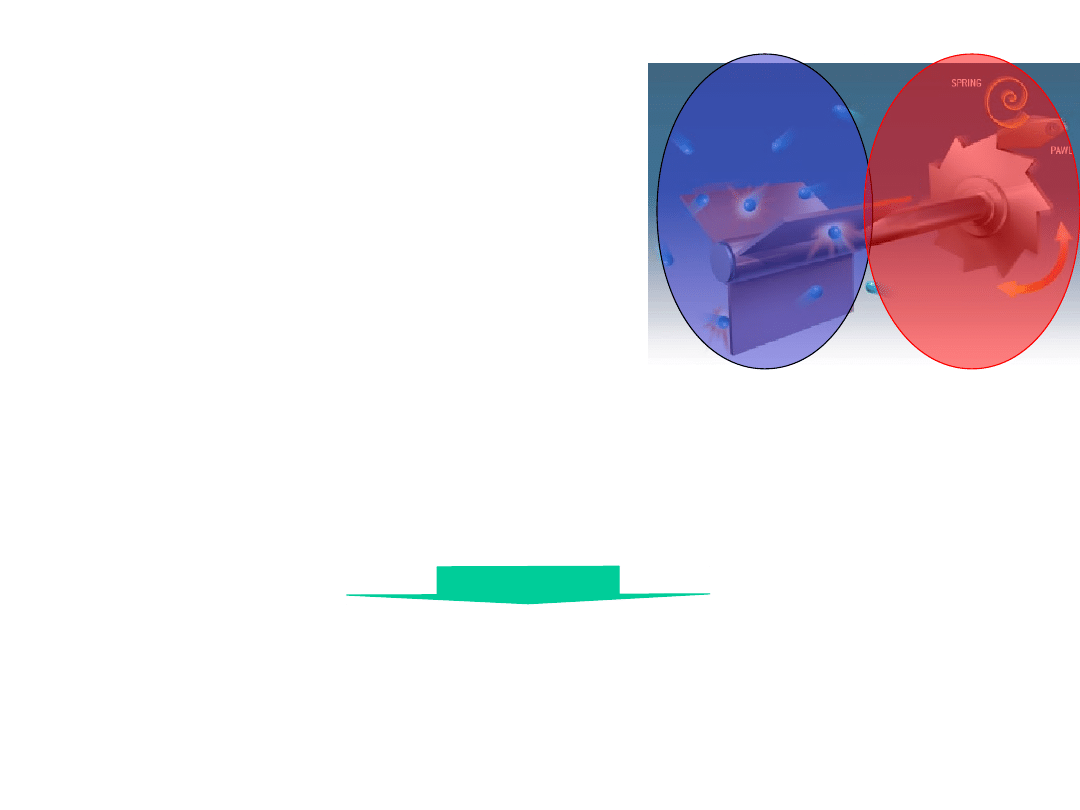
The Feynman Thermal
Ratchet
T
1
T
2
Works only if
T
1
>T
2
!!
The decay of temperature gradient
over 10 nm disappears within ns.
Wrong
model !!
Motor protein
conformational
change take place
within µs.
Molecular machine is an
isothermal engine
(not heat engines)
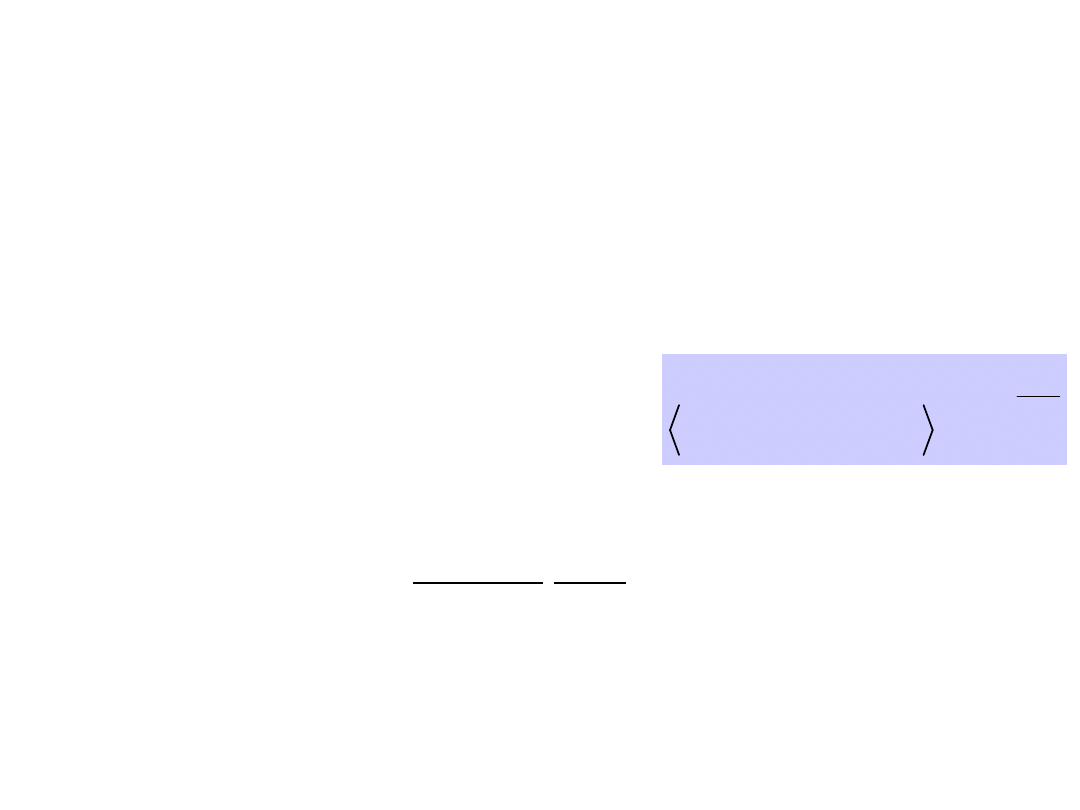
Principles of directed motion
of molecular motors and
pumps
Moving between wells occurs by thermal activation
over barriers.
Machine operates even after
macroscopic equilibrium has been
reached.
Time scales are determined
by depth of energy wells and
height of barriers.
T
k
G
B
e
time
waiting
They operate at energies close to kT and
fluctuations
play a central role.
Molecular motors operate in an environment
where viscosity dominates inertia (velocity if
proportional to force).
Only potential energy is
stored.
Friction dominates inertia.

1.
The chemical cycle is
autonomous.
2.
The binding energy to the track depends on
the state of the chemical cycle.
3.
The motor feels an asymmetric potential
energy as a function of position.
The structure of a molecular machine
A track with spatial
asymmetry.
Track
Machine
A site which binds to the track.
Out-of-equilibrium process coupled to a location.
A
catalitic
site
hydrolyzing ATP.
ATP
The allosteric interaction coupluing the
ATPase cycle to the track binding.
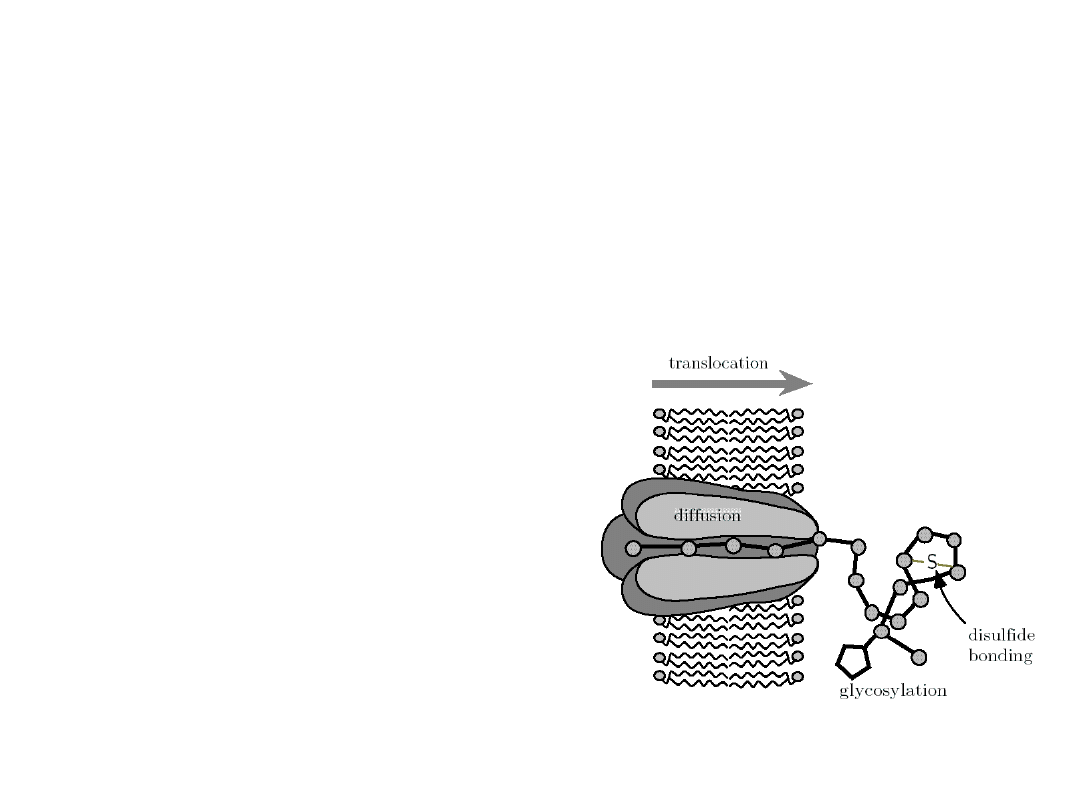
Molecular devices found in cells
Catalists
– enhance the rate of a chemical
reaction (enzymes)
Machines
– actively reverse the natural
flow of some chemical or mechanical
process by coupling it to another one.
Cyclic machines
–
process some external
source of free energy.
One-shot machines
–
exhaust some internal
source of free energy
Protein
translocat
ion
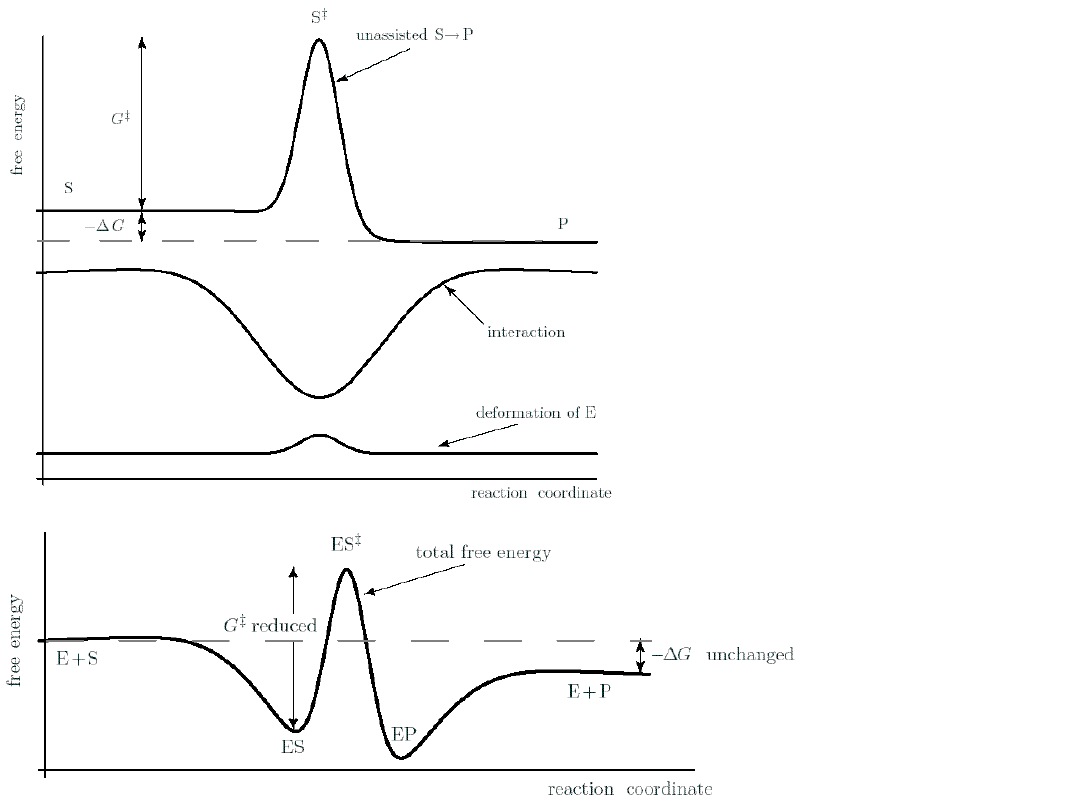
Enzymes work
by reduces the
activation
energy of a
reaction.
An enzyme
cannot alter the
net
G of the
reaction.
An enzyme
binds to the
transition
state.
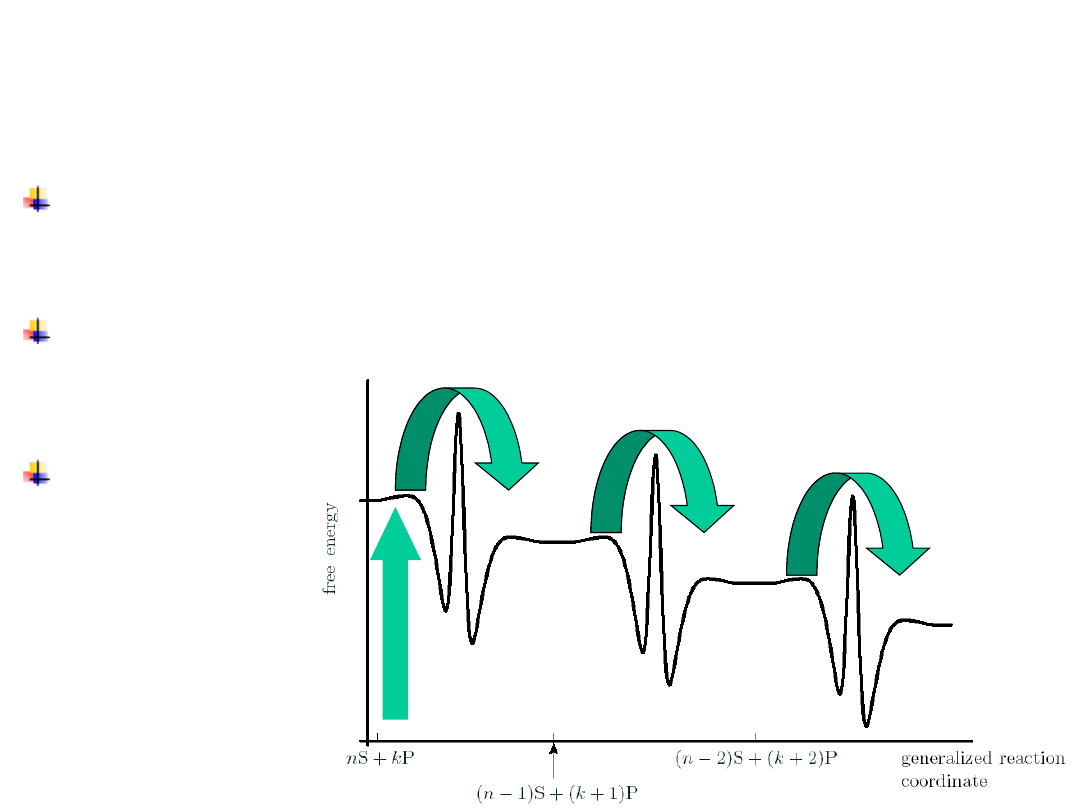
A large c
P
can
reverse
the sign of G
reversing the
reaction.
Enzymes can be regarded as
a cyclic machines.
High c
S
rises the left end of the free
energy landscape.
It operates in cycles, but the system’s free energy
falls by G in each step.
The potential energy surface
is periodic.
Energy balance on a molecular
motor
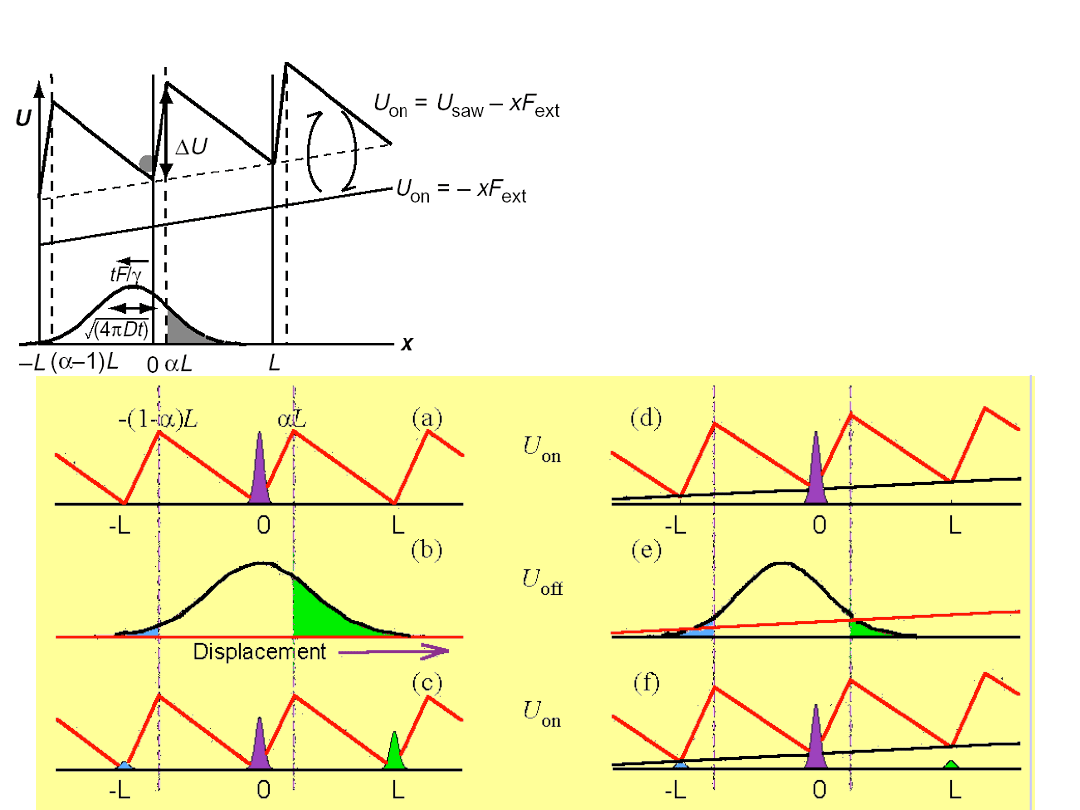
The mechanism of the flashing
Brownian ratchet
The ratchet potential
exhibits
broken
spatial symmetry
V(x + L) = V(x)
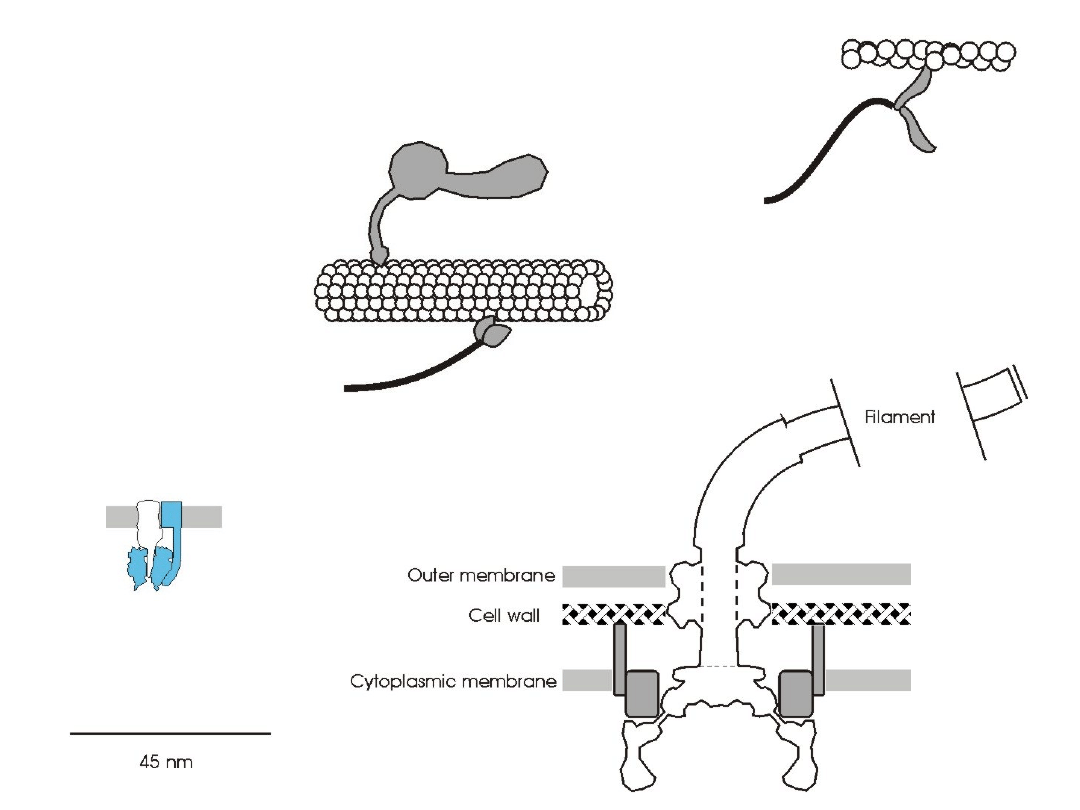
Molecular motors
F
0
F
1
-ATPase
Myosin
Flagella motor
Dynein
Kinesin
Document Outline
- Slide 1
- Slide 2
- Slide 3
- Slide 4
- Slide 5
- Slide 6
- Slide 7
- Slide 8
- Slide 9
- Slide 10
- Slide 11
- Slide 12
- Slide 13
- Slide 14
- Slide 15
- Slide 16
- Slide 17
- Slide 18
- Slide 19
- Slide 20
- Slide 21
- Slide 22
- Slide 23
- Slide 24
- Slide 25
- Slide 26
- Slide 27
- Slide 28
- Slide 29
- Slide 30
Wyszukiwarka
Podobne podstrony:
wykład 12 pamięć
Socjologia wyklad 12 Organizacja i zarzadzanie
Wykład 12(3)
Wykład 12
Wykład 12 Zarządzanie sprzedażą
Wykład 12 1
biofizyka wyklad 09
wyklad 12
Wyklad 1 12
wyklad 12 MNE
biofizyka wyklad 04
wykład 12
ZARZ SRODOWISKIEM wyklad 12
wykład 7 12
Wyklad 12 ppt
więcej podobnych podstron