
Journal of Environmental Sciences 2010, 22(5) 777–783
An autotrophic nitrogen removal process: Short-cut nitrification
combined with ANAMMOX for treating diluted e
ffluent
from an UASB reactor fed by landfill leachate
Jie Liu
1
,2
, Jian’e Zuo
1,
∗
, Yang Yang
1
, Shuquan Zhu
2
,
Sulin Kuang
3
, Kaijun Wang
1
1. Environmental Simulation and Pollution Control State Key Joint Laboratory, Department of Environmental Science and Engineering,
Tsinghua University, Beijing 100084, China. E-mail: liuj1101@126.com
2. School of Chemical and Environmental Engineering, China University of Mining and Technology (Beijing), Beijing 100083, China
3. Liulitun Sanitary Landfill, Beijing 100094, China
Received 30 July 2009; revised 27 November 2009; accepted 01 December 2009
Abstract
A combined process consisting of a short-cut nitrification (SN) reactor and an anaerobic ammonium oxidation upflow anaerobic
sludge bed (ANAMMOX) reactor was developed to treat the diluted e
ffluent from an upflow anaerobic sludge bed (UASB) reactor
treating high ammonium municipal landfill leachate. The SN process was performed in an aerated upflow sludge bed (AUSB) reactor
(working volume 3.05 L), treating about 50% of the diluted raw wastewater. The ammonium removal e
fficiency and the ratio of NO
2
−
-
N to NOx
−
-N in the e
ffluent were both higher than 80%, at a maximum nitrogen loading rate of 1.47 kg/(m
3
·day). The ANAMMOX
process was performed in an UASB reactor (working volume 8.5 L), using the mix of SN reactor e
ffluent and diluted raw wastewater at a
ratio of 1:1. The ammonium and nitrite removal e
fficiency reached over 93% and 95%, respectively, after 70-day continuous operation,
at a maximum total nitrogen loading rate of 0.91 kg
/(m
3
·day), suggesting a successful operation of the combined process. The average
nitrogen loading rate of the combined system was 0.56 kg
/(m
3
·day), with an average total inorganic nitrogen removal efficiency 87%.
The nitrogen in the e
ffluent was mostly nitrate. The results provided important evidence for the possibility of applying SN-ANAMMOX
after UASB reactor to treat municipal landfill leachate.
Key words: landfill leachate; short-cut nitrification; anaerobic ammonia oxidation; autotrophic nitrogen removal
DOI: 10.1016
/S1001-0742(09)60176-5
Introduction
Municipal landfill leachate represents a special case of
high-strength wastewater. It is a complex mix of many
kinds of organic and inorganic contaminants generated
through the decomposition of municipal solid wastes
during landfill and is further supplemented by rainwater
percolating through the waste material (Ganigue et al.,
2007). This leachate has extremely high environmental
pollution potential, due to its high concentrations of or-
ganics, ammonium, and inorganic salts, including, in some
cases, heavy metals (Horan et al., 1997).
Compared with organic contaminants in landfill
leachate, the high concentration ammonium is more dif-
ficult to be treated. Nitrogen removal from wastewaters
is traditionally performed by a conventional biological
nitrification
/denitrification process (Munch et al., 1996).
However, complete nitrogen removal can be limited by
the availability of biodegradable organic carbon sources
(Puig et al., 2004). The ratio of C
/N in municipal landfill
* Corresponding author. E-mail: jiane.zuo@tsinghua.edu.cn
leachate is quite low, which makes the traditional nitrifica-
tion
/denitrification process inapplicable or too expensive
(Cema et al., 2006; Fux et al., 2004; Strous et al., 1997).
A complete autotrophic nitrogen removal process, the
anaerobic ammonium oxidation (ANAMMOX) process
has been newly developed, in which the ammonium is
oxidized directly with nitrite, without the requirement of
any organic carbon matters, thereby this process is con-
sidered an one of the most promising biological treatment
process for nitrogen removal, which may provide a novel
alternative solution to the nitrogen removal from leachate.
The ANAMMOX process was first observed in an
autotrophic denitrification fluidized bed reactor fed by the
e
ffluent of an anaerobic reactor containing some ammoni-
um (Mulder et al., 1995). It is actually an anoxic process,
in which the ammonium is oxidized by nitrite instead
of oxygen in the traditional nitrification process as the
electron acceptor, and the nitrite is reduced into nitrogen
gas by ammonia instead of organic carbon matter in the
traditional denitrification process as the electron donner
(van de Graaf et al., 1996). ANAMMOX is an autotrophic
778
Jie Liu et al.
Vol. 22
nitrogen removal process performed by the ANAMMOX
bacteria, which are characterized by an extremely slow
growth rate (doubling time of 11 days) (Strous et al., 1998).
This makes the start-up period longer compared to other
nitrogen removal technologies.
Prior to ANAMMOX process, a short-cut nitrification
(SN)-converting ammonium to nitrite-process should be
performed to supply enough nitrite for the ANAMMOX
process. In recent years, increasing attention has been
paid to the application of this process for wastewater
treatment, such as the supernatant from sludge and swine
wastewater digester, municipal landfill leachate. The first
full-scale ANAMMOX reactor was build up in Rotterdam,
the Netherlands, which treats up to 750 kg-N
/day (load of
10.71 kg
/(m
3
·day)) (Wouter et al., 2007). A process con-
sisting of a partial nitrification reactor and an ANAMMOX
reactor, followed by two ground soil infiltration systems,
was applied to treat municipal landfill leachate (Liang
and Liu, 2008), and a quite stable operation was obtained
when the ANAMMOX load was below 0.12 kg
/(m
3
·day).
The long-term stability of partially nitrification of swine
wastewater digester liquor and the subsequent treatment by
ANAMMOX process were also studied, and very stable
nitrogen removal e
fficiency was obtained in 70 days at
a nitrogen removal loading rate of 0.22 kg N
/(m
3
·day)
(Yamamoto et al., 2008).
In Liulitun Sanitary Landfill, Beijing, China, the munic-
ipal landfill leachate is currently treated by a full scale
upflow anaerobic sludge bed (UASB) reactor, but the
e
ffluent from the UASB reactor still contains high concen-
trations of ammonium and relatively low concentrations
of organic carbon matter. Thus, due to lack of carbon
sources, the traditional nitrification
/denitrification process
is not suitable to be applied for nitrogen removal.
In the present article, a novel complete autotrophic nitro-
gen removal process, consisting of SN and ANAMMOX,
has been developed for treating the diluted e
ffluent from
the UASB reactor fed by municipal landfill leachate. The
ammonium in about 50% of diluted raw wastewater is
completed oxidized to nitrite, then mixed with the other
50% diluted raw wastewater, and the mix is supplied to the
ANAMMOX process, thus ammonium and nitrite are re-
moved simultaneously. In this way, ANAMMOX process,
combined with SN process, a new SN-ANAMMOX pro-
cess provides a completely autotrophic nitrogen removal
technology. Compared with the conventional biological
treatment technologies, this process may have promising
technical and economic advantages for the treatment of
landfill leachate, due to less oxygen consumption, no
requirement for organic carbon matter addition, and low
sludge production (Schmidt et al., 2003; Khin and Annach-
hatre, 2004).
1 Materials and methods
1.1 Raw wastewater
The raw wastewater studied was obtained from the
e
ffluent of an UASB reactor in Liulitun municipal solid
waste (MSW) sanitation landfill plant in Beijing, Chi-
na. The NH
4
+
-N concentration was high and C
/N ratio
(COD
cr
/NH
4
+
-N) was low in the raw wastewater (Table
1). Due to the high concentration of ammonium, the raw
wastewater was diluted with tap water to an NH
4
+
-N
concentration of about 600–900 mg
/L prior entering into
the system.
1.2 Experimental set-up
This process consisted of two major components: a
SN reactor and an ANAMMOX reactor. In the combined
sequential process, the diluted raw wastewater was first
fed to the SN reactor for production of nitrite. The e
ffluent
from this reactor was then mixed with an approximately
equal quantity of the diluted raw wastewater, and this was
then fed into the ANAMMOX reactor for total nitrogen re-
moval. The experimental flow chart and schematic diagram
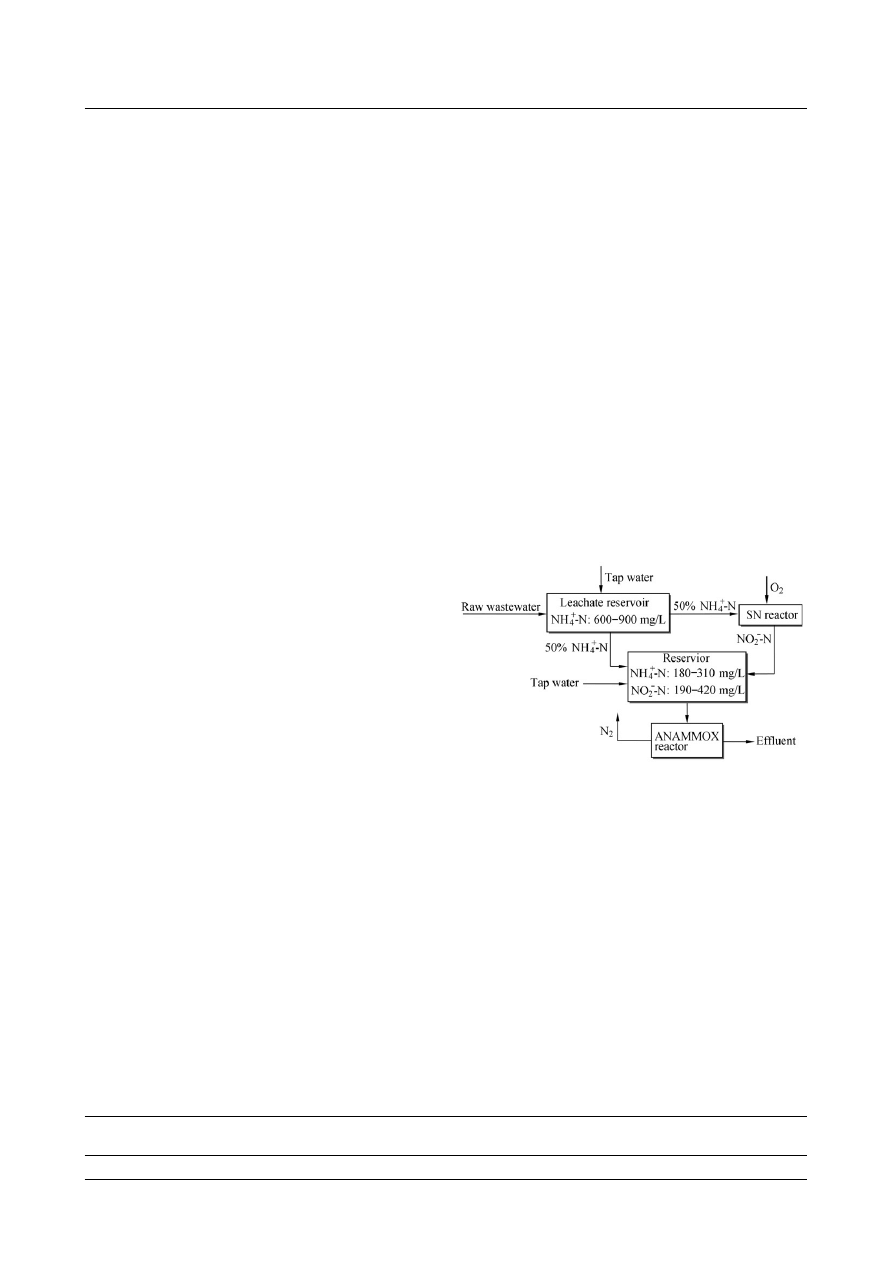
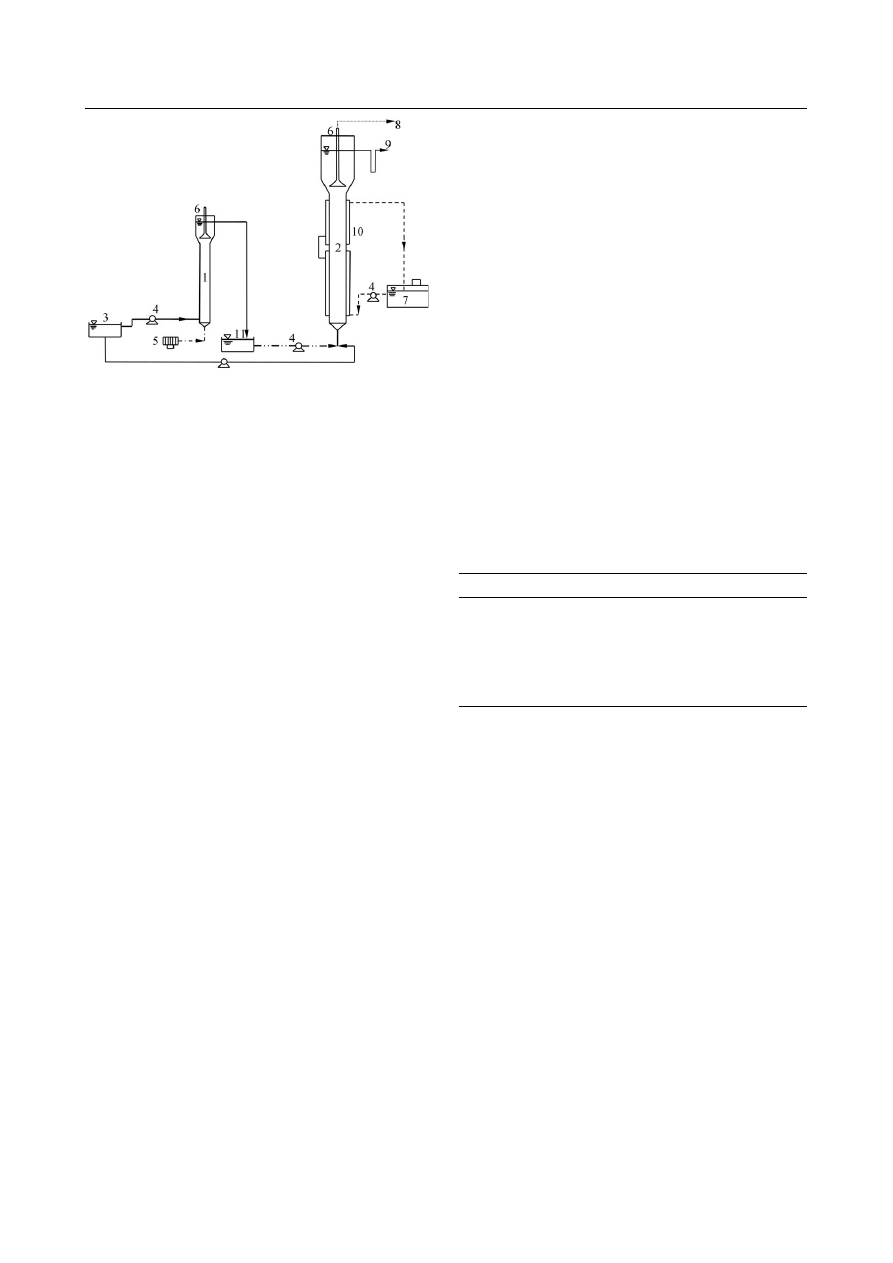
are shown in Figs. 1 and 2, respectively.
Fig. 1
Flow chart of the SN-ANAMMOX process.
1.2.1 Short-cut nitrification system and operational
scheme
The SN reactor, an aerated upflow sludge bed (AUSB)
reactor, was made of polymethyl methacrylate, with a
total volume of 3.05 L and a reaction zone of 1.45 L.
The aerobic activated sludge from a nitrification reac-
tor in the same laboratory was used as the inoculum
(initial concentration was 2.3 g MLVSS
/L). During the
period of the previous 85 days operation, the reactor
was operated with the inorganic synthetic wastewater,
which was prepared by adding ammonium (NH
4
HCO
3
),
KH
2
PO
4
, NaHCO
3
, MgSO
4
·7H
2
O, CaCl
2
and trace el-
ements ((g
/L) Na
2
EDTA
·2H
2
O 15, ZnSO
4
·7H
2
O 0.43,
CoCl
2
·6H
2
O 0.24, MnCl
2
·4H
2
O 0.99, CuSO
4
·5H
2
O 0.25,
Table 1
Raw wastewater characteristics (e
ffluent from UASB reactor fed by landfill leachate)
COD
cr
BOD
5
NH
4
+
-N
Alkalinity
PO
4
3
−
-P
TOC
pH
(mg
/L)
(mg
/L)
(mg
/L)
(CaCO
3
) (mg
/L)
(mg
/L)
(mg
/L)
1900–2200
160–200
1900–2400
11000–13000
3–5
215–574
8.0–8.4
No. 5
An autotrophic nitrogen removal process: Short-cut nitrification combined with ANAMMOX for treating diluted e
ffluent······
779
Fig. 2 Schematic diagram of SN-ANAMMOX process. (1) SN reactor;
(2) ANAMMOX reactor; (3) influent; (4) peristaltic pump; (5) air pump;
(6) gas separator and collector; (7) hot water bath; (8) nitrogen gas; (9)
e
ffluent; (10) hot water jacket; (11) mix tank.
NaMoO
4
·2H
2
O 0.22, NiCl
2
·6H
2
O 0.19, Na
2
SeO
3
0.08,
and H
3
BO
3
0.02) to tap water under the operating condi-
tions of temperature 25°C, pH 7.8–8.0, dissolved oxygen
(DO) 1.5–3mg
/L, and hydraulic retention time (HRT) 18
hr. At the end of the operation, the ammonium removal
e
fficiency and ratio of NO
2
−
-N to NOx
−
-N in the e
ffluent
were both more than 90% at a loading rate of 0.82 kg
N
/(m
3
·day). From day 86, the SN reactor was fed with
the diluted raw wastewater, HRT was about 14 hr and the
nitrogen loading rate was about 1 kg TN
/(m
3
·day), at the
beginning, the pH and DO concentration were controlled
at 7.4 and 3.5 mg
/L respectively, result in the decrease of
ratio of the NO
2
−
-N to NOx
−
-N in e
ffluent. Then the pH
and DO were adjusted to 8 and 1.5 mg
/L, respectively,
and the SN process was achieved soon afterwards. The
required ammonium nitrogen-loading rate was acquired by
adjusting the influent flow and influent ammonium con-
centration. The reactor was performed at room temperature
(about 25°C). The pH value and alkalinity were regulated
by the addition of sodium bicarbonate to the influent.
1.2.2 ANAMMOX system and operational scheme
The ANAMMOX reactor was an UASB reactor made
of polymethyl methacrylate with a total volume of 8.45
L, and a reaction zone of 6.05 L. Due to the long dou-
bling time of the ANAMMOX bacteria, BMTM biofilm
carriers (
ϕ12×10 mm for each cube of carrier, Dalian
Shengyuan Water Treatment Equipment Development
Co., Ltd., China) were used to immobilize the anaero-
bic microorganisms. Prior to the operation with mixture
of the diluted raw wastewater and the e
ffluent of the
SN reactor, the reactor was operated with the synthetic
wastewater, which was prepared by adding NH
4
HCO
3
,
NaNO
2
, KH
2
PO
4
, NaHCO
3
, MgSO
4
·7H
2
O, CaCl
2
, trace
elements I ((g
/L) EDTA 5, FeSO
4
5) and trace elements
II ((g
/L): EDTA 15, H
3
BO
4
0.014, MnCl
2
·4H
2
O 0.99,
CuSO
4
·5H
2
O 0.25, ZnSO
4
·7H
2
O 0.43, NiCl
2
·6H
2
O 0.19,
NaSeO
4
·10H
2
O 0.21, NaMoO4
·2H
2
O 0.22) to a mineral
medium for 260 days, under the operating conditions of
temperature 31°C, pH 7.6, HRT 17 hr. At the end of the
experiment, the ANAMMOX reactor achieved a successful
start-up and stable running, with ammonium and nitrite
removal e
fficiencies of over 95% and the total nitrogen
loading rate of 0.59 kg N
/(m
3
·day). From day 261, the
ANAMMOX reactor was operated with the mixture of
the raw wastewater and the e
ffluent of the SN reactor,
at the beginning of the operation, the NH
4
+
-N removal
e
fficiency reduced to about 64%, while the NO
2
−
-N re-
moval e
fficiency still kept above 95%, which indicate that
the ammonium was overdosed. Afterwards, the ratio of
raw wastewater to the e
ffluent of SN reactor was strictly
controlled, then the removal e
fficiency of NH
4
+
-N soon
risen to above 93%. The desired loading rate was attained
by adjusting the ammonium and nitrite concentration in
influent. Temperature in the reactor was maintained at (31
± 1)°C by a hot water jacket. The pH value in the reactor
was controlled in the range of 7.5–8.0 by dosing sodium
bicarbonate into the influent. The HRT of the reactor was
maintained at 17 hr.
1.2.3 Analytic methods
The analytic methods and equipments used for nitrogen
(NH
4
+
-N, NO
2
−
-N, NO
3
−
-N), pH, DO, chemical oxygen
demand (COD) and temperature are shown in Table 2.
Table 2
Analytic methods and equipments
Analytic method and equipment
NH
4
+
-N
Nesster’s reagent colorimetric method
NO
2
−
-N
N-(1-Naphthyl)-ethylenediamine spectrophotometry
NO
3
−
-N
UV spectrophotometry
pH
Orion828 acidometer
DO
Thermo 810A dissolved oxygen meter
COD
The standard method of potassium dichromate*
Temperature
Alcohol thermometer (0–50°C)
* Monitoring and Analyzing Methods of Water and Wastewater (4th ed.).
Environmental Protection Agency of China.
2 Results and discussion
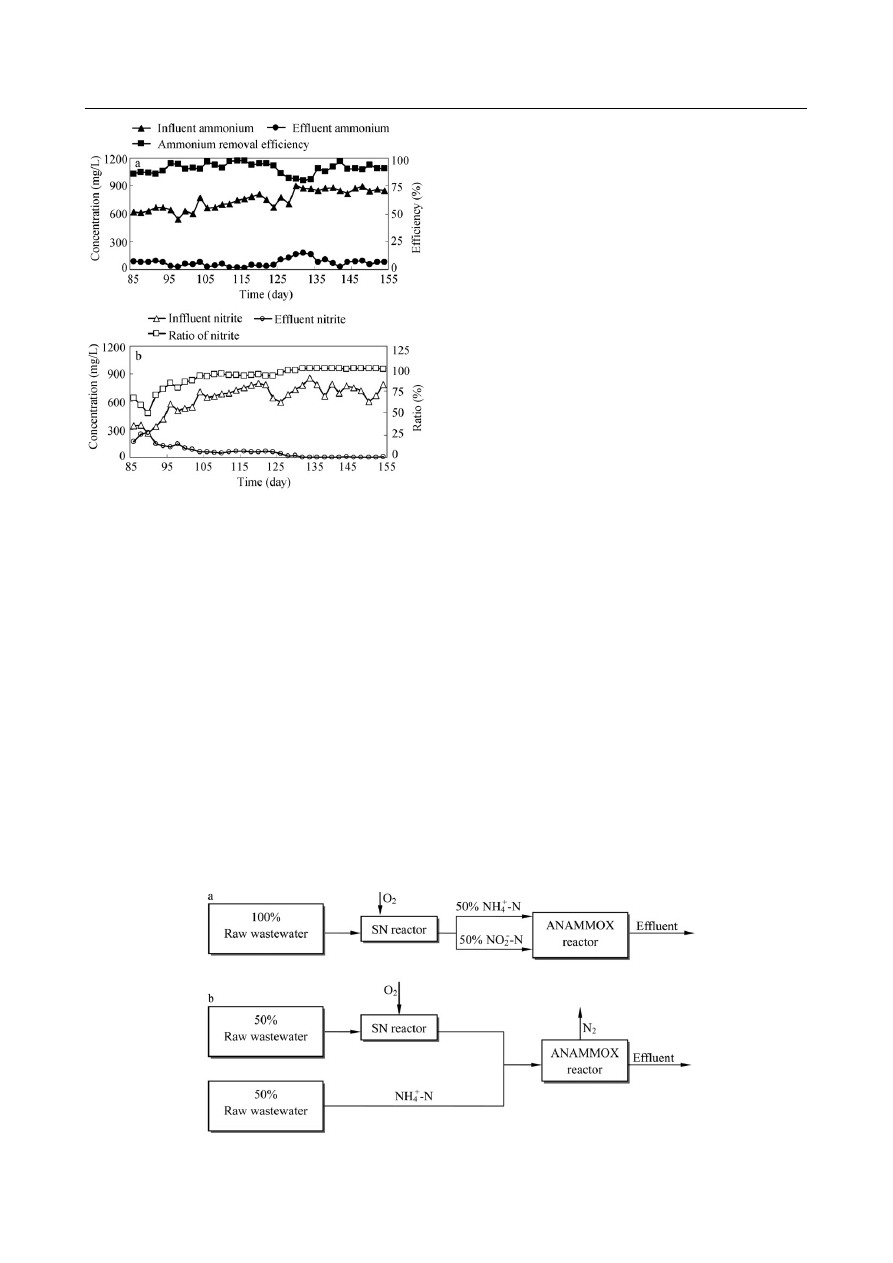
2.1 Short-cut nitrification process
The concentration of NH
4
+
-N, NO
2
−
-N, NO
3
−
-N of
the SN reactor and the corresponding NH
4
+
-N removal
e
fficiency and ratio of NO
2
−
-N to NOx
−
-N in the e
ffluent
are presented in Fig. 3. Since an 85-day operation with the
synthetic wastewater was prior to the combined process,
the results of the SN reactor are presented from day 85.
After the diluted raw wastewater was used as the influent,
the pH value and the DO concentration in the reactor
were maintained at 7.4 and 3.5 mg
/L, respectively, at the
beginning of the operation, while the HRT was 14 hr and
the temperature was 25°C. The NH
4
+
-N removal e
fficiency
was maintained at around 90%, the ratio of NO
2
−
-N to
NOx
−
-N in the e
ffluent fluctuated around 50% between day
85 and day 97. From day 91, the pH value in the reactor
was increased to about 8.0, the DO concentration in the
reactor was adjusted to about 1.5 mg
/L, and the ratio of
NO
2
−
-N to NOx
−
-N in the e
ffluent increased to 86% and
maintained at about 90% after day 102. This suggested that
at certain HRT and temperature conditions, the pH and DO
780
Jie Liu et al.
Vol. 22
Fig. 3
Performance of short-cut nitrification reactor. (a) NH
4
+
-N re-
moval e
fficiency and NH
4
+
-N concentrations in influent and e
ffluent; (b)
NO
2
−
-N concentrations in influent and e
ffluent, and the ratio of NO
2
−
-
N
/NOx
−
-N in e
ffluent.
concentration in the reactor are two important parameters
for obtaining a higher ratio 90% of NO
2
−
-N to NOx
−
-
N in the e
ffluent. An the end of the operation, with an
average nitrogen loading rate of about 1.4 kg
/(m
3
·day), the
NH
4
+
-N removal e
fficiency and the 90% ratio of NO
2
−
-
N to NOx
−
-N in the e
ffluent could be still maintained,
suggesting a successful operation of the SN reactor fed
by the diluted raw wastewater at room temperature (about
25°C) conditions.
As the electron acceptor of the ANAMMOX process, the
NO
2
−
-N concentration in influent is one of key parameters
to the combined SN-ANAMMOX system. Compared to
previously reported SHARON-ANANMMOX process, the
strategy for obtaining NO
2
−
-N in this article was somewhat
di
fferent (Fig. 4b), in which about 50% of the diluted
raw wastewater was fed into the SN reactor first, and
more than 90% of NH
4
+
-N was oxidized into NO
2
−
-N,
the e
ffluent of the SN reactor was mixed with the other
about 50% of the diluted raw wastewater and then fed
to the ANAMMOX reactor. Compared with the report-
ed SHARON-ANAMMOX process, the SN-ANAMMOX
process has some obvious advantages. First, the DO con-
centration is not necessary to be controlled to the limited
level, and a higher nitrogen loading rate of the reactor may
be maintained. Second, a proper ratio of ammonium to
nitrite in the influent of the ANAMMOX reactor can be
easily controlled only by changing the ratio of the diluted
raw wastewater to e
ffluent of the SN reactor according
for a operational situation of the SN reactor, the nitrite
concentration and the ratio of ammonium to the nitrite
in the influent of the ANAMMOX reactor are the most
important parameters for a stable performance of the
ANAMMOX process. Third, the sludge in the AUSB SN
reactor can be cultivated to granule sludge, and this makes
a relative higher sludge concentration in the SN reactor,
then results in a higher nitrogen loading rate. The operation
of AUSB SN reactor is much easier and simpler, owing to
the dynamic operation strategy for stable operation of the
short-cut nitrification process (Yang et al., 2007), in which
some operational parameters, including temperature, pH
value, DO, and total ammonia concentration, etc., could
a
ffect the growth rate of ammonium oxidizing bacteria
(AOB) and nitrite oxidizing bacteria (NOB) in di
fferent
ways. A non-linear mathematic dynamic model (based on
the modified Monod equations), including all these param-
eters, was proposed and certified for a stable operation
of SN process. According to this model, stable operation
of the SN reactor should be achieved by controlling the
operating conditions, under which the growth rate of AOB
(
μ
AOB
) is greater than that of the NOB (
μ
NOB
). For instance,
in the present article, process conditions were controlled as
follows: temperature was about 25°C, pH value was about
8, DO concentration was about 1.5 mg
/L, total ammonia
(NH
4
+
and NH
3
) concentration in the reactor was about
80 mg
/L. The calculation of the model showed that under
the above condition, the
μ
AOB
is about 0.57 day
−1
, and the
μ
NOB
is about 0.0050 day
−1
. That means the AOB will
grow faster than the NOB in this reactor, i.e., the stable
performance of the SN reactor can be achieved in this
Fig. 4
Flow chart of two NO
2
−
-N obtaining strategies. (a) Reported NO
2
−
-N obtaining strategy of SHARON-ANAMMOX process; (b) NO
2
−
-N
obtaining strategy of SN-ANAMMOX system used in this article.
No. 5
An autotrophic nitrogen removal process: Short-cut nitrification combined with ANAMMOX for treating diluted e
ffluent······
781
reactor, so a SN process was achieved. During that period,
the maximum nitrogen load was 1.47 kg
/(m
3
·day) and both
the ammonium removal e
fficiency and the ratio of NO
2
−
-
N to NOx
−
-N in the e
ffluent of the SN reactor were both
more than 90%.
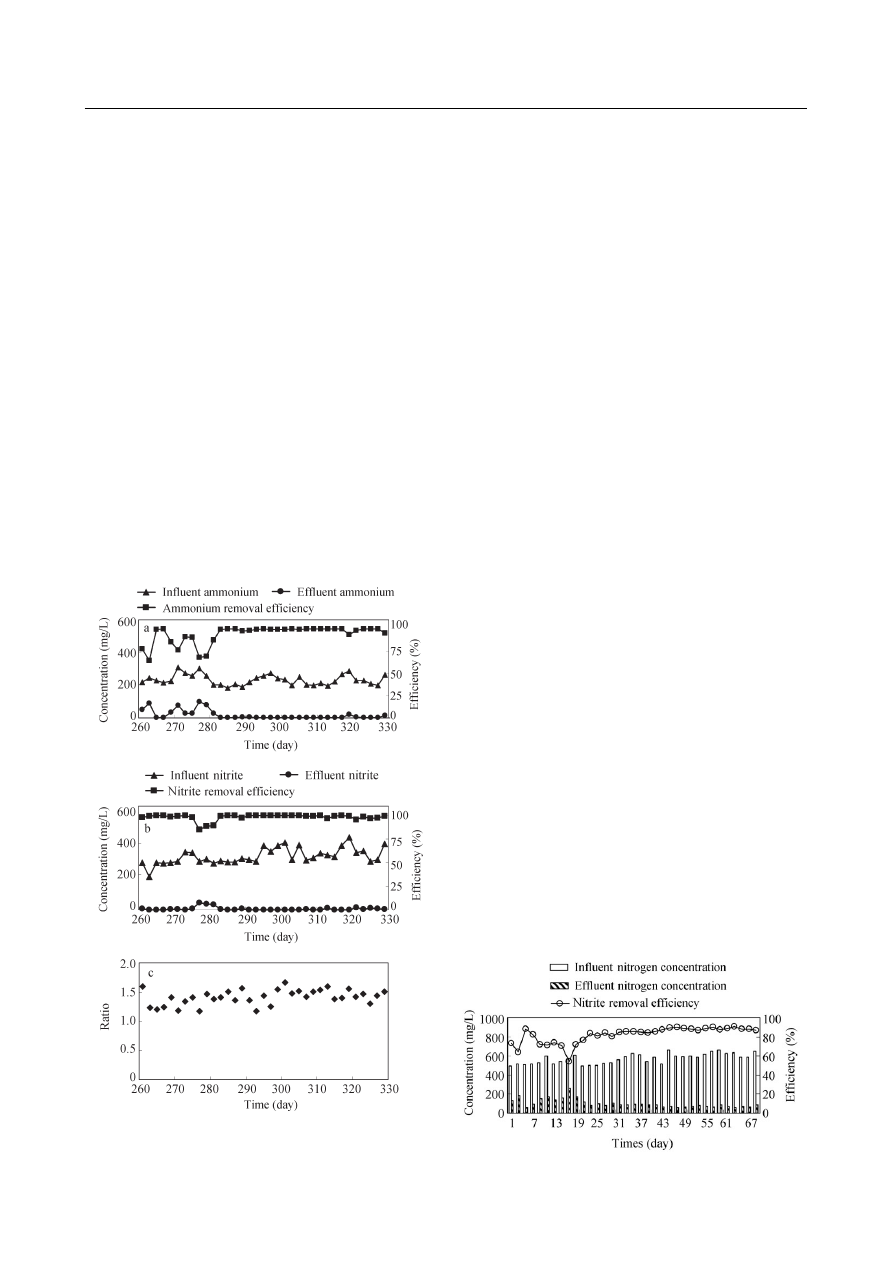
2.2 ANAMMOX process
The concentration of NH
4
+
-N, and NO
2
−
-N of the
ANAMMOX reactor, the corresponding NH
4
+
-N and
NO
2
−
-N removal e
fficiency, and the ratio of removed
NO
2
−
-N to the removed NH
4
+
-N are presented in Fig. 5.
The pH value and temperature were maintained around
7.6 and (31
± 1)°C, respectively, while the HRT was
maintained at 17 hr. As the reactor had been operated
with synthetic water for 260 days prior to the operation
with the diluted raw wastewater and the e
ffluent of the
SN reactor, the results of the ANAMMOX reactor is
presented from day 260. The NH
4
+
-N removal e
fficiency
fluctuated around 75% during day 260 to day 279, due to
an inappropriate ratio of the diluted raw wastewater to the
e
ffluent of the SN reactor. Therefore, the mix ratio was
strictly controlled from day 279 on, then the NH
4
+
-N and
NO
2
−
-N removal e
fficiency subsequently reached above
95%. This suggests that an appropriate ratio of the raw
wastewater to the e
ffluent of the SN reactor is crucial to ob-
Fig. 5
Performance of ANAMMOX reactor. (a) NH
4
+
-N removal
e
fficiency and NH
4
+
-N concentrations in influent and e
ffluent; (b) NO
2
−
-
N removal e
fficiency and NO
2
−
-N concentrations in influent and e
ffluent;
(c) ratio of removed NO
2
−
-N to NH
4
+
-N.
tain higher nitrogen removal e
fficiency in the ANAMMOX
reactor. Subsequently, the NH
4
+
-N and NO
2
−
-N removal
e
fficiency stabilized above 93% and 95%, respectively,
with a maximum influent nitrogen loading rate of about
0.91 kg
/(m
3
·day).
The average ratio of the removed NO
2
−
-N to NH
4
+
-N
was 1.41 : 1, which was similar to the value reported (1.32
: 1) reported by other researchers (Strous et al., 1998).
This suggested that the organic matter in raw wastewater
did not result in a bloom of denitrification bacteria, most
of nitrogen removed in ANAMMOX reactor was removed
by ANAMMOX bacteria. Meanwhile, the average COD
removal e
fficiency was less than 18%, suggesting that the
organic matter remained in raw wastewater was mostly
non-biodegradable; thus, its e
ffect on ANAMMOX bacte-
ria was limited.
Due to strict anaerobic environments are required for
the growth of ANAMMOX bacteria, an oxygen removal
process for influent, in which ammonium and nitrite
concentration were both about 50 mg
/L, was applied
during the start-up period. After 103-day operation, the
removal e
fficiency of ammonium and nitrite were both
above 90%, suggesting a successful start-up. In addition,
the application of BMTM biofilm carriers in ANAMMOX
reactor favored better the growth of ANAMMOX bacteria
on the surfaces and pores of carriers. Except plenty of
biofilm attached to the carriers, in the lower part of the
ANAMMOX reactor, some granular sludge can be found
at the end of the operation, which can contribute to the
nitrogen removal in the reactor.
On the basis of the above results, a novel combined
SN and ANAMMOX process was proven to be a feasible
process to treat the diluted e
ffluent of a UASB reactor
fed by landfill leachate. When the combined system is
practically applied, the e
ffluent of the system could be
used to dilute raw wastewater, so that a proper influent
ammonium concentration and loading rate can be applied
to the SN reactor. In addition, a proper influent ammonium
and nitrite concentrations and total nitrogen loading rate
can be applied to the ANAMMOX reactor.
2.3 Combined process
The concentration of total inorganic nitrogen in the
influent and e
ffluent of the combined system and the
corresponding removal e
fficiency are presented in Fig. 6.
During the period of stable operation, the total average
Fig. 6
Performance of combined SN-ANAMMOX system.

782
Jie Liu et al.
Vol. 22
Fig. 7
SEM photographs of sludge samples at end of operation of SN and ANAMMOX reactors. (a) SN sludge; (b) ANAMMOX sludge.
inorganic nitrogen concentration in influent was 573.6
mg
/L, while the total average nitrogen concentration in
e
ffluent was 77.3 mg/L, with NH
4
+
-N, NO
2
−
-N and NO
3
−
-
N concentrations of 2.2 mg
/L, 2.7 mg/L and 72.3 mg/L,
respectively. This suggested that NO
3
−
-N was a byproduct
of the system, which is a main factor a
ffecting the total
nitrogen removal e
fficiency of the system. At the end of
the operation, the average nitrogen loading rate was 0.56
kg
/(m
3
·day), while the average inorganic nitrogen removal
e
fficiency of the system was 87%. Thus, the feasibility of
applying the SN-ANAMMOX process for treating diluted
raw wastewater was confirmed.
2.4 Morphological characterization of microorganisms
in reactors
When the diluted raw wastewater was applied as influ-
ent, the color of the sludge in SN and ANAMMOX reactors
gradually changed from yellow and red to brown and
grey brown, with time went on. High-magnification SEM
showed the morphological diversity of the microorganisms
that inhabited the SN reactor, which included spherical-,
rod- and long filamentous-shaped microorganisms among
others (Fig. 7a). The configuration of the granule sludge
and its magnified surface from the ANAMMOX reactor are
shown in Fig. 7b. The configuration of the granule was a
regular oval, while the microorganisms inhabiting on the
surface of the granule included some ANAMMOX-like
bacteria (Yang et al., 2006) and a few long filamentous-
shaped microorganisms.
3 Conclusions
The stable operation of SN process was achieved in an
AUSB reactor fed by diluted raw wastewater (the e
ffluent
of UASB reactor treating landfill leachate). The maximum
inorganic nitrogen load of 1.47 kg
/(m
3
·day) resulted in an
ammonium removal e
fficiency above 80% and about an
over 90% of the ratio of NO
2
−
-N to NOx
−
-N in the e
ffluent.
The successful operation of ANAMMOX was achieved
in an UASB reactor fed by a mixture of diluted raw
wastewater (e
ffluent of the UASB reactor treating land-
fill leachate) and the e
ffluent of the SN reactor, with a
maximum inorganic nitrogen loading of 0.91 kg
/(m
3
·day)
and an above 93% of both ammonium and nitrite removal
e
fficiency.
The SN-ANAMMOX combined process was confirmed
to be a feasible process for treating the diluted e
ffluent
of the UASB reactor fed by landfill leachate. The com-
bined process successfully achieved stable performance
for 70 days, with a maximum nitrogen loading rate of
0.63 kg
/(m
3
·day), and a removal efficiency of ammonium
and nitrite both above 93%. However, the average total
inorganic nitrogen removal e
fficiency was only about 87%,
mainly due to the production of nitrate in the ANAMMOX
process.
Acknowledgments
This work was financially supported by the Special Fund
of State Key Joint Laboratory of Environment Simulation

No. 5
An autotrophic nitrogen removal process: Short-cut nitrification combined with ANAMMOX for treating diluted e
ffluent······
783
and Pollution Control, China (No. 08Y03ESPCT) and the
Key Projects in the National Science & Technology Pillar
Program in the Eleventh Five-Year Plan Period of China
(No. 2006BACl9B01).
References
Cema G, Szatkowska B, Plaza E, Trela J, Surmacz-Go’rska J,
2006. Nitrogen removal rates at a technical-scale pilot plant
with the one-stage partial nitritation
/Anammox process.
Water Science and Technology, 54(8): 209–217.
Fux C, Siegrist H, 2004. Nitrogen removal from sludge
digester liquids by nitrification
/denitrification or partial
nitritation
/Anammox: Environmental and economical con-
siderations. Water Science and Technology, 50(10): 19–26.
Ganigue R, Lopez H, Balaguera M D, Colprim J, 2007. Partial
ammonium oxidation to nitrite of high ammonium content
urban landfill leachates. Water Research, 41(15): 3317–
3326.
Horan N J, Gohar H, Hill B, 1997. Application of a granular
activated carbon-biological fluidised bed for the treatment
of landfill leachates containing high concentrations of am-
monia. Water Science and Technology, 36(2-3): 369–375.
Khin T, Annachhatre A P, 2004. Novel microbial nitrogen re-
moval processes. Biotechnology Advances, 22(7): 519–532.
Liang Z, Liu J X, 2008. Landfill leachate treatment with a novel
process: Anaerobic ammonium oxidation (Anammox) com-
bined with soil infiltration system. Journal of Hazardous
Materials, 15(1): 202–212.
Mulder A, van de Graaf A A, Robertson L A, Kuenen J G, 1995.
Anaerobic ammonium oxidation discovered in a denitri-
fying fluidized bed reactor. FEMS Microbiology Ecology,
16(3): 177–184.
Munch E V, Lant P, Keller J, 1996. Simultaneous nitrification and
denitrification in bench-scale sequencing batch reactors.
Water Research, 30(2): 277–284.
Puig S, Vives M T, Corominas L, Balaguer M D, Colprim J, 2004.
Wastewater nitrogen removal in SBRs, applying a step-
feed strategy: From lab-scale to pilot-plant operation. Water
Science and Technology, 50(10): 89–96.
Schmidt I, Sliekers O, Schmid M, Bock E, Fuerst J, Kuenen J G
et al., 2003. New concepts of microbial treatment processes
for the nitrogen removal in wastewater. FEMS Microbiology
Reviews, 27(4): 481–492.
Strous M, Gerven E V, Zheng P, Kuenen J G, Jetten M S, 1997.
Ammonium removal from concentrated waste streams with
the anaerobic ammonium oxidation (Anammox) process
in di
fferent reactor configurations. Water Research, 31(8):
1955–1962.
Strous M, Heijnen J J, Kuenen J G, Jetten M S M, 1998.
The sequencing batch reactor as a powerful tool for the
study of slowly growing anaerobic ammonium-oxidizing
microorganisms. Applied Microbiology and Biotechnology,
50(5): 589–596.
van de Graaf A A, Peter de B, Lesley A R, Mike S M J, Gijs
J K, 1996. Autotrophic growth of anaerobic ammonium-
oxidizing microorganisms in a fluidized bed reactor. Micro-
biology, 142(8): 2187–2196.
Wouter R L van der Star, Wiebe R A, Dennis B, Jan-Willem M,
Takaaki T, Marc S et al., 2007. Startup of reactors for anoxic
ammonium oxidation: Experiences from the first full-scale
anammox reactor in Rotterdam. Water Research, 41(18):
4149–4163.
Yamamoto T, Takaki K, Koyama T, Furukawa K, 2008. Long-
term stability of partial nitritation of swine wastewater
digester liquor and its subsequent treatment by Anammox.
Bioresource Technology, 99(14): 6419–6425.
Yang Y, Zuo J E, Lu Y Q, 2007. Establishment and verification of
dynamic criterion function for stable operation of short-cut
nitrification process. China Environmental Science, 27(4):
543–548.
Yang Y, Zuo J E, Quan Z X, Sung-taik L, Shen P, Gu X S,
2006. Analysis of the species group of anaerobic ammonia
oxidation sludge in UASB reactors. China Environmental
Science, 26(1): 52–56.
Wyszukiwarka
Podobne podstrony:
28, moje 28
CellRESET moje 28 dni biały dzień(1)
CellRESET moje 28 dni biały dzień
CellRESET moje 28 dni zielony dzień
moje 28
CellRESET moje 28 dni zielony dzień(2)
CellRESET moje 28 dni zielony dzień
CellRESET moje 28 dni biały dzień
CellRESET moje 28 dni zielony dzień
CellRESET moje 28 dni biały dzień(3)
moje, Katecheza 28, Katecheza 28
28 Relative Clauses, Tragedia Piotra Włostowica, Moje dokumenty
PRAWO NOTATKI 28.11.2010 moje !!, Prawo
moje sprawozdanie 28
Kosci, kregoslup 28[1][1][1] 10 06 dla studentow
Ch 28 Pelites
więcej podobnych podstron