
Unit 5: Chapter 6
Alkyl Halides: Nucleophilic
Substitution and Elimination
Organic Chemistry, 5
th
Edition
L. G. Wade, Jr.
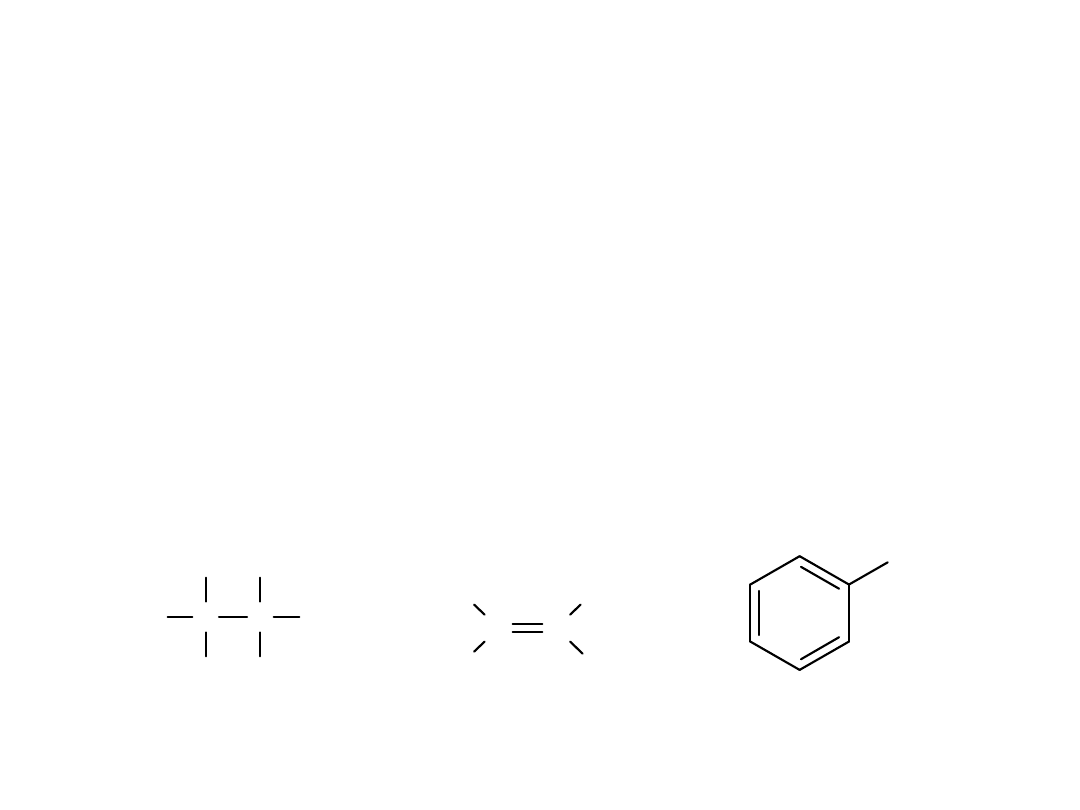
Alkyl Halides
2
Classes of Halides
• Alkyl
: Halogen, X, is directly bonded to
sp
3
carbon.
• Vinyl
: X is bonded to sp
2
carbon of alkene.
• Aryl:
X is bonded to sp
2
carbon on
benzene ring. Examples:
C
H
H
H
C
H
H
Br
alkyl halide
C C
H
H
H
Cl
vinyl halide
I
aryl halide
=
>
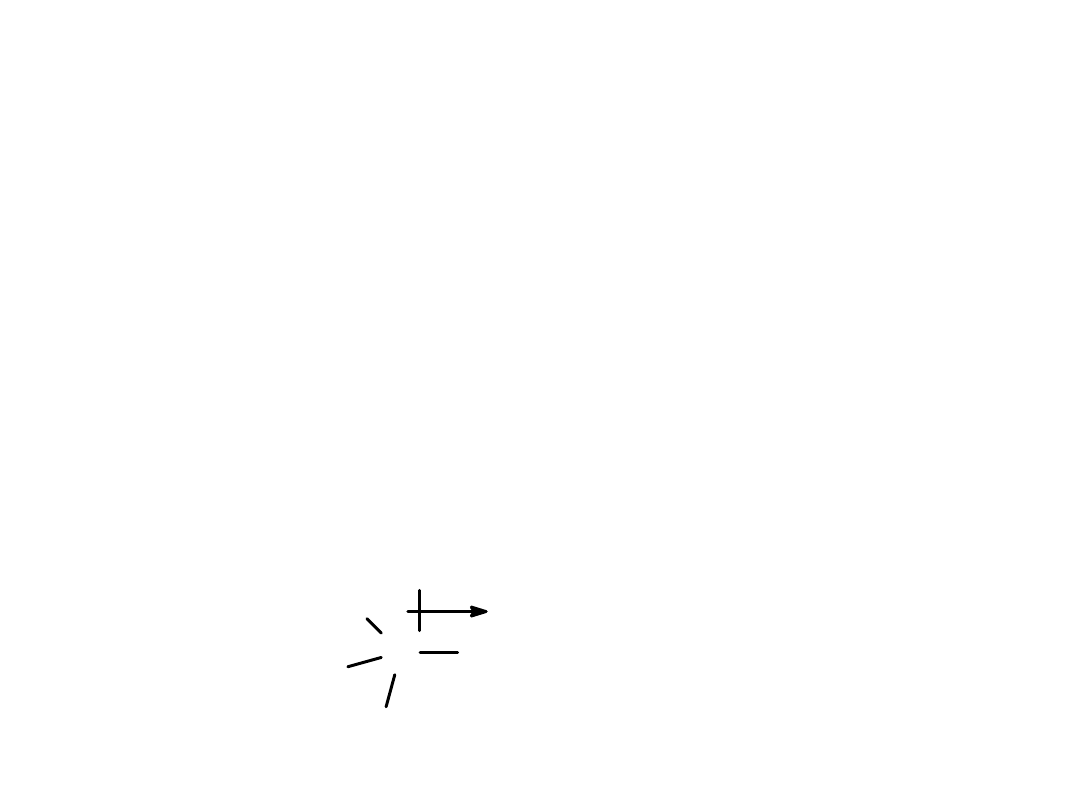
Alkyl Halides
3
Polarity and Reactivity
• Halogens are more electronegative than C.
• Carbon-halogen
bond is polar
, so carbon
has partial positive charge.
• Carbon can be
attacked
by a nucleophile.
• Halogen can
leave
with the electron pair.
=>
C
H
H
H
Br
+ -

Alkyl Halides
4
Classes of Alkyl Halides
• Methyl halides
: only one C, CH
3
X
• Primary
: C to which X is bonded has
only one C-C bond, CH
3
CH
2
X.
• Secondary
: C to which X is bonded has
two C-C bonds, (CH
3)2
HCX.
.
• Tertiary
: C to which X is bonded has
three C-C bonds , (CH
3)3
CX.
=>
Alkyl Halides
5
Classify These:
CH
3
CH CH
3
Cl
CH
3
CH
2
F
(CH
3
)
3
CBr
CH
3
I
=>
Alkyl Halides
6
Dihalides
• Geminal dihalide
: two halogen atoms
are bonded to the same carbon
• Vicinal dihalide
: two halogen atoms
are bonded to adjacent carbons.
C
H
H
H
C
H
Br
Br
geminal dihalide
C
H
H
Br
C
H
H
Br
vicinal dihalide
=
>
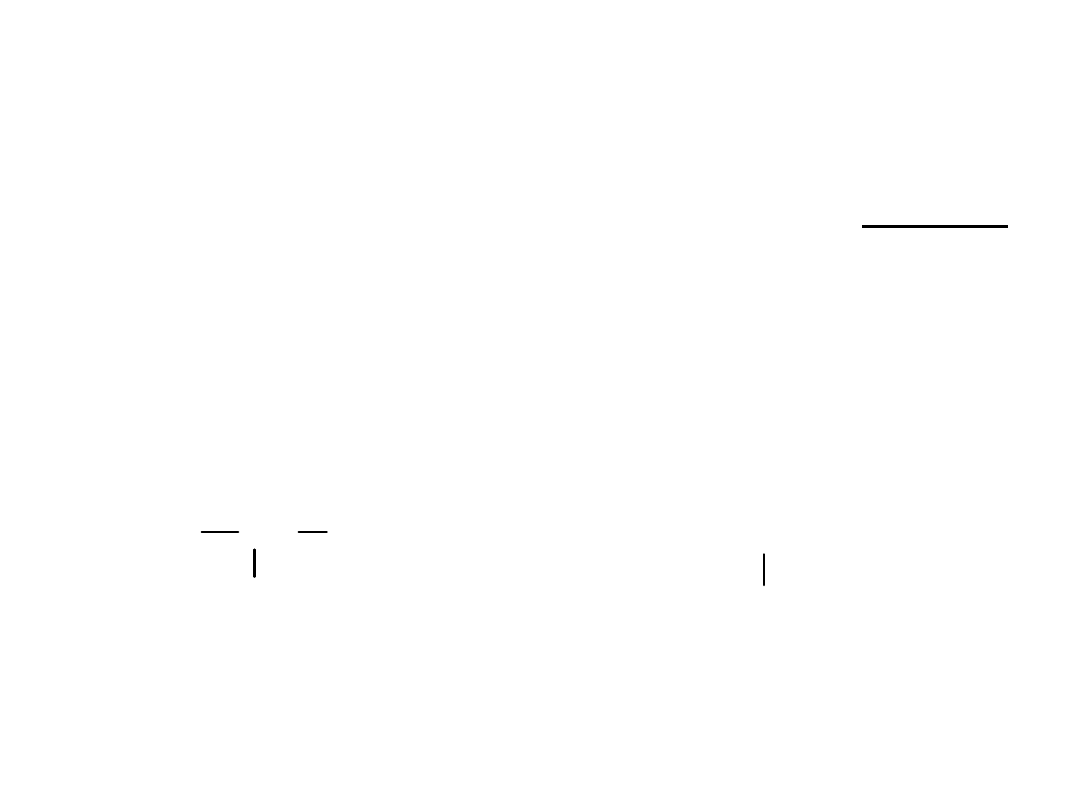
Alkyl Halides
7
IUPAC Nomenclature
• Name as
halo
alkane.
• Choose the
longest carbon chain
, even if
the halogen is not bonded to any of those
C’s.
• Use
lowest possible
numbers for position.
CH
3
CH CH
2
CH
3
Cl
CH
3
(CH
2
)
2
CH(CH
2
)
2
CH
3
CH
2
CH
2
Br
2-chlorobutane
4-(2-bromoethyl)heptane
=
>
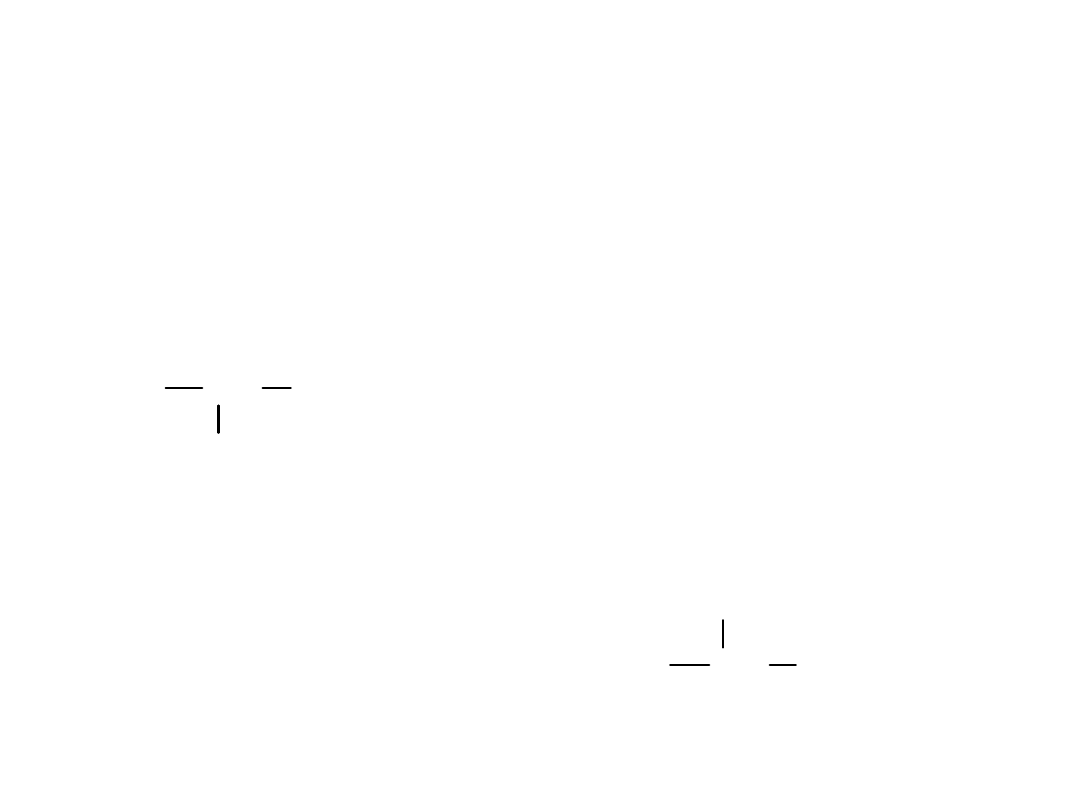
Alkyl Halides
8
Systematic
Common
Common
Names
• Name as alkyl
halide
.
• Useful only for
small
alkyl groups.
• Name these:
CH
3
CH CH
2
CH
3
Cl
(CH
3
)
3
CBr
CH
3
CH
CH
3
CH
2
F
=>
secbutyl
chloride
2-chlorobutane
tert-Butyl
bromide
2bromo-2-methylpropane
1-flouro-2-methylpropane

Alkyl Halides
9
“Trivial” Names
• CH
2
X
2
called methylene halide.
• CHX
3
is a haloform.
• CX
4
is carbon tetrahalide.
• Examples:
CH
2
Cl
2
is methylene chloride
CHCl
3
is chloroform
CCl
4
is carbon tetrachloride.
=>

Alkyl Halides
10
Uses of Alkyl Halides
• Solvents
- degreasers and dry cleaning fluid
• Reagents
for synthesis of other compounds
• Anesthetic
: Halothane is CF
3
CHClBr
CHCl
3
used originally (toxic and carcinogenic)
• Freons
, chlorofluorocarbons or CFC’s
Freon 12, CF
2
Cl
2
, now replaced with Freon 22,
CF
2
CHCl, not as harmful to ozone layer.
• Pesticides
- DDT banned in U.S.
=>

Alkyl Halides
11
Dipole Moments
•
=
4.8 x
x d
, where is the charge
(proportional to EN, electronegativity) and
d is the distance (bond length) in Angstroms.
• Electronegativities
: F > Cl > Br > I
• Bond lengths
: C-F < C-Cl < C-Br < C-I
• Bond dipoles
:
C-Cl > C-F > C-Br > C-I
1.56 D 1.51 D 1.48 D 1.29 D
• Molecular dipoles
depend on shape, too!
=>

Alkyl Halides
12
Boiling Points
• Greater intermolecular forces, higher b.p.
dipole-dipole attractions not significantly
different for different halides
London forces greater for larger atoms
• Greater mass, higher b.p
.
• Spherical shape decreases b.p.
(CH
3
)
3
CBr CH
3
(CH
2
)
3
Br
73C 102C
=>

Alkyl Halides
13
Densities
• Alkyl fluorides and chlorides
less dense
than water.
• Alkyl dichlorides, bromides, and iodides
more dense
than water.
=>
Alkyl Halides
14
Preparation of RX
• Free radical halogenation
produces mixtures, not good lab
synthesis
unless: all H’s are equivalent, or
halogenation is highly selective.
• Free radical allylic halogenation
produces alkyl halide with double bond
on the neighboring carbon.
=>
Alkyl Halides
15
Chlorination of Methane
• Requires
heat or light
for initiation.
• The most effective wavelength is
blue
,
which is absorbed by chlorine gas.
• Lots of product formed from absorption of
only
one photon
of light (
chain reaction
).
=>
C
H
H
H
H + Cl
2
heat or light
C
H
H
H
Cl + HCl

Alkyl Halides
16
Free-Radical Chain
Reaction
• Initiation
generates a reactive
intermediate
.
• Propagation
: the
intermediate reacts
with
a stable molecule to produce another
reactive
intermediate
(and a product
molecule).
• Termination
: side reactions that destroy
the reactive intermediate.
=>
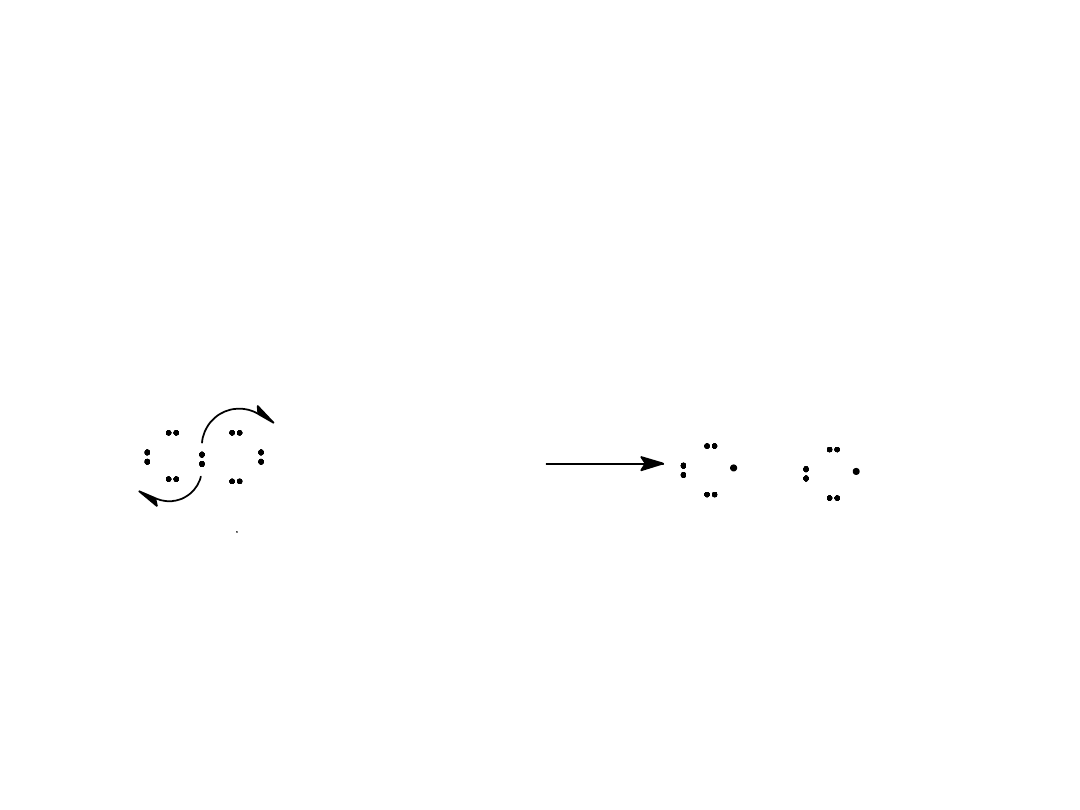
Alkyl Halides
17
Initiation Step
A chlorine molecule splits
homolytically
into chlorine atoms
(free radicals)
=
>
Cl Cl + photon (
h
)
Cl + Cl
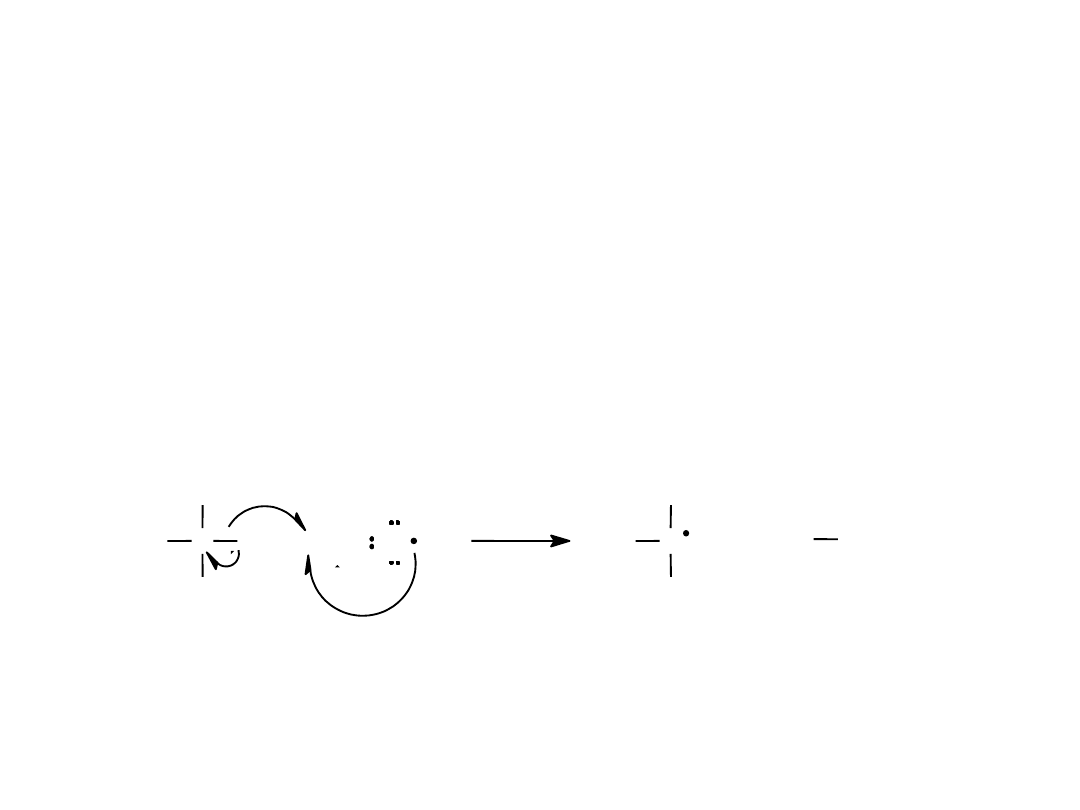
Alkyl Halides
18
Propagation Step (1)
The chlorine
atom collides with a
methane molecule and abstracts
(removes) a H, forming another
free
radical
and one of the products (HCl).
C
H
H
H
H
Cl
+
C
C
H
H
H
H
H
H
+ H Cl
=
>
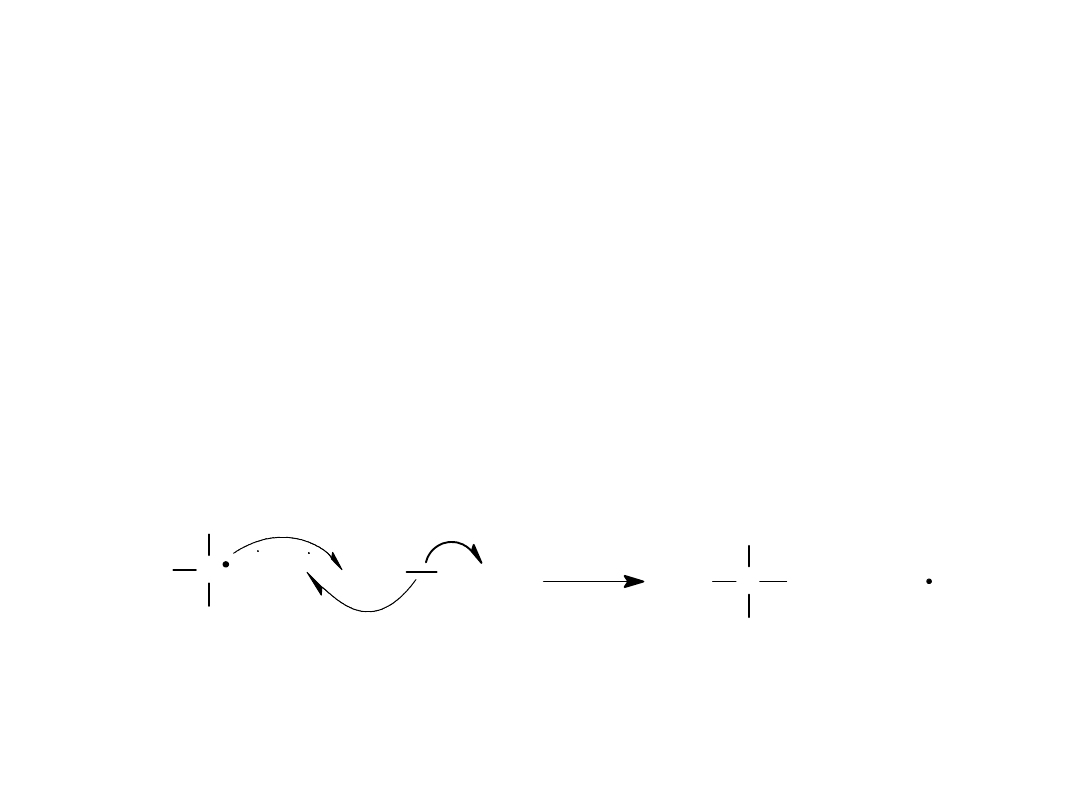
Alkyl Halides
19
Propagation Step (2)
The methyl free radical collides with
another chlorine molecule, producing
the other product (methyl chloride)
and regenerating the chlorine radical.
C
H
H
H
+
Cl Cl
C
H
H
H
Cl
+
Cl
=
>
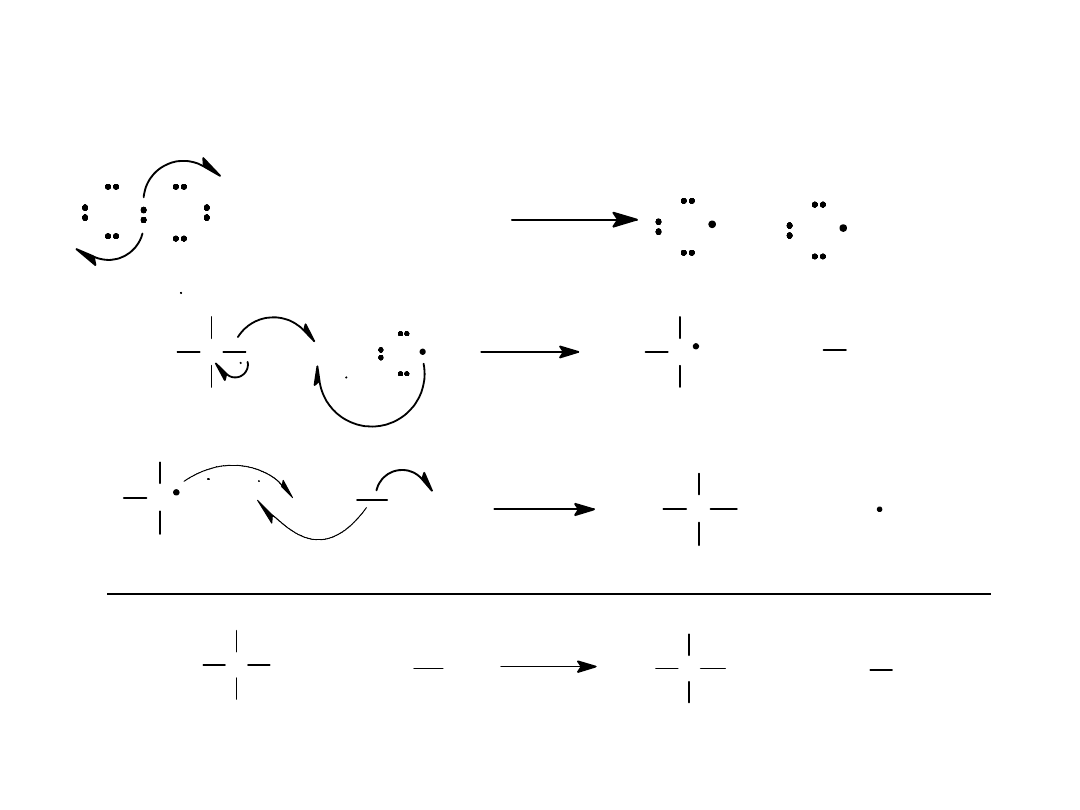
Alkyl Halides
20
Overall Reaction
C
H
H
H
H
Cl
+
C
H
H
H
+ H Cl
C
H
H
H
+
Cl Cl
C
H
H
H
Cl
+
Cl
C
H
H
H
H + Cl Cl
C
H
H
H
Cl
+ H Cl
=
>
Cl Cl + photon (
h
)
Cl + Cl
Alkyl Halides
21
Termination Steps
• Collision of any two free radicals
• Combination of free radical with
contaminant or collision with wall.
C
H
H
H
Cl
+
C
H
H
H
Cl
Can you suggest others?
=>
Alkyl Halides
22
Halogenation of
Alkanes
• All H’s equivalent. Restrict amount of
halogen to prevent di- or trihalide
formation
• Highly selective: bromination of 3C
=>
+ HBr
H
Br
h
Br
2
+
H
H
90%
+ HBr
CH
3
C
CH
3
CH
3
Br
h
Br
2
+
CH
3
C
CH
3
CH
3
H
Alkyl Halides
23
Allylic Halogenation
• Allylic radical is resonance stabilized.
• Bromination occurs with good yield at
the allylic position (sp
3
C next to C=C).
• Avoid a large excess of
Br
2
by using
N-bromosuccinimide (NBS)
to generate
Br
2
as product HBr is formed.
N
O
O
Br + HBr
N
O
O
H + Br
2
=>
Alkyl Halides
24
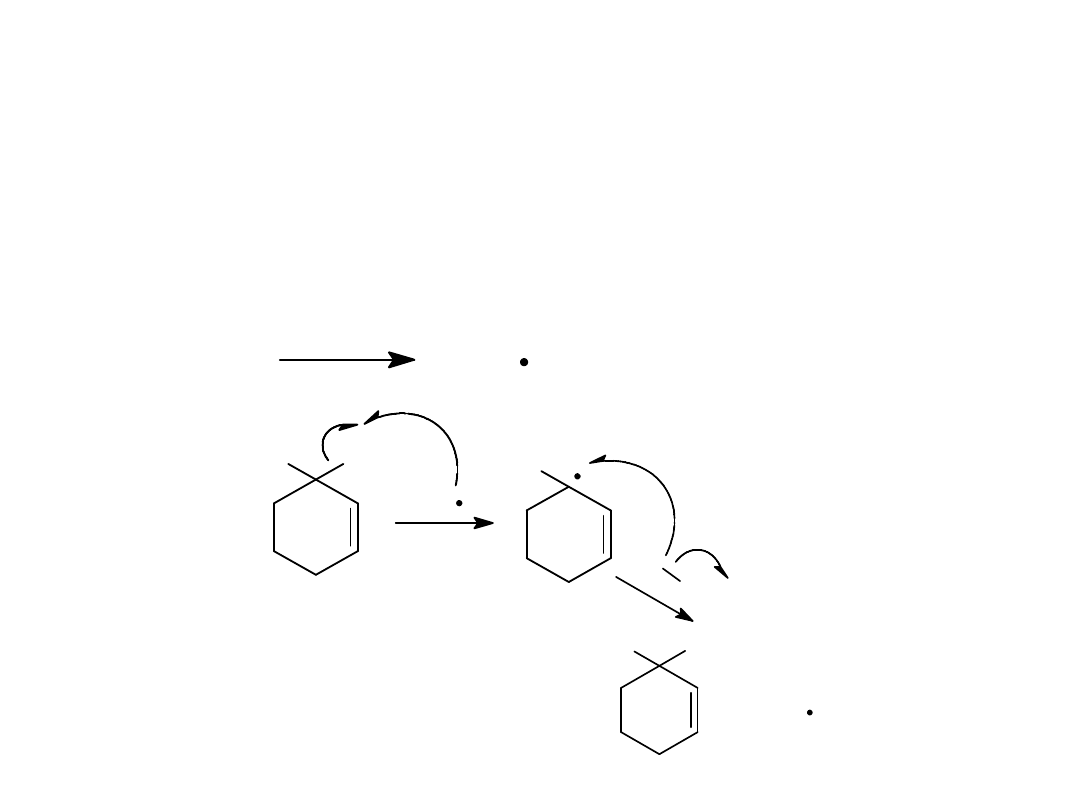
Reaction Mechanism
Free radical chain reaction
initiation, propagation,
termination.
H
H
Br
H
+ HBr
Br
Br
H
Br
+ Br
=>
2Br
Br
2
h
Allylic Halogenation
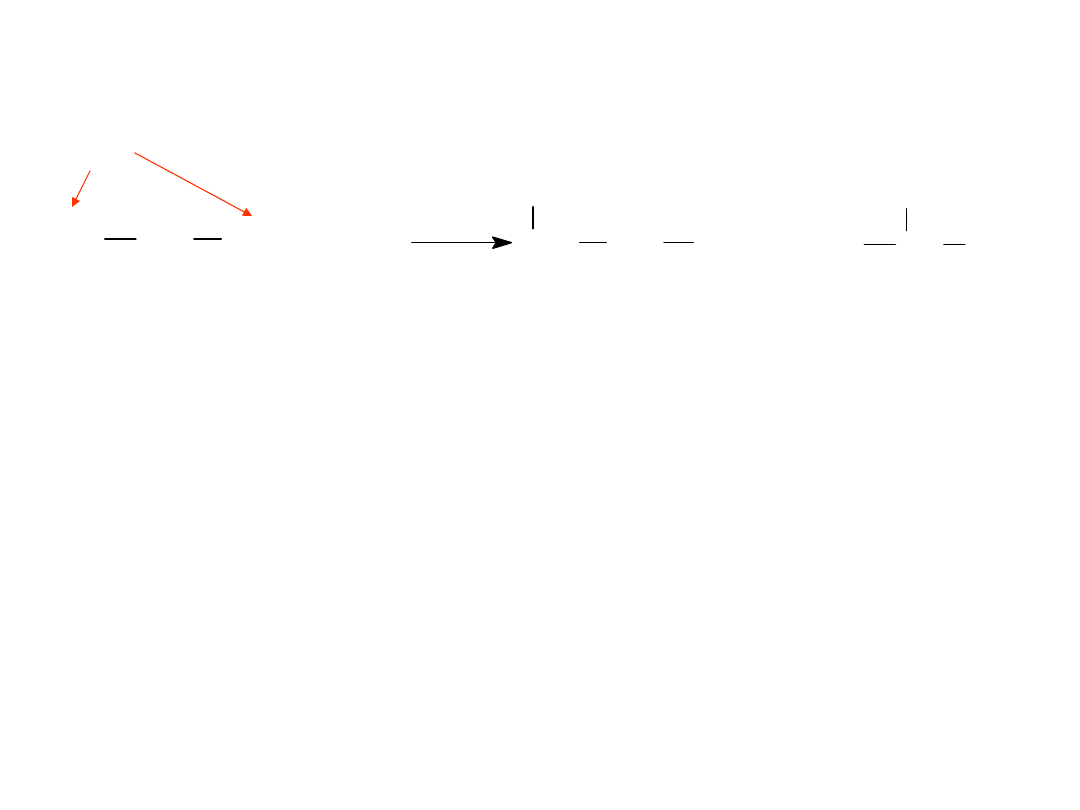
Alkyl Halides
25
Chlorination of Propane
• There are
six 1 H’s
and
two 2’s
. We
expect 3:1
product mix, or 75% 1-
chloropropane and 25% 2-chloropropane.
• Typical product mix:
40%
1-chloropropane
and
60%
2-chloropropane.
• Therefore, not all H’s are equally reactive.
=>
1 C
2 C
CH
3
CH
2
CH
3
+ Cl
2
h
CH
2
Cl
CH
2
CH
3
+ CH
3
CH
Cl
CH
3
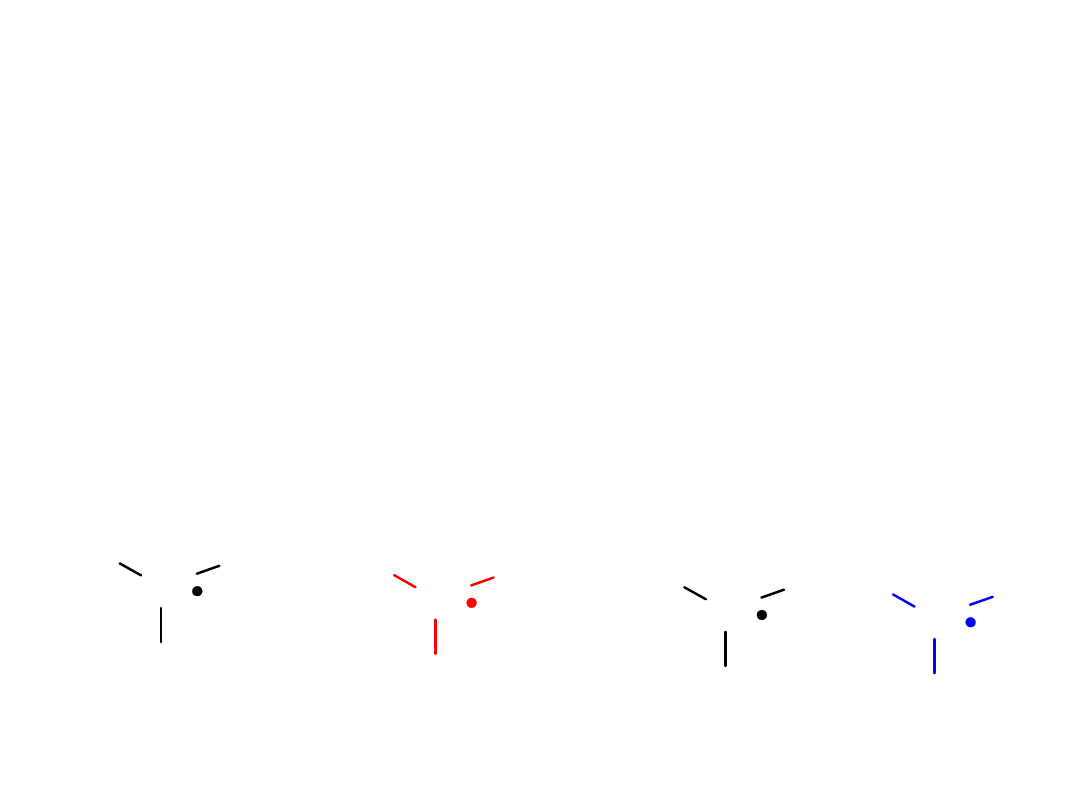
Alkyl Halides
26
Energy required to break a C-H bond
decreases as substitution on the carbon
increases.
Stability: 3° > 2 ° > 1° > methyl
H(kcal) 91, 95, 98, 104
Free Radical Stabilities
C
+
CH
3
CH
3
C
H
3
C
+
CH
3
H
C
H
3
C
+
H
H
C
H
3
C
+
H
H
H

Alkyl Halides
27
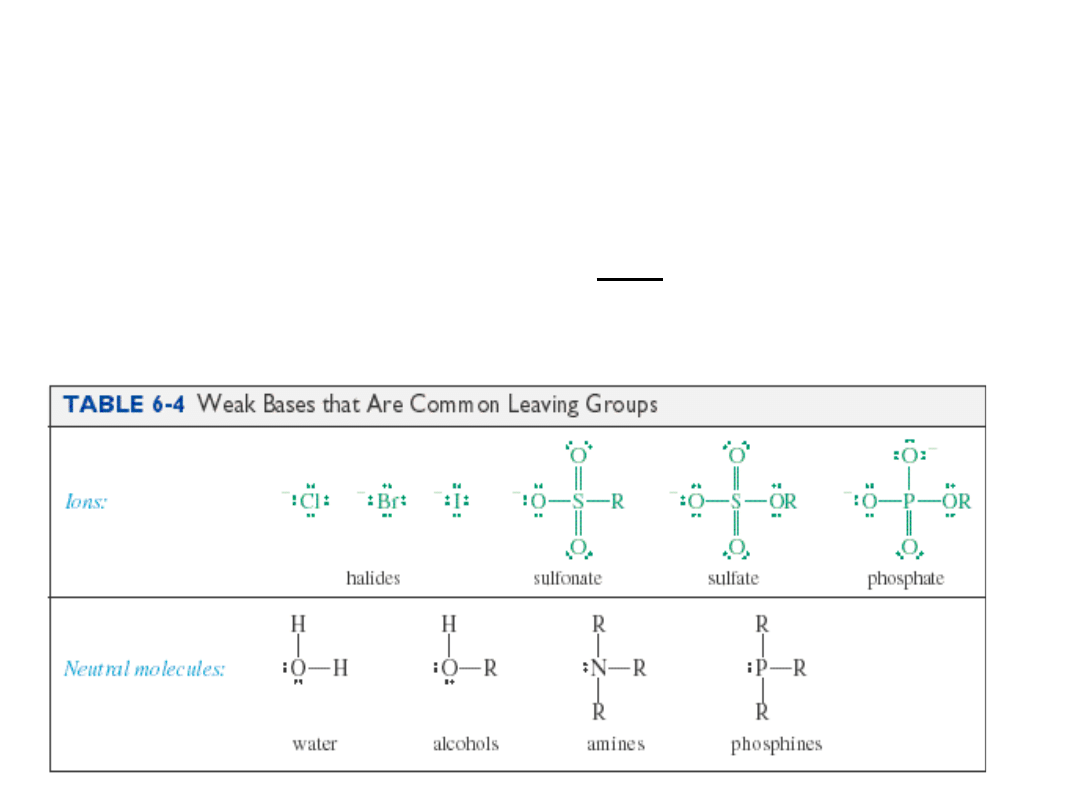
Substitution Reactions
• The halogen atom on the alkyl halide is
replaced
with another group.
• Since the halogen is more electronegative
than carbon, the
C-X
bond breaks
heterolytically and
X
-
leaves.
• The group replacing X
-
is a
nucleophile.
=>
C C
H X
+ Nuc:
-
C C
H Nuc
+ X:
-
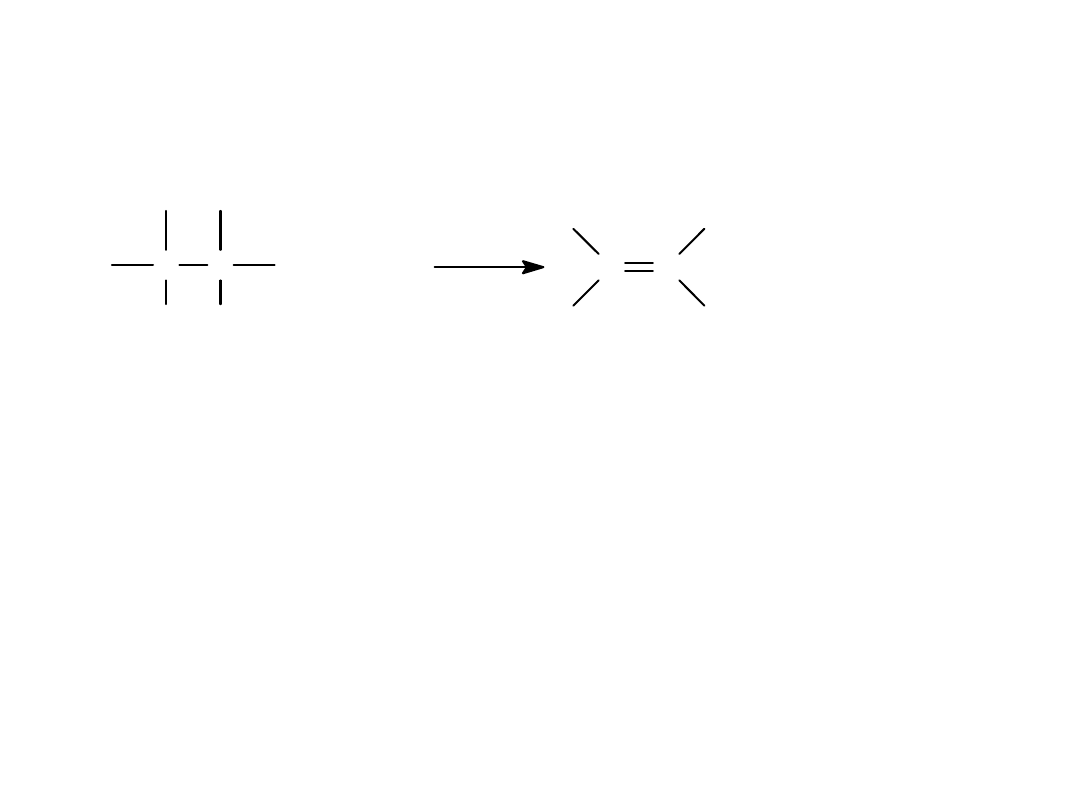
Alkyl Halides
28
Elimination Reactions
• The alkyl halide loses halogen as
a halide
ion, and also loses
H
+
on the adjacent
carbon to a base.
• A
pi bond
is formed. Product is
alkene
.
• Also called
dehydrohalogenation (-HX).
=>
C C
H X
+ B:
-
+ X:
-
+ HB
C C
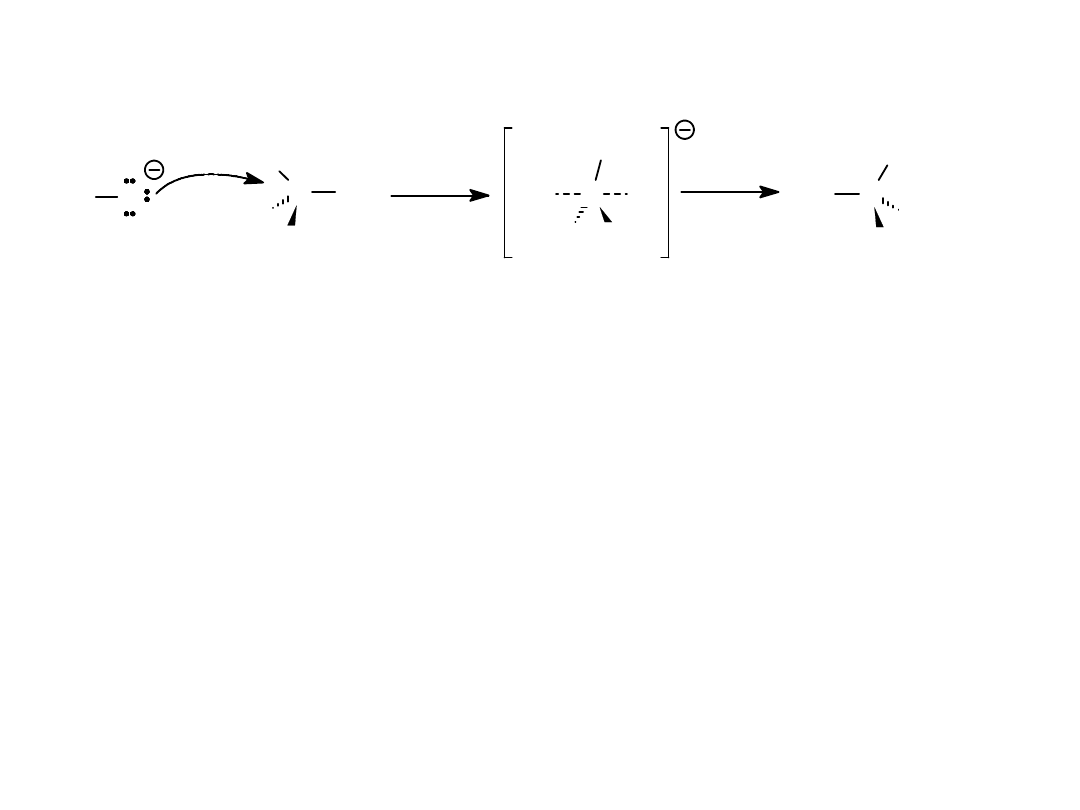
Alkyl Halides
29
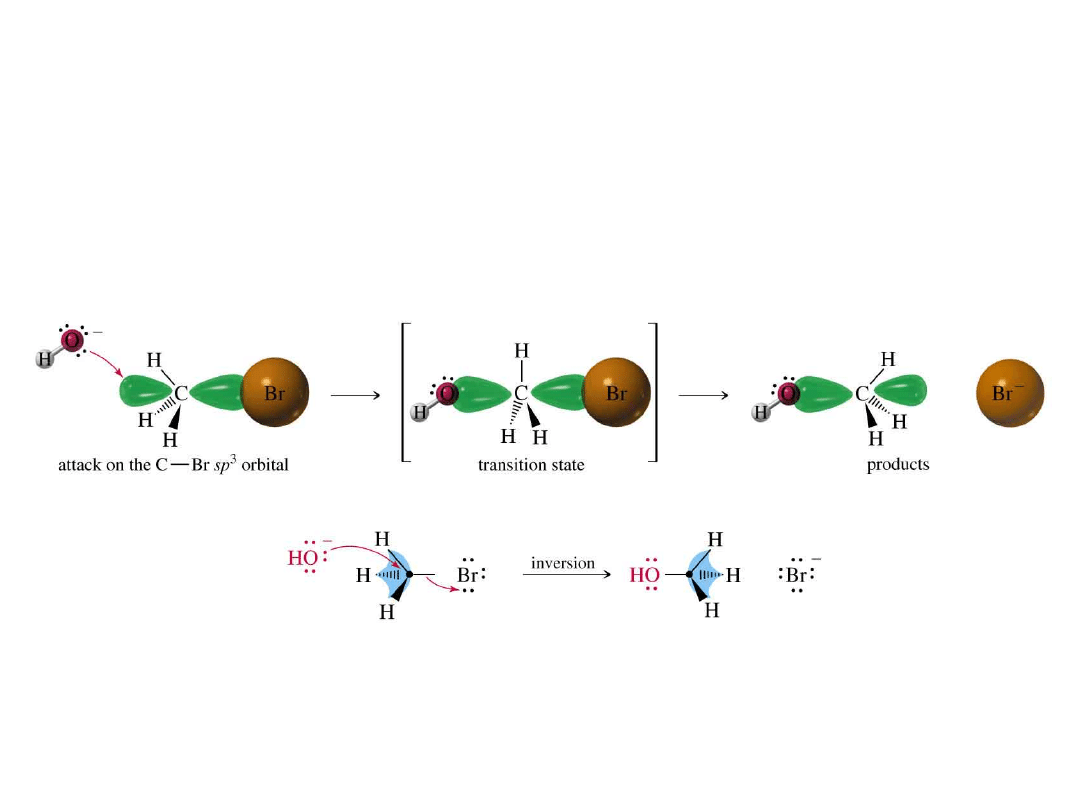
S
N
2 Mechanism
• Bimolecular
nucleophilic substitution.
• Concerted reaction
: new bond forming
and old bond breaking at same time.
•
Rate is first order in each reactant.
Rate is first order in each reactant.
• Walden inversion.
=>
C
H
Br
H
H
H O
C
HO
Br
H
H
H
C
HO
H
H
H
+ Br
-
Alkyl Halides
30
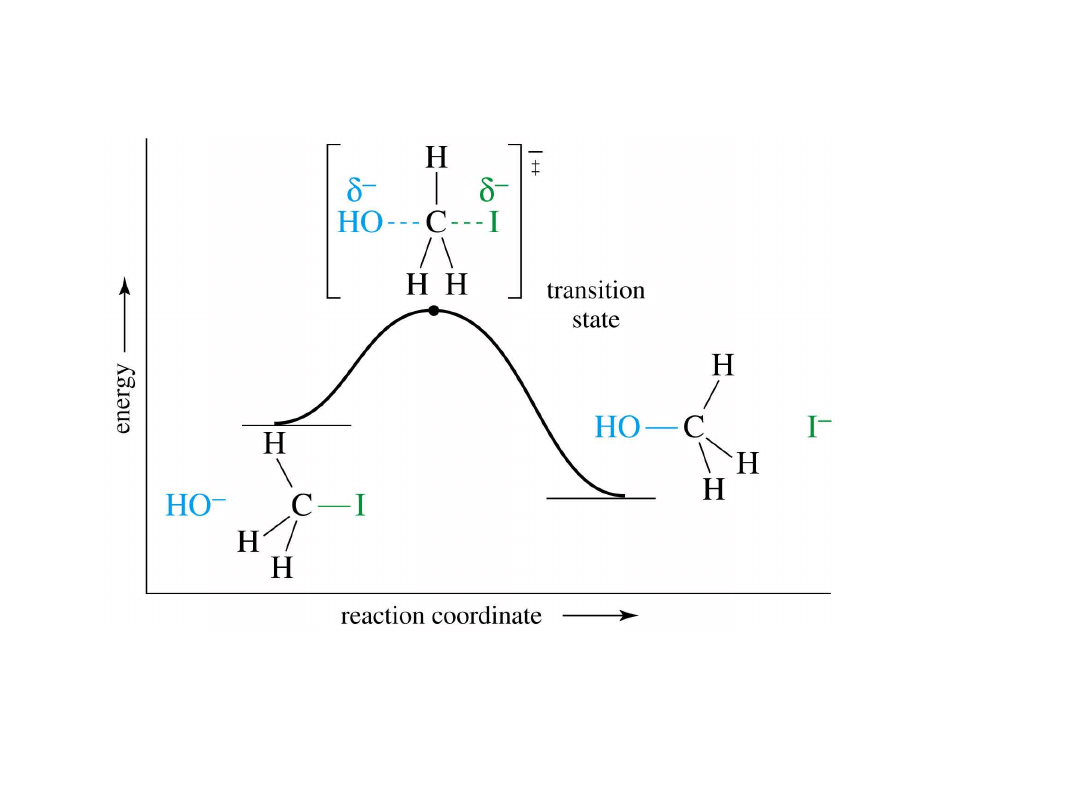
S
N
2 Energy Diagram
• One-step reaction
.
• Transition state is highest in energy. =>
Alkyl Halides
31

Alkyl Halides
32
7_104.exe
S
N
2 reaction of oxonum ion
7_108.exe
Reaction of alcohols with thionyl chloride
Alkyl Halides
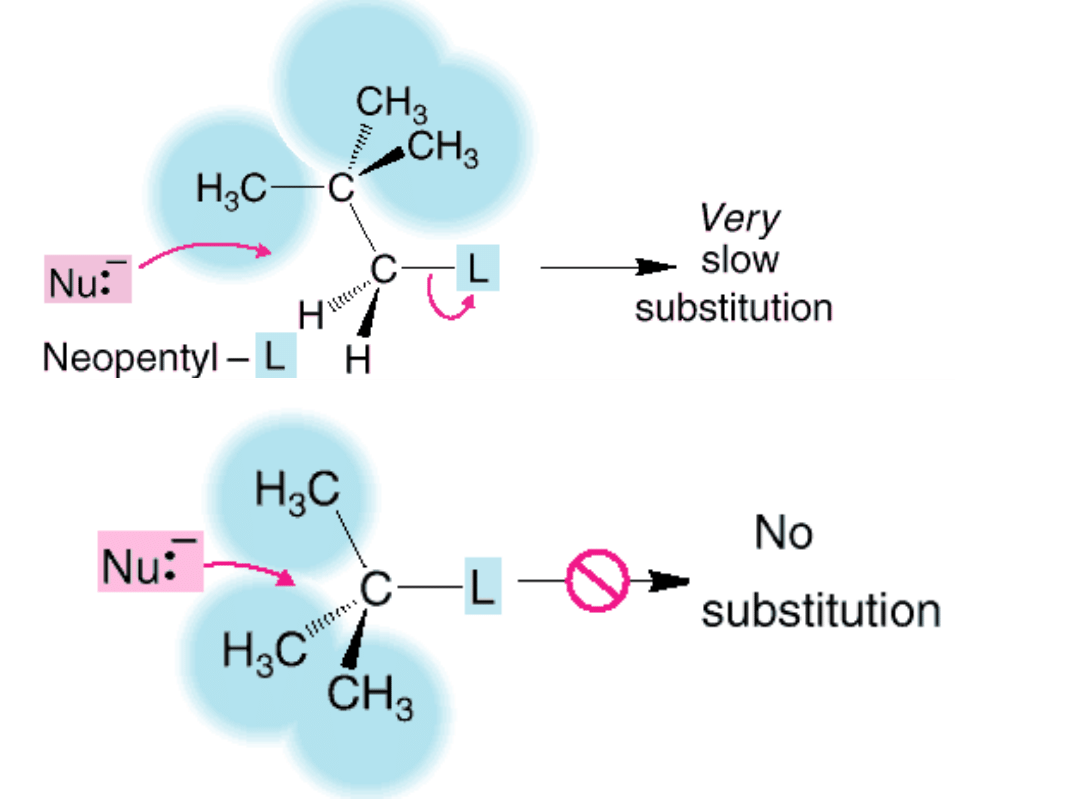
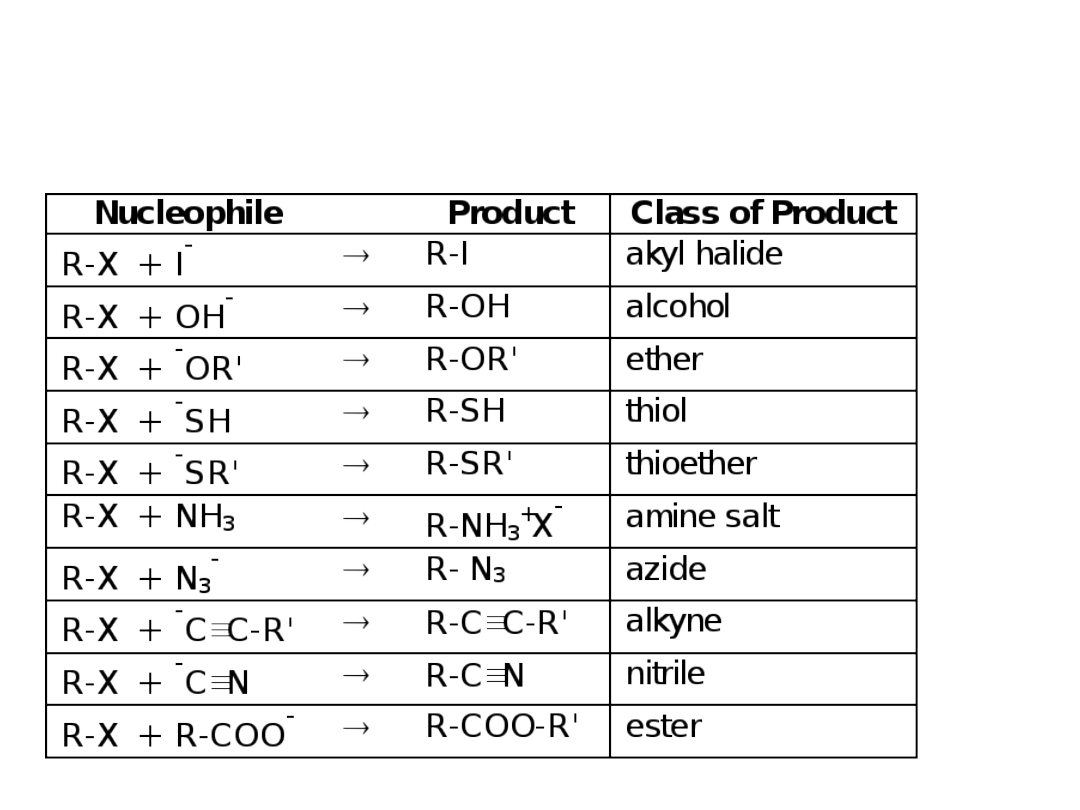
33
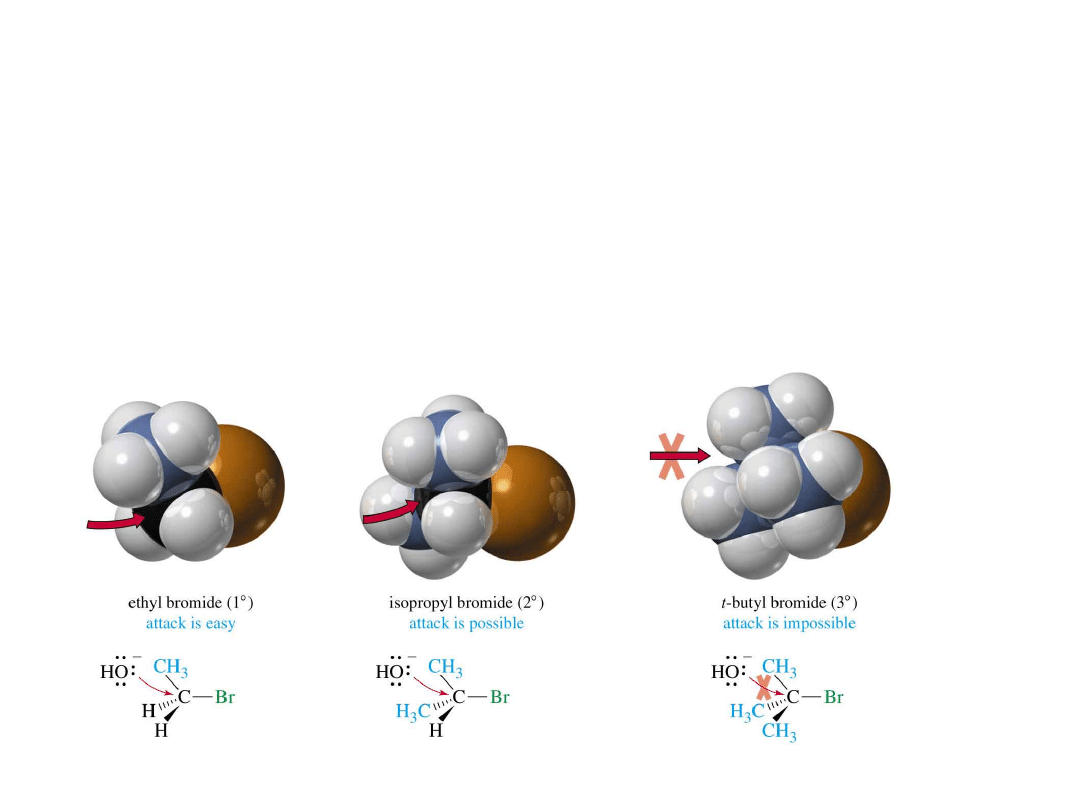
Steric
Hindrance
Relative rate of reaction for some alkyl halides
Alkyl Halide Type of C
Relative Rate
H
CH
2
X
methyl
30
CH
3
CH
2
X
primary
1
CH
3
CH
2
CH
2
X
primary 0.4
CH
3
CH
2
CH
2
CH
2
X
primary
0.4
(CH
3
)
2
CHX
secondary
0.025
(CH
3
)
3
CX
tertiary
~0.00
Size of
nucleophile
and hindrance
Rate = Ze
Rate = Ze
-
-
Ea/RT
Ea/RT
Alkyl Halides
34
Uses for S
N
2 Reactions
• Synthesis of other classes of compounds.
• Halogen exchange reaction.
=
>

Alkyl Halides
35
S
N
2: Nucleophilic Strength
• Stronger nucleophiles react faster.
• Strong bases are strong nucleophiles,
but not all strong nucleophiles are basic.
=>

Alkyl Halides
36
Trends in Nuc. Strength
• Of a conjugate acid-base pair, the
base
is stronger:
OH
-
> H
2
O, NH
2-
> NH
3
• Decreases
left to right
on Periodic Table. More
electronegative atoms less likely to form new bond:
OH
-
> F
-
, NH
3
> H
2
O
• Increases
down Periodic
Table, as size and
polarizability increase:
I
-
> Br
-
> Cl
-
=>
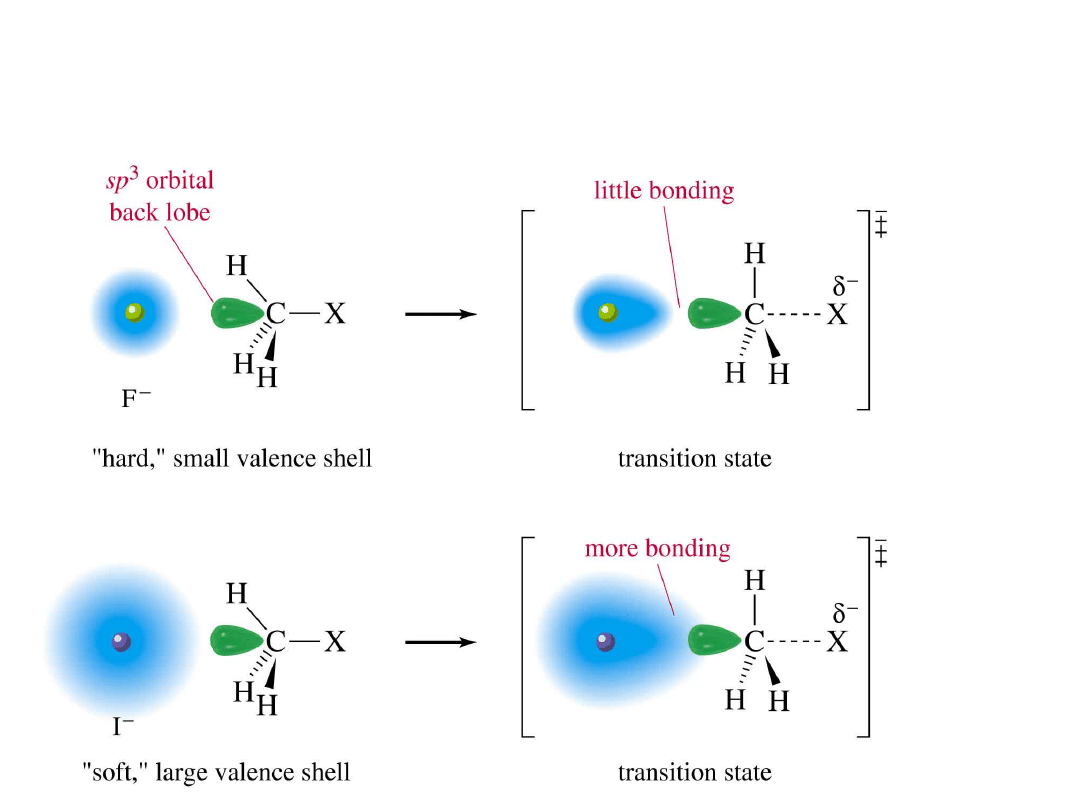
Alkyl Halides
37
Polarizability Effect
=
>
Alkyl Halides
38
Bulky Nucleophiles
Sterically hindered for attack on
carbon, so weaker nucleophiles.
CH
3
CH
2
O
ethoxide (unhindered)
weaker base, but stronger nucleophile
C
CH
3
H
3
C
CH
3
O
t-butoxide (hindered)
stronger base, but weaker nucleophile
=>
Alkyl Halides
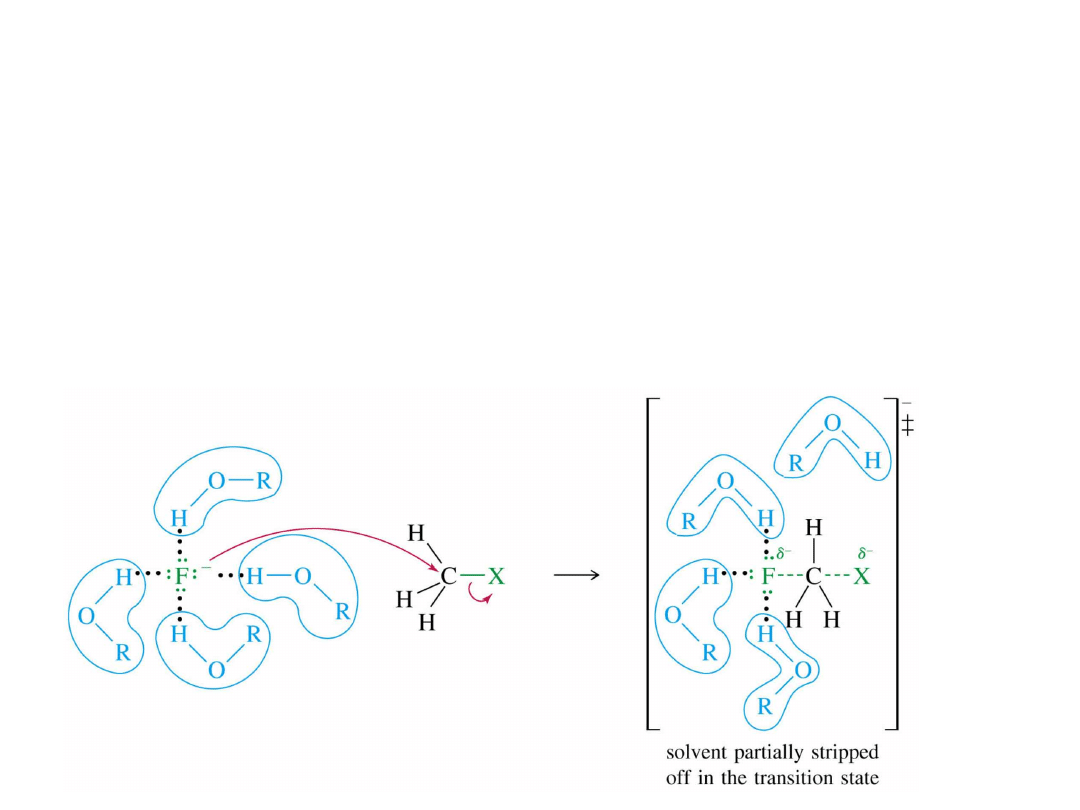
39
Solvent Effects (1)
Polar protic solvents
(O-H or N-H) reduce
the strength of the nucleophile.
Hydrogen bonds must be broken before
nucleophile can attack the carbon.
=
>
Alkyl Halides
40
Solvent Effects (2)
• Polar
aprotic
solvents (no O-H or N-H) do
not form hydrogen bonds with nucleophile
• Examples:
CH
3
C N
acetonitrile
C
O
H
3
C
CH
3
acetone
=>
dimethylformamide
(DMF)
C
H
O
N
CH
3
CH
3
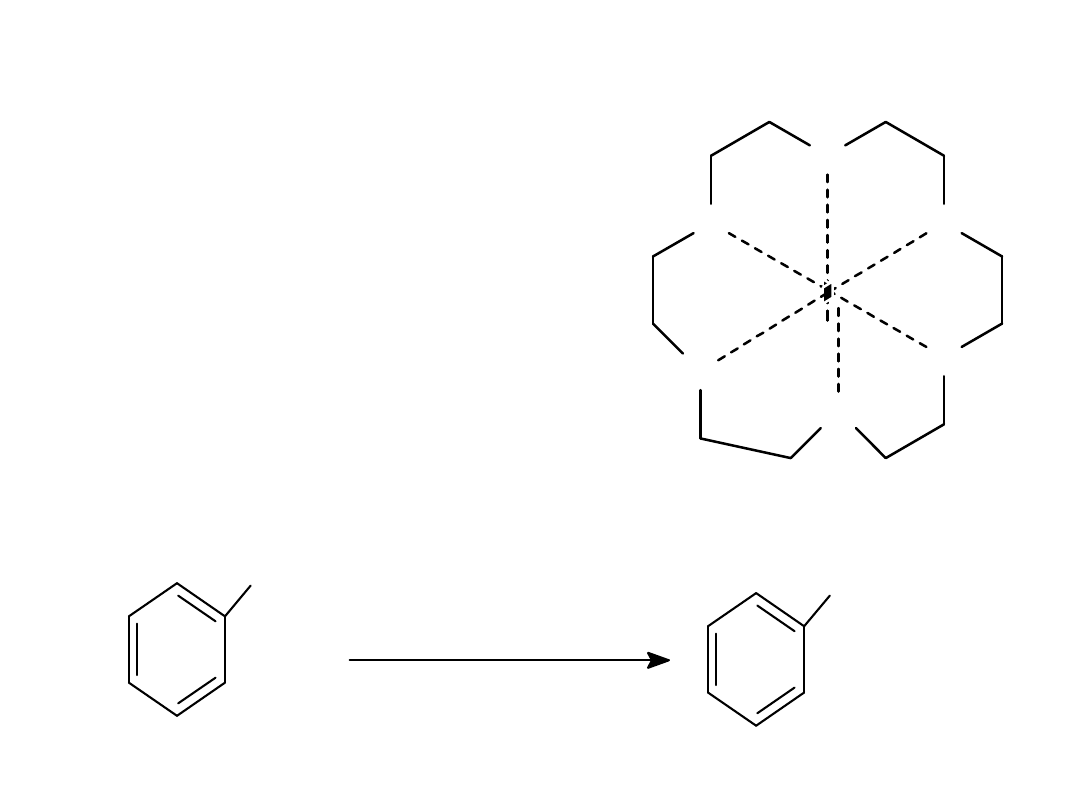
Alkyl Halides
41
Crown Ethers
• Solvate the
cation
, so
nucleophilic strength
of the anion increases.
• Fluoride becomes a
good nucleophile.
O
O
O
O
O
O
K+
18-crown-6
CH
2
Cl
KF, (18-crown-6)
CH
3
CN
CH
2
F
=>
Alkyl Halides
42
S
N
2: Reactivity of Substrate
• Carbon must be
partially positive
.
• Must have a
good leaving group
• Carbon must
not be sterically hindered
.
=>
Alkyl Halides
43
Leaving Group Ability
• Electron-withdrawing
• Stable once it has left (not a strong base)
• Polarizable to stabilize the transition state.
=>

Alkyl Halides
44
Structure of Substrate
• Relative rates for S
N
2:
CH
3
X > 1° > 2°
>> 3°
• Tertiary halides do not react via the
S
N
2 mechanism, due to steric
hindrance. =>
Alkyl Halides
45
Stereochemistry of S
N
2
Walden inversion
=>

Alkyl Halides
46
S
N
1 Reaction
• Unimolecular
nucleophilic substitution.
• Two step reaction
with carbocation
intermediate.
• Rate is first order
in the alkyl halide,
zero order in the nucleophile.
• Racemization
occurs.
=>
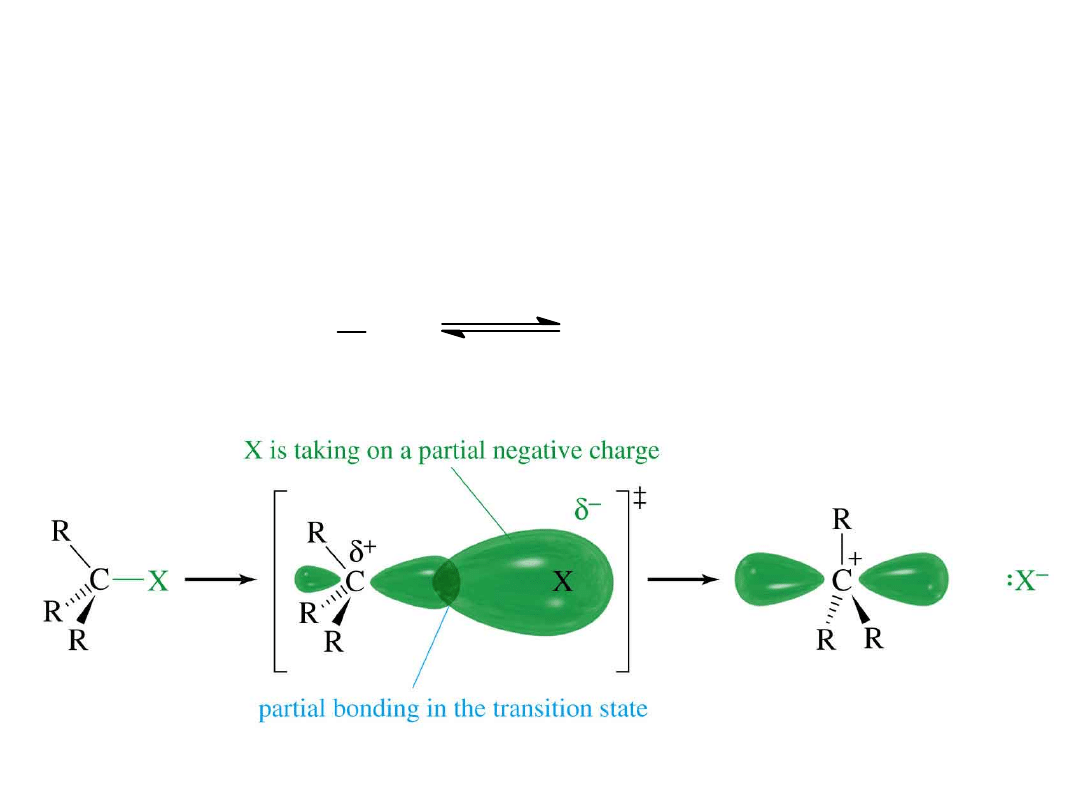
Alkyl Halides
47
S
N
1 Mechanism (1)
Formation of carbocation (slow)
(CH
3
)
3
C Br
(CH
3
)
3
C
+
+ Br
-
=>
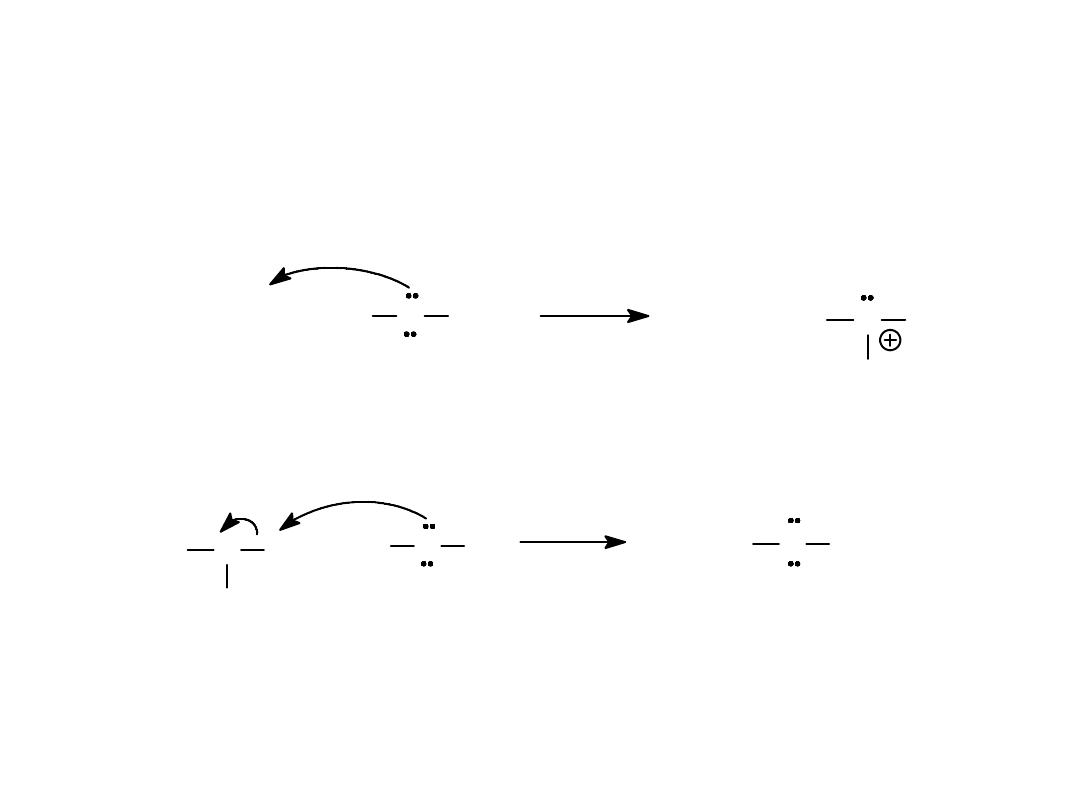
Alkyl Halides
48
S
N
1 Mechanism (2)
• Nucleophilic attack
(CH
3
)
3
C
+
+ H O H
(CH
3
)
3
C O H
H
(CH
3
)
3
C O H
H
H O H
+
(CH
3
)
3
C O H + H
3
O
+
=>
• Loss of H
+
(if needed)
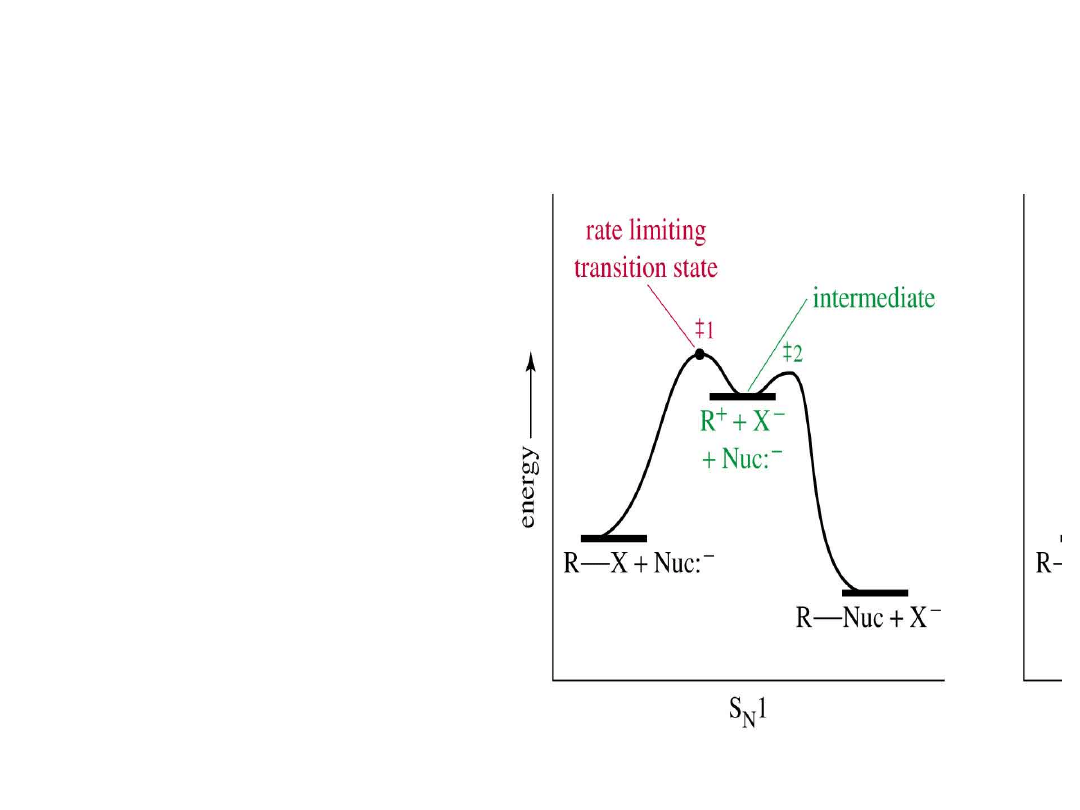
Alkyl Halides
49
S
N
1 Energy Diagram
• Forming the
carbocation is
endothermic
• Carbocation
intermediate is in
an energy well.
=>

Alkyl Halides
50
7_106.exe
S
N
1 with hindrance
7_105.exe
S
N
1 and S
N
2 reactions of alcohols with acids
7_107.exe
Racemic from S
N
1

Alkyl Halides
51
Rates of S
N
1 Reactions
• 3° > 2° > 1° >> CH
3
X
Order follows stability of carbocations
(opposite to S
N
2)
More stable ion requires less energy to
form
• Better leaving group, faster reaction (like
S
N
2)
• Polar protic solvent best: It solvates ions
strongly with hydrogen bonding.
=>
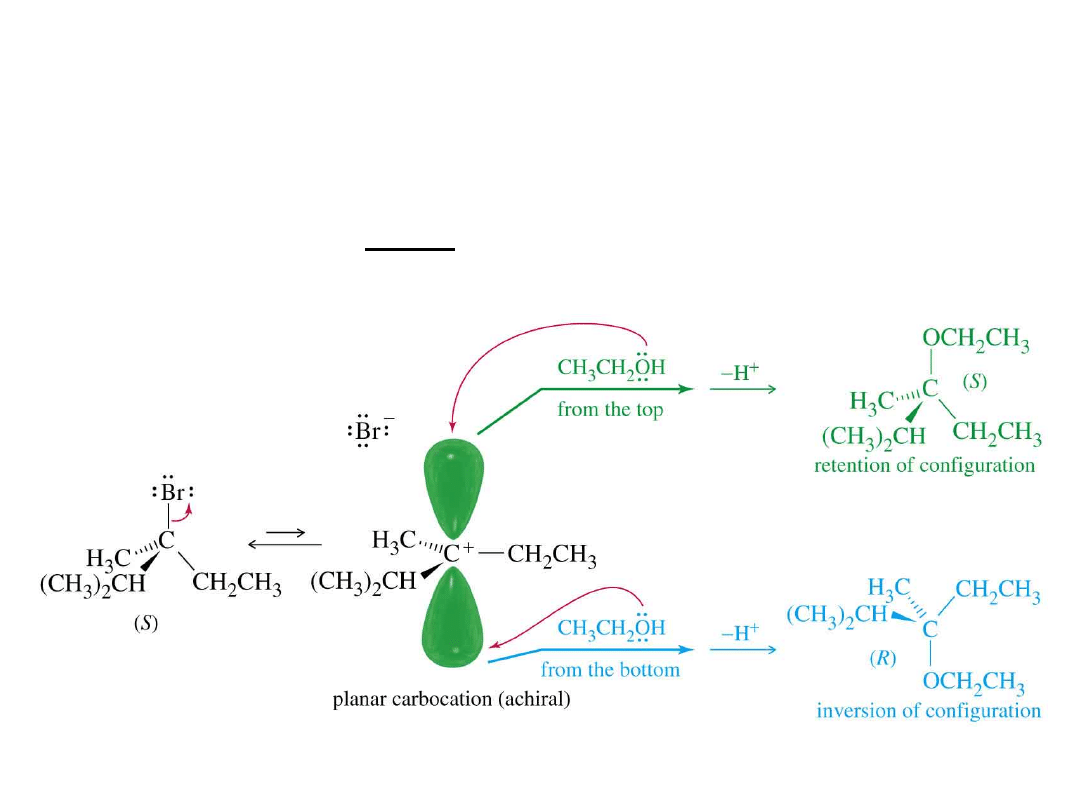
Alkyl Halides
52
Stereochemistry of S
N
1
Racemization:
inversion and retention
=>

Alkyl Halides
53
Rearrangements
• Carbocations can rearrange to form a
more stable carbocation.
• Hydride shift: H
-
on adjacent carbon
bonds with C
+
.
• Methyl shift: CH
3-
moves from adjacent
carbon if no H’s are available.
=>
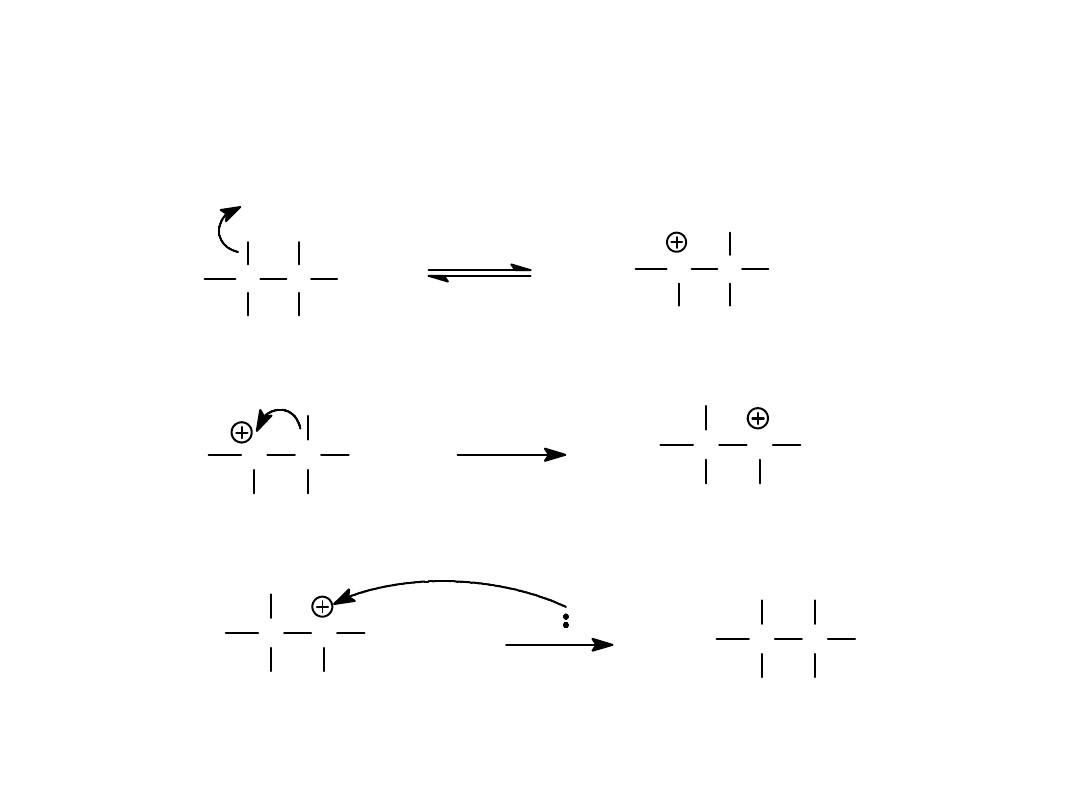
Alkyl Halides
54
Hydride Shift
CH
3
C
Br
H
C
H
CH
3
CH
3
CH
3
C
H
C
H
CH
3
CH
3
CH
3
C
H
C
H
CH
3
CH
3
CH
3
C
H
C
CH
3
CH
3
H
CH
3
C
H
C
CH
3
CH
3
H
Nuc
CH
3
C
H
C
CH
3
CH
3
H Nuc
=>
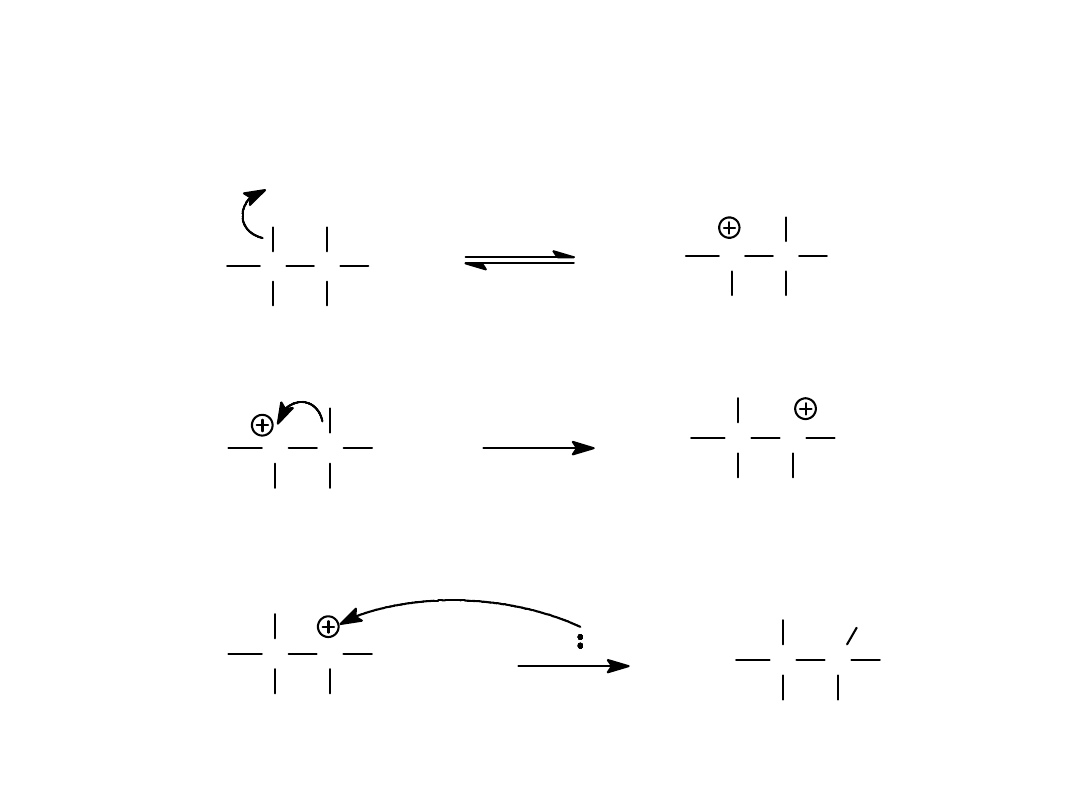
Alkyl Halides
55
Methyl Shift
CH
3
C
Br
H
C
CH
3
CH
3
CH
3
CH
3
C
H
C
CH
3
CH
3
CH
3
CH
3
C
H
C
CH
3
CH
3
CH
3
CH
3
C
H
C
CH
3
CH
3
CH
3
CH
3
C
H
C
CH
3
CH
3
CH
3
Nuc
CH
3
C
H
C
CH
3
CH
3
CH
3
Nuc
=>

Alkyl Halides
56
S
N
2 or S
N
1?
• Primary or methyl
• Strong nucleophile
• Polar aprotic
solvent
• Rate = k[halide]
[Nuc]
• Inversion at chiral
carbon
• No rearrangements
• Tertiary
• Weak nucleophile (may
also be solvent
)
• Polar protic solvent,
silver salts
• Rate = k[halide]
• Racemization of
optically active
compound
• Rearranged products
=>

Alkyl Halides
57
E1 Reaction
• Unimolecular elimination
• Two groups lost (usually X
-
and H
+
)
• Nucleophile acts as base
• Also have S
N
1 products (mixture)
=>
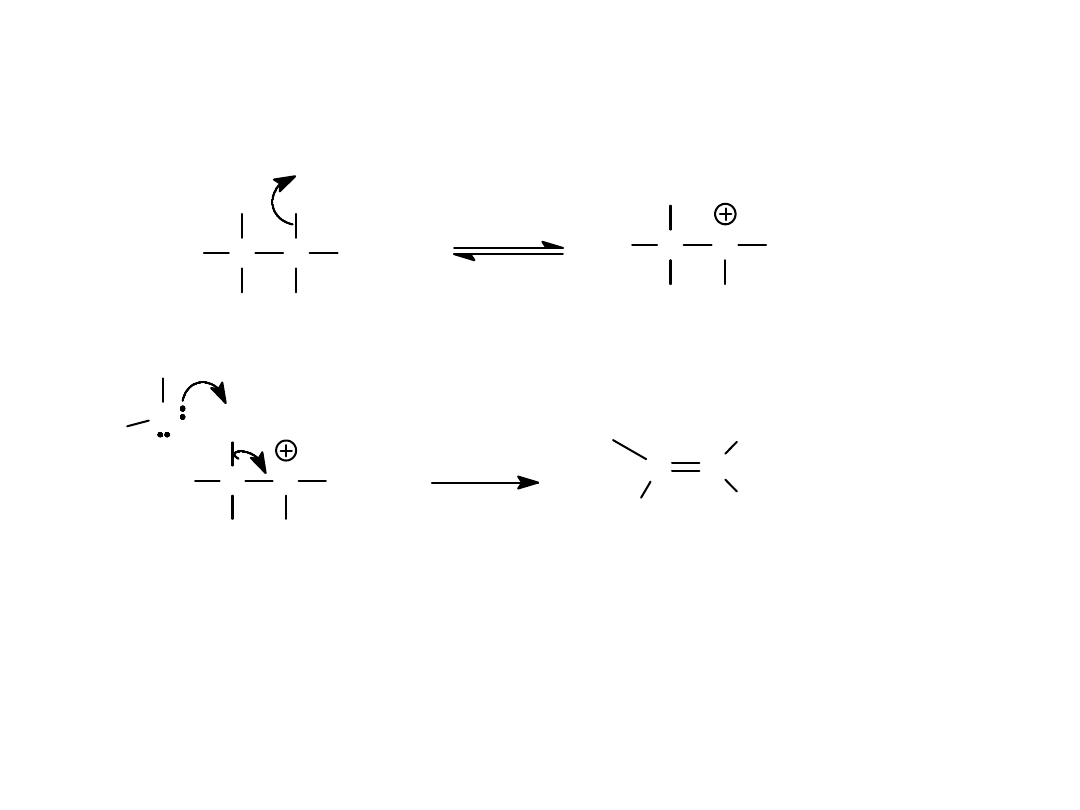
Alkyl Halides
58
E1 Mechanism
• Halide ion leaves, forming carbocation.
• Base removes H
+
from adjacent carbon.
• Pi bond forms. =>
H C
H
H
C
CH
3
CH
3
Br
C
H
H
H
C CH
3
CH
3
O
H
H
C
H
H
H
C CH
3
CH
3
C C
H
CH
3
CH
3
H
+ H
3
O
+
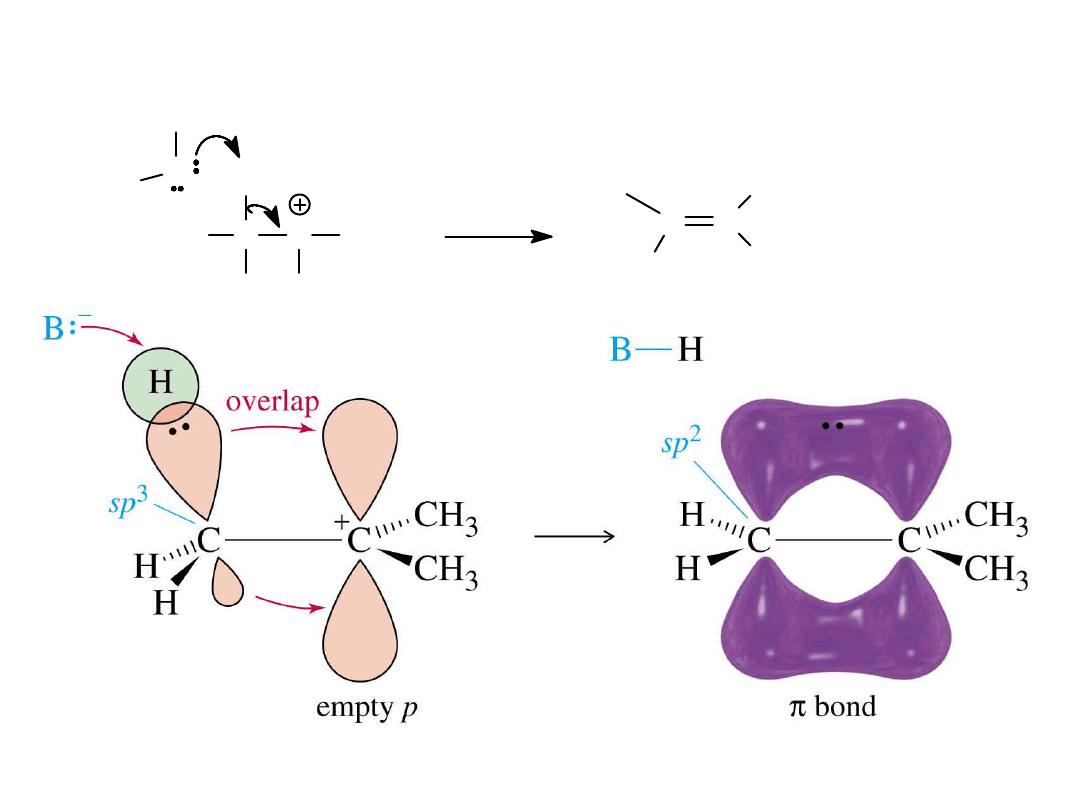
Alkyl Halides
59
A Closer Look
O
H
H
C
H
H
H
C CH
3
CH
3
C C
H
CH
3
CH
3
H
+ H
3
O
+
=>
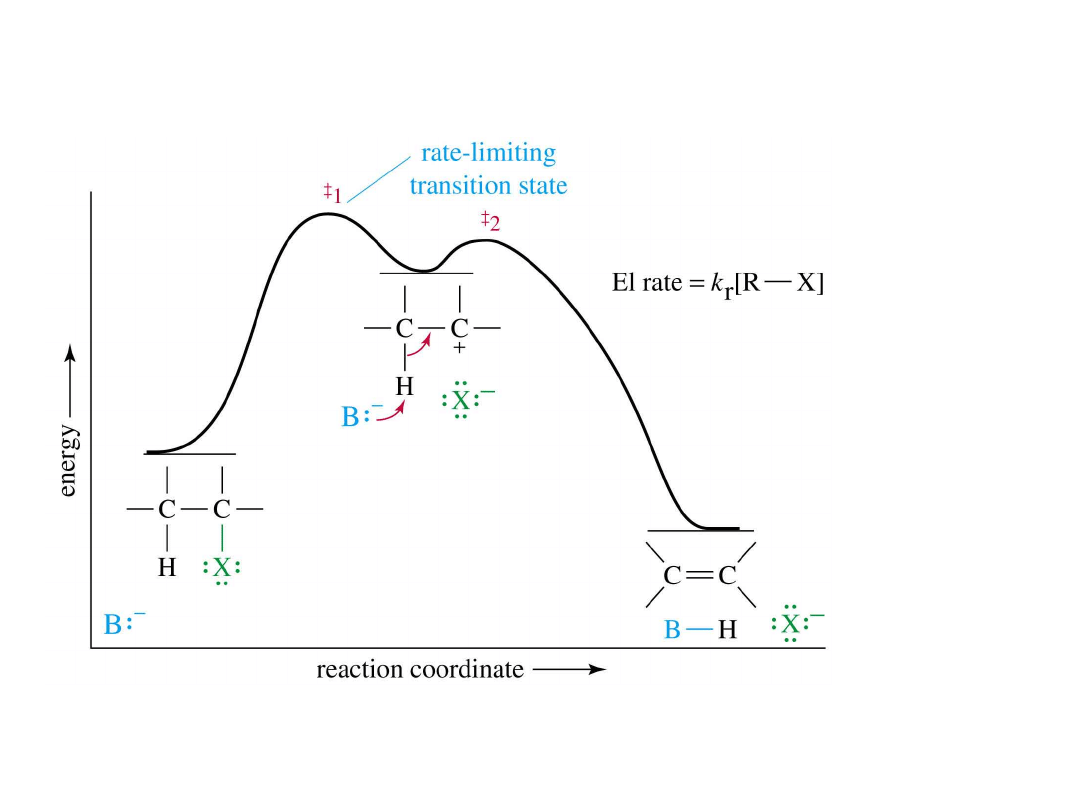
Alkyl Halides
60
E1 Energy Diagram
• Note: first step is same as S
N
1
=
>

Alkyl Halides
61
E2 Reaction
• Bimolecular
elimination
• Requires a
strong base
• Halide leaving and proton
abstraction happens
simultaneously - no intermediate.
=>
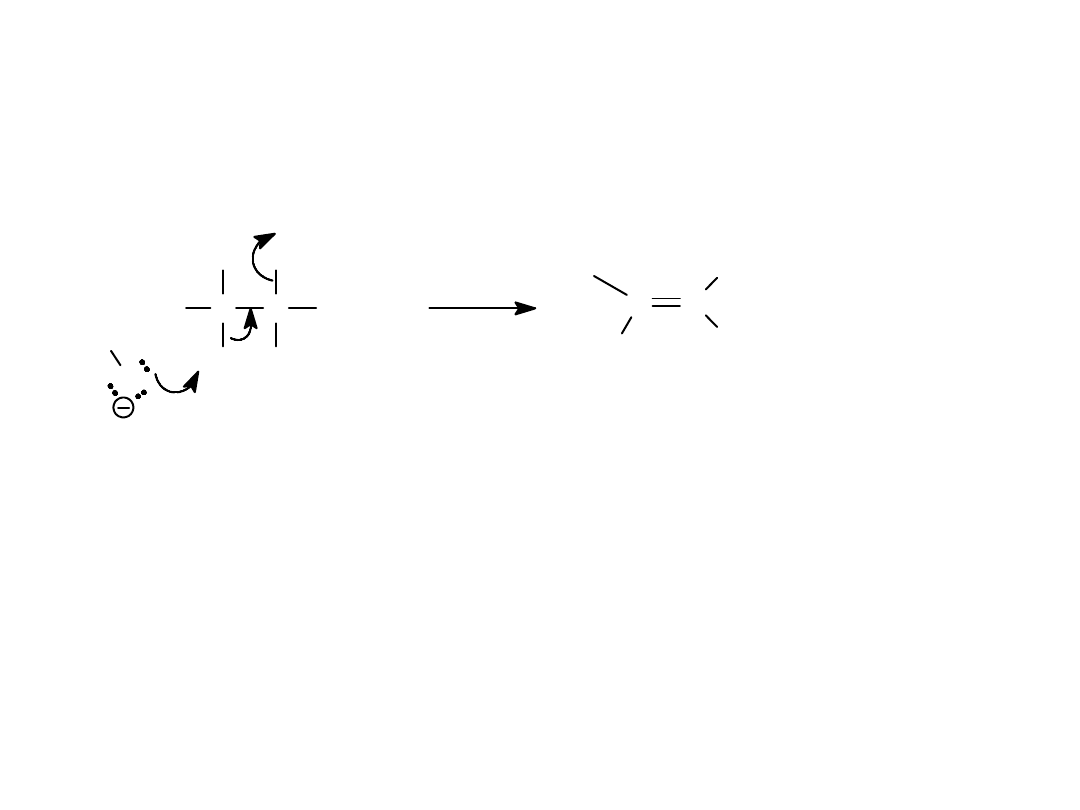
Alkyl Halides
62
E2 Mechanism
H C
H
H
C
CH
3
CH
3
Br
C C
H
CH
3
CH
3
H
O
H
+ H
2
O B
r
-
+
=
>
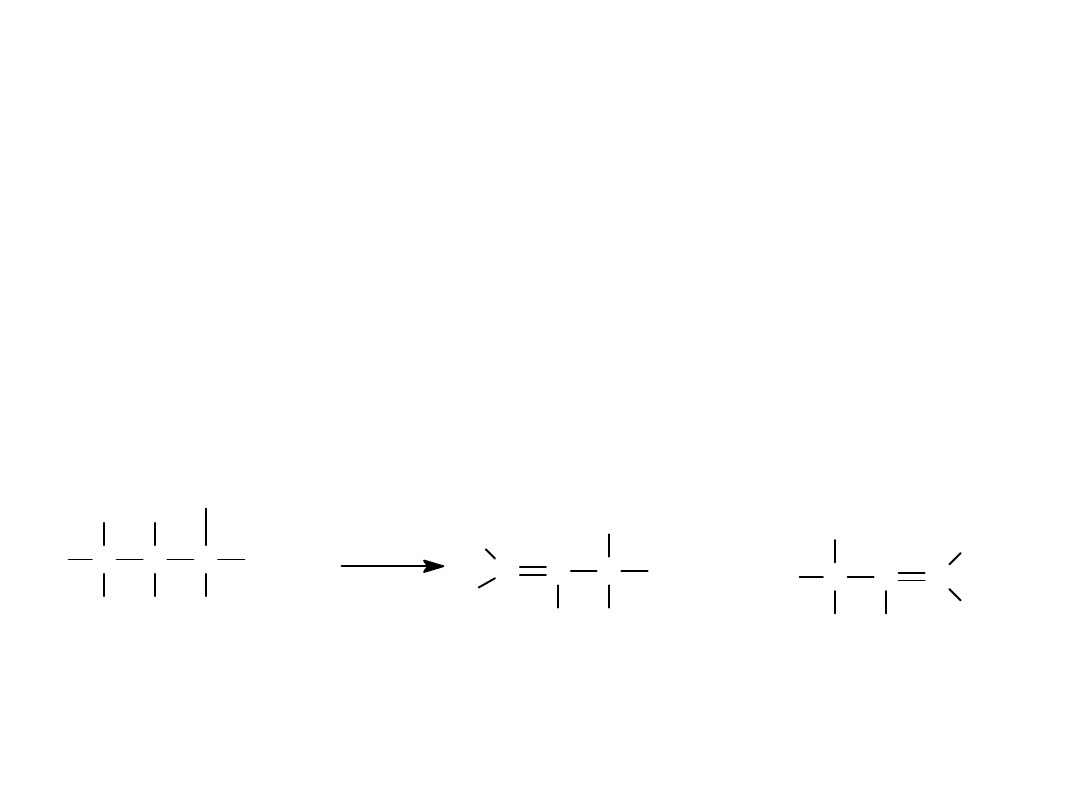
Alkyl Halides
63
Saytzeff’s Rule
• If more than one elimination product is
possible, the
most-substituted
alkene is the
major product (most stable).
• R
2
C=CR
2
>R
2
C=CHR>RHC=CHR>H
2
C=CHR
tetra > tri > di > mono
C C
Br
H
C
H
CH
3
H
H
H
CH
3
OH
-
C C
H
H
C
H H
CH
3
CH
3
C
H
H
H
C
H
C
CH
3
CH
3
+
=>
minor
minor
major
major
Alkyl Halides
64
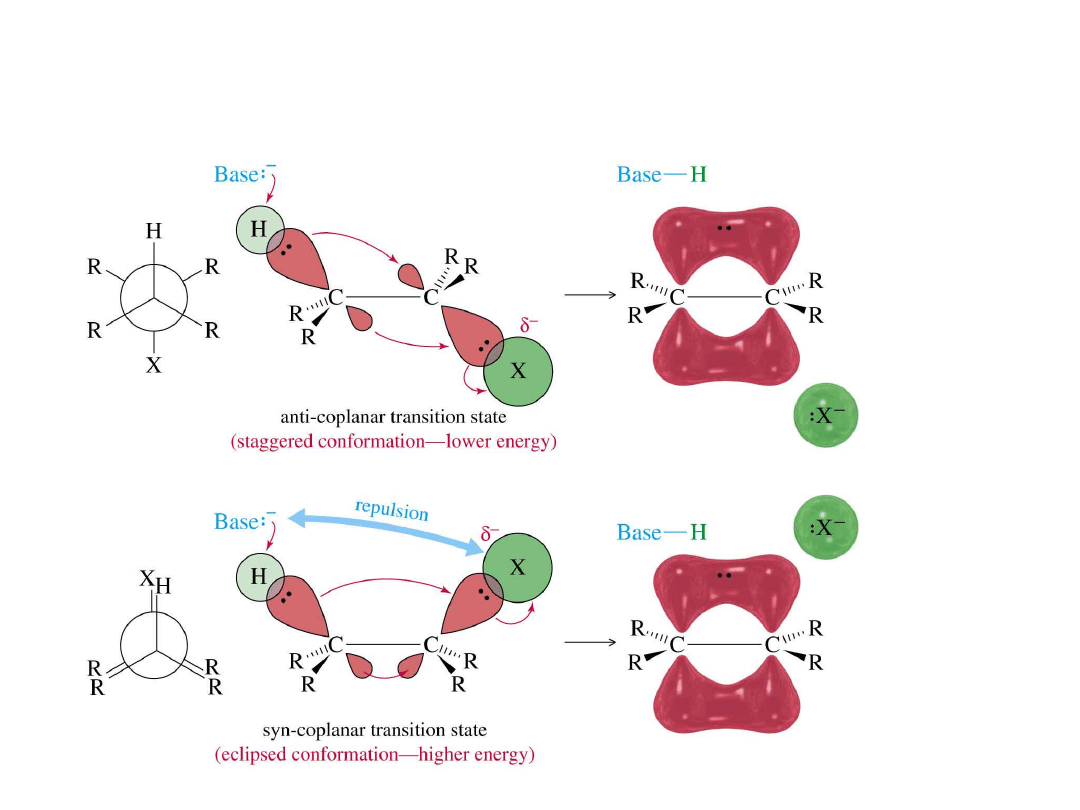
E2 Stereochemistry
=>

Alkyl Halides
65
E1 or E2?
• Tertiary > Secondary
• Weak base
• Good ionizing
solvent
• Rate = k[halide]
• Saytzeff product
• No required
geometry
• Rearranged products
• Tertiary > Secondary
• Strong base required
• Solvent polarity not
important
• Rate = k[halide][base]
• Saytzeff product
• Coplanar leaving
groups (usually anti)
• No rearrangements
=>

Alkyl Halides
66
Substitution or Elimination?
• Strength
of the nucleophile determines
order: Strong nuc. will go S
N
2 or E2.
• Primary
halide
usually S
N
2.
• Tertiary halide
mixture of S
N
1, E1 or E2
• High
temperature
favors elimination.
• Bulky
bases
favor elimination.
• Good nucleophiles, but weak bases,
favor substitution. =>
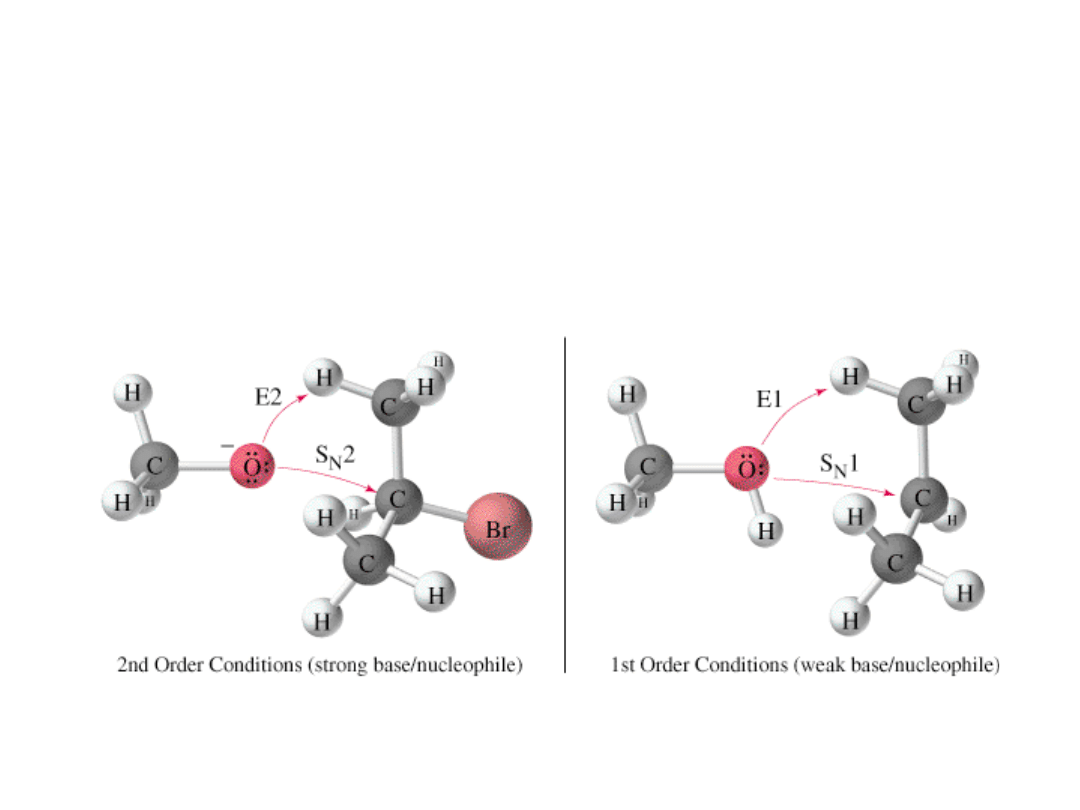
Alkyl Halides
67
Secondary Halides?
Mixtures of products are common.
=
>

Alkyl Halides
68
End of Unit 5
Document Outline
- Unit 5: Chapter 6 Alkyl Halides: Nucleophilic Substitution and Elimination
- Classes of Halides
- Polarity and Reactivity
- Classes of Alkyl Halides
- Classify These:
- Dihalides
- IUPAC Nomenclature
- Systematic Common Names
- “Trivial” Names
- Uses of Alkyl Halides
- Dipole Moments
- Boiling Points
- Densities
- Preparation of RX
- Chlorination of Methane
- Free-Radical Chain Reaction
- Initiation Step
- Propagation Step (1)
- Propagation Step (2)
- Overall Reaction
- Termination Steps
- Halogenation of Alkanes
- Allylic Halogenation
- Reaction Mechanism
- Chlorination of Propane
- Free Radical Stabilities
- Substitution Reactions
- Elimination Reactions
- SN2 Mechanism
- SN2 Energy Diagram
- PowerPoint Presentation
- Slide 32
- Steric Hindrance
- Uses for SN2 Reactions
- SN2: Nucleophilic Strength
- Trends in Nuc. Strength
- Polarizability Effect
- Bulky Nucleophiles
- Solvent Effects (1)
- Solvent Effects (2)
- Crown Ethers
- SN2: Reactivity of Substrate
- Leaving Group Ability
- Structure of Substrate
- Stereochemistry of SN2
- SN1 Reaction
- SN1 Mechanism (1)
- SN1 Mechanism (2)
- SN1 Energy Diagram
- Slide 50
- Rates of SN1 Reactions
- Stereochemistry of SN1
- Rearrangements
- Hydride Shift
- Methyl Shift
- SN2 or SN1?
- E1 Reaction
- E1 Mechanism
- A Closer Look
- E1 Energy Diagram
- E2 Reaction
- E2 Mechanism
- Saytzeff’s Rule
- E2 Stereochemistry
- E1 or E2?
- Substitution or Elimination?
- Secondary Halides?
- Slide 68
Wyszukiwarka
Podobne podstrony:
reguly ustalania nazw systematycznych dla alkanow oraz halogenoalkanow ppt
kwasy halogenokarboksylowe ppt
03 Sejsmika04 plytkieid 4624 ppt
Choroby układu nerwowego ppt
10 Metody otrzymywania zwierzat transgenicznychid 10950 ppt
10 dźwigniaid 10541 ppt
03 Odświeżanie pamięci DRAMid 4244 ppt
Prelekcja2 ppt
2008 XIIbid 26568 ppt
WYC4 PPT
rysunek rodziny ppt
więcej podobnych podstron